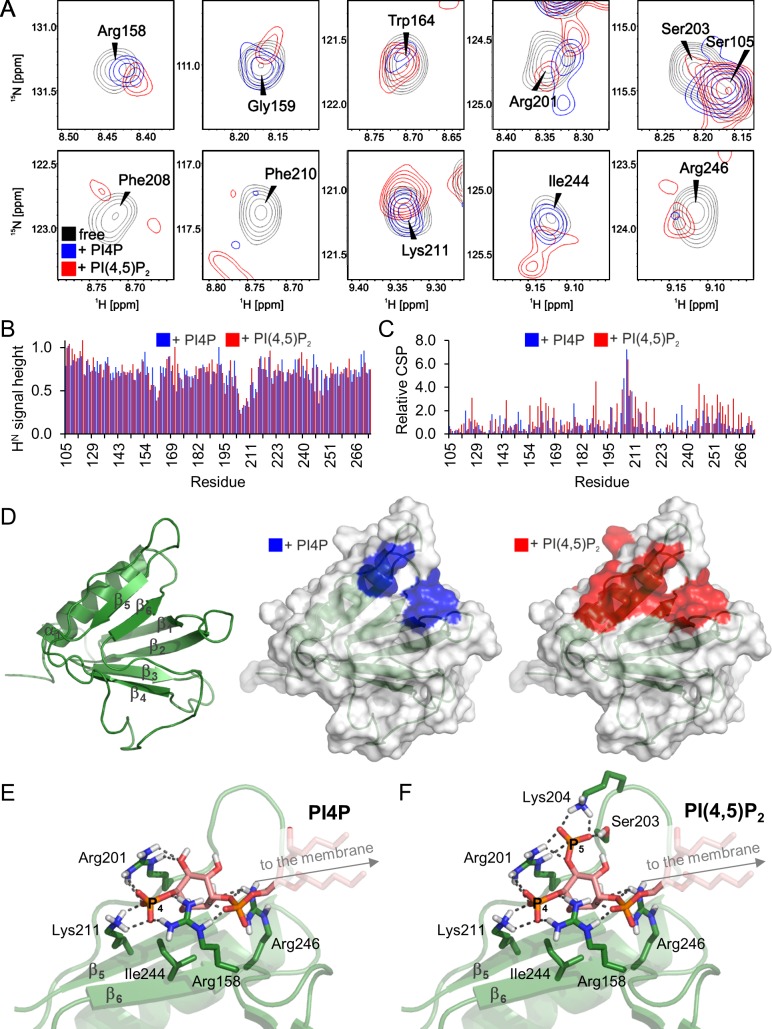
Figure 4.
Both PI4P and PI(4,5)P2 bind to the PH domain of ORP8. (A) The interaction with PI4P/PI(4,5)P2 induced specific changes in the NMR spectra of ORP8. (B and C) We observed both significant reduction of signal intensity (B) and changes in positions of signals (C) in the 2D 15N/1H HSQC spectra of 15N-labeled ORP8 PH domain upon the addition of the ligand. The graphs represent simple differences in signal positions and intensities between the free and ligand-bound protein obtained from a single experiment. (D) The PH domain adopts the canonical fold, and both PI4P and PI(4,5)P2 bind to the same interface as suggested by highlighting the relative changes observed in the NMR spectra on the 3D protein structure. (E and F) The NMR-data driven models of the PH/PI4P (E) and PH/PI(4,5)P2 (F) complexes suggest that the phosphate groups of PI4P/PI(4,5)P2 are stabilized by an extensive network of electrostatic interactions with the positively charged side chains of Arg158, Arg201, Lys211, and Arg246. PI(4,5)P2 is additionally stabilized by an interaction with Lys204 and Ser203.