Abstract
Background
Asthma is the most common chronic disease during pregnancy and it may have influence on pregnancy outcome.
Objectives
Our goal was to assess the association between maternal asthma and the perinatal risks as well as possible effects of asthma medication.
Methods
The study was based on a nationwide Finnish register-based cohort between the years 1996 and 2012 in the Drug and Pregnancy Database. The register data comprised 962 405 singleton live and stillbirths, 898 333 (93.3%) pregnancies in mothers with neither confirmed asthma nor use of asthma medication (controls), and 26 674 (2.8%) pregnancies with confirmed maternal asthma. 71% of mothers with asthma used asthma medication. The diagnosis of asthma was based on the mothers’ right for subsidised medication which is carefully evaluated by strict criteria including pulmonary function testing. Odds ratio was used in comparison. Premature birth (PB), low birth weight, small for gestational age (SGA), neonatal death were the main outcome measures.
Results
Maternal asthma was associated with adjusted odds ratios (aORs) for perinatal mortality 1.24 (95% CI 1.05 to 1.46), preterm birth 1.18 (1.11 to 1.25), low birth weight 1.29 (1.21 to 1.37), fetal growth restriction (SGA) 1.32, (1.24 to 1.40), and asphyxia 1.09 (1.02 to 1.17). Asthma treatment reduced the increased risk of preterm birth aOR 0.85 (95% CI 0.76 to 0.96) but mothers with treated asthma had higher risks of fetal growth restriction (SGA) aOR 1.26 (1.10 to 1.45), and asphyxia aOR 1.37 (1.17 to 1.61) than mothers with untreated asthma.
Conclusion
Asthma is associated with increased risks of perinatal mortality, preterm birth, low birth weight, fetal growth restriction (SGA), and asphyxia. Asthma treatment reduces the risk of preterm delivery, but it does not seem to reduce other complications such as perinatal mortality.
Introduction
Asthma is the most common chronic disease during pregnancy[1]. In Finland, the prevalence of physician diagnosed asthma has increased from six to ten percent from 1996 to 2006, asthma mortality and hospitalisation rates are low, and asthma treatment results are excellent in general [2,3].
The importance of public health questions on maternal asthma and asthma medication use during pregnancy has inspired several studies. However, previous cohort study findings based on single small cohorts on the relationships between maternal asthma and risk of perinatal complications have been conflicting [4–9]. By combining cohorts by meta-analysis, asthma has been shown to increase the risks of preeclampsia, preterm birth, low birth weight, fetal growth restriction (SGA), neonatal death, and congenital anomalies [10,11]. There are large cohort studies of mothers with asthma: three Swedish cohorts of 24 750, 26 586, and a recent cohort of 108 225, one US of 17,044, and one Canadian cohort of 13 100 mothers with asthma [4,12–16]. All these cohorts confirmed the association between maternal asthma and the risks of preterm birth and SGA. In addition, the Canadian cohort also showed an association between maternal asthma and increased perinatal mortality. Maternal asthma has been associated also with several mild neonatal complications such as hyperbilirubinemia and transient respiratory problems [10,11,14,17,18]. Based on small clinical studies, improved asthma management is suggested to decrease the asthma related increased perinatal risks[19–21].
We studied pregnancy complications in mothers with asthma in Finland based on complete, high quality registry data[22]. The data were acquired from the Drugs and Pregnancy database, which combines information from Finnish health registers. Our aim was to investigate the pregnancy complication risks associated with asthma and asthma medication.
Methods
This is a national, population register based, cohort investigation including information on all the singleton live and stillbirths in Finland between 1996 and 2012. The data were acquired from the Finnish Drugs and Pregnancy database [23]. This database is a collaboration by THL National Institute for Health and Welfare, KELA Social Insurance Institution, and FIMEA Finnish Medicines Agency. The database contains medical and sociodemographic information on parents, pregnancy and perinatal outcomes, and reimbursed medication purchases. The linkage of registers has been approved by the data keepers (THL and KELA). An ethical committee has approved the formation of this database, and its utilisation in research studies of general interest. Since the registered women were not contacted, the ethical committee did not require informed consent from the study population. Each citizen and permanent resident of Finland has a personal identity code enabling data linkage between registers. All data were fully anonymized before analysis. Reporting to the official healthcare registers by public and private hospitals, and primary health care is mandatory by law.
The Medical Birth Register (MBR) was used to identify all the pregnancies and information on the outcomes. The registry data is recorded at the time of birth, and include all the live births and stillbirths (≥22 weeks of pregnancy or birth weight ≥500 grams).
The National Drug Reimbursement Register (DRR) contains individual-level data on reimbursed prescription medication purchases and data on the reimbursement for discounted medication for a restricted number of diseases. In Finland, all of the included asthma medications require a prescription for purchase. For asthma, the right for subsidised medication is granted according to strict criteria which include asthma symptoms, six months of regular medication use, and a reversible airway obstruction diagnostic for asthma in pulmonary function testing (Table 1). Once a person fulfils the criteria they are offered a right for subsidised medication, which is permanent for applicants over 16 years of age. In this study, the right for subsidised asthma medication indicates confirmed asthma. Asthma medication use was defined as asthma medication purchases during the period between three months prior the pregnancy and the end of the pregnancy. The three month period before the pregnancy was selected, because in Finland, the pharmacies are entitled to sell medications for up to three months at one time.
Table 1. The criteria used for the right for justification of reimbursement.
For reimbursement, a person has to fulfil criteria in all three categories: symptoms, pulmonary function testing and medication purchase. Once a person fulfils the criteria they are offered reimbursement, which is permanent for applicants over 16 years of age.
| Physicians certificate–general requirements |
| Typical development and clinical picture of asthma |
| Possible indicators of eosinophilic inflammation |
| Results of allergy test, pulmonary function tests and bronchial challenge tests |
| Persistent and ongoing symptoms of asthma for at least six months |
| Need for a controller medication for at least six months |
| Pulmonary function testing–at least one of the following: |
| Reversible obstruction during PEF monitoring of two weeks at least three times: daily variation of at least 20% and 60 l/min or improvement of PEF at least 15% and 60 l/min after bronchodilative medicine |
| In flow volume spirometry, an improvement of at least 12% and 200ml in FEV1 or FVC |
| After inhaled glucocorticoid treatment an increase of at least 15% and 200ml in FEV1 or increase in PEF level of 20% |
| Moderate or severe hyper responsiveness in bronchial challenge test to histamine or methacholine |
| A decrease of 15% or more in PEF or FEV1 in exercise test |
| Medication purchases |
| Regular use of asthma controller medication for at least six months from ATC-group R03AK (inhaled combination of β2 agonists and glucocorticoids) or R03BA (inhaled glucocorticoids) or (R03DC) leucotrienantagonist indicated by medication purchases is required |
PEF = peak expiratory flow, FEV1 = forced expiratory volume in one second, FVC forced vital capacity
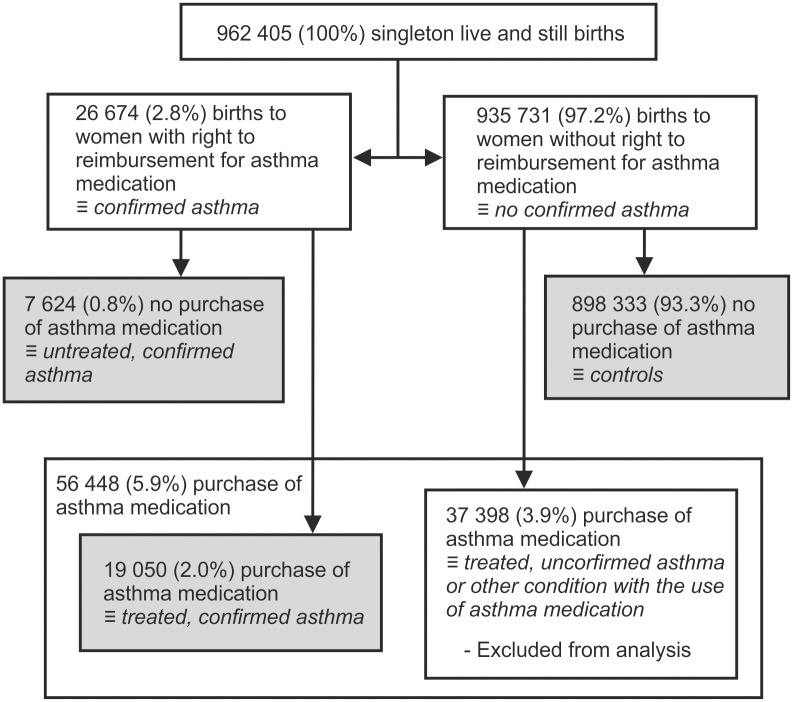
For comparisons, we identified three groups of mothers: 1) controls as mothers with neither confirmed asthma nor current use of asthma medication, 2) untreated asthma as mothers with confirmed asthma but no use of asthma medication, and 3) treated asthma as mothers with confirmed asthma and use of asthma medication (Fig 1). Also, a fourth group was recognised as mothers without confirmed asthma but use of asthma medication. This group was excluded from analysis (Fig 1).
Fig 1. Demographics of the 962 405 singleton live and stillbirths between 1996 and 2012.
The separation into different groups.
For study purposes, the Anatomical Therapeutic Chemical (ATC) grouping was used, and the following categories were identified: inhaled short- and long-acting β2 agonists (R03AC), inhaled glucocorticoids (R03BA), inhaled combination of β2 agonists and glucocorticoids (R03AK), inhaled short- and long-acting anticholinergics (R03BB), xanthines (R03DA), leukotriene receptor antagonists (R03DC), and oral glucocorticoids (H02AB).
The primary outcomes of this study were preterm birth (<37 weeks), low birth weight (<2500 grams), perinatal mortality (stillbirths and early neonatal deaths), and small for gestational age (defined by population based sex-specific references).[24] In perinatology, the length of the pregnancy and perinatal mortality are the most commonly used core outcomes of pregnancy.[25]
The secondary outcomes were umbilical cord arterial pH <7.10, Apgar score <7, and urgent section. Apgar score is a well-established classification system of newborn health. The 5 minute Apgar scores were available in the Medical Birth Register since 2004. The asphyxia diagnose is made postnatally from the arterial or venous cord pH and Apgar score, or prenatally from cardiotocogram. The information regarding urgent sections was selected as an outcome measure to represent an observed consequence of the fetal distress and asphyxia. Urgent section takes place within 30 minutes on section decision.
The used confounding factors were maternal age, smoking while pregnant, socioeconomic status based on maternal occupation during pregnancy, and parity. As the reported prevalence of epilepsy did not differ between the maternal asthma groups, and as the amount of mothers with type 1 and 2 diabetes was small, these factors were not used for adjusting.
Differences between different exposure groups were evaluated by chi-squared test for dichotomous and categorical variables. Adjusted odds ratios (aOR) and 95% confidence intervals (CI) were estimated to measure all associations, using unconditional logistic regression analysis. Possible confounding factors were selected based on the literature (see above). Individuals with missing outcome variables were excluded from the analysis.
Results
The demographic data and criteria for confirmed asthma diagnosis are presented in Fig 1 and Table 1. Supplemental S1 Table shows characteristics of mothers in detail. The study cohort included of 962 405 live and stillbirths, 898 333 (93.3%) control mothers and 26 674 (2.8%) mothers with confirmed asthma, and 5 292 perinatal deaths. For subgroup analysis, asthmatic mothers were subdivided into 19 059 (2.0%) mothers with treated confirmed asthma, and 7 624 (0.8%) mothers with untreated confirmed asthma. Of the mothers with treated confirmed asthma, 64.6% used at least two asthma medications belonging to separate ATC-groups.
The main study results are presented in Table 2, and S2 and S3 Tables. S3 Table represents all adjusted and unadjusted odds ratios. Compared to control pregnancies, the infants born to mothers with asthma showed increased risk of perinatal mortality, preterm birth, low birth weight, fetal growth restriction (SGA), umbilical arterial pH below 7.1, Apgar score below 7 at 1 minutes and 5 minutes of age, delivery by urgent section, and birth asphyxia. In a subgroup analysis comparing pregnancies between mothers with asthma, asthma treatment reduces the increased risk of preterm birth. However, mothers with treated asthma had higher risks of fetal growth restriction (SGA) and asphyxia than mothers with untreated asthma.
Table 2. Perinatal outcomes in mothers with confirmed, treated or untreated asthma for singleton live and stillbirths.
| Confirmed asthma vs control | Treated vs. untreated. confirmed asthma | Untreated. confirmed asthma vs control | Treated. confirmed asthma vs. control | |||||
|---|---|---|---|---|---|---|---|---|
| aOR* (95% CI) | P | aOR* (95% CI) | P | aOR* (95% CI) | P | aOR* (95% CI) | P | |
| Perinatal mortality | 1.26 (1.07, 1.48) | 0.0067 | 0.93 (0.66, 1.32) | 0.6903 | 1.33 (1.00, 1.79) | 0.0533 | 1.22 (1.01, 1.49) | 0.0447 |
| Premature birth | 1.18 (1.12, 1.25) | <0.0001 | 0.87 (0.77, 0.98) | 0.0210 | 1.31 (1.19, 1.45) | <0.0001 | 1.13 (1.05, 1.21) | 0.0005 |
| Low birth weight | 1.29 (1.21, 1.37) | <0.0001 | 1.12 (0.97, 1.29) | 0.1144 | 1.19 (1.05, 1.34) | 0.0058 | 1.33 (1.23, 1.43) | <0.0001 |
| SGA | 1.32 (1.24, 1.40) | <0.0001 | 1.25 (1.09, 1.44) | 0.0015 | 1.12 (0.99, 1.26) | 0.0691 | 1.39 (1.30, 1.49) | <0.0001 |
| Umbilical arterial pH < 7.1 | 1.17 (1.08, 1.26) | <0.0001 | 1.10 (0.92, 1.31) | 0.3027 | 1.10 (0.94, 1.28) | 0.2449 | 1.20 (1.09, 1.31) | 0.0001 |
| 1 min Apgar score 0 to 6 | 1.26 (1.19, 1.32) | <0.0001 | 1.01 (0.91, 1.14) | 0.8172 | 1.24 (1.13, 1.36) | <0.0001 | 1.26 (1.19, 1.34) | <0.0001 |
| 5 min Apgar score 0 to 6 | 1.21 (1.09, 1.35) | 0.0005 | 1.05 (0.82, 1.35) | 0.6829 | 1.15 (0.93, 1.43) | 0.1934 | 1.23 (1.09, 1.40) | 0.0009 |
| Urgent section | 1.23 (1.18, 1.28) | <0.0001 | 1.12 (1.03, 1.23) | 0.0119 | 1.13 (1.05, 1.23) | 0.0019 | 1.27 (1.21, 1.33) | <0.0001 |
| Asphyxia | 1.07 (1.00, 1.14) | 0.0454 | 1.32 (1.12, 1.55) | 0.0007 | 0.88 (0.77, 1.01) | 0.0774 | 1.14 (1.06, 1.23) | 0.0007 |
* Adjusted for maternal age, parity, smoking, socio-economic status, year of birth
Table 3 and S4 Table present risk factors associated with each medication type and their combinations in pregnancies. The use of short-acting β2 agonists was associated with increased perinatal mortality, but on the other hand, 71% of mothers with treated asthma used these short-acting β2 agonists, and the increased risk of perinatal mortality could be a result of the underlying disease. The number of used asthma medications increased the risk of low birth weight and fetal growth restriction in logistic regression analysis on 0, 1, 2, and 3 or more medications. Each purchased medication group increased the risk of low birth weight by 6% aOR 1.06 (95% CI 1.00 to 1.13, P = 0 .0356), and fetal growth restriction (SGA) by 13% aOR 1.13 (95% CI 1.07 to 1.19, P < 0.0001). However, the risk of perinatal mortality or preterm birth did not increase in logistic manner.
Table 3. Perinatal outcomes of treated, confirmed asthma by ATC-groups compared to controls for singleton live and stillbirths.
| Perinatal mortality | Premature birth | Low birth weight | SGA | |||||
|---|---|---|---|---|---|---|---|---|
| aOR* (95% CI) | P | aOR* (95% CI) | P | aOR* (95% CI) | P | aOR* (95% CI) | P | |
| Any asthmamedication | 1.22 (1.01, 1.49) | 0.0447 | 1.13 (1.05, 1.21) | 0.0005 | 1.33 (1.23, 1.43) | <0.0001 | 1.39 (1.30, 1.49) | <0.0001 |
| No asthma medication | 1.33 (1.00, 1.79) | 0.0533 | 1.31 (1.19, 1.45) | <0.0001 | 1.19 (1.05, 1.34) | 0.006 | 1.12 (0.99, 1.26) | 0.069 |
| One asthma medication | 1.29 (0.92, 1.80) | 0.1477 | 1.19 (1.05, 1.33) | 0.0048 | 1.21 (1.06, 1.39) | 0.004 | 1.19 (1.05, 1.36) | 0.009 |
| Two asthma medications | 1.23 (0.92, 1.64) | 0.1601 | 1.06 (0.96, 1.18) | 0.2557 | 1.37 (1.24, 1.52) | <0.0001 | 1.46 (1.32, 1.61) | <0.0001 |
| Three or more asthma medications | 1.12 (0.74, 1.71) | 0.5859 | 1.18 (1.03, 1.36) | 0.0168 | 1.39 (1.20, 1.60) | <0.0001 | 1.53 (1.33, 1.75) | <0.0001 |
* Adjusted for maternal age, parity, smoking, socio-economic status, year of birth
Discussion
Main findings
This comprehensive register based study shows that confirmed maternal asthma is associated with the risk of perinatal mortality with the adjusted odds ratio of 1.24 (95% CI 1.05 to 1.46). In addition, like previously shown, maternal asthma is associated with increased risks of preterm birth, low birth weight, fetal growth restriction (SGA), and asphyxia. When comparing treated and untreated group of mothers with asthma, there was no significant difference in perinatal mortality between the groups, but asthma treatment decreased the increased risk of preterm birth. However, the risks of SGA and asphyxia remained higher in mothers with treated asthma.
Strengths and limitations
The major strengths of this study are national coverage of the two obligatory registers, the large size of the study cohort, the strict criteria applied for the diagnosis of persistent asthma, the exclusion of the use of any asthma medication in the group of control mothers, and possibility to control several confounding factors such as parity, maternal age, socioeconomic status, self-reported smoking, and epilepsy.[22] In this study, we have been able to cover some of the possible weaknesses of previous three published large cohort studies on this subject. The Swedish study by Källén and associates was based on self-reported use of asthma medication on maternity care visit between 10 and 12 weeks of pregnancy [4,17,26]. The US cohort was based on asthma diagnosis (ICD-9) in patient records, in well-selected hospitals across US and contained no information on use of asthma medications [14,15,27]. The Canadian cohort was based on purchases of asthma medication of substantial but not whole population of one province in Canada [16].
Our cohort included 26 674 (2.8%) children born to mothers with confirmed asthma, and 71% of these mothers used asthma medication. We estimated the use of medication through the medicine reimbursement registry with full coverage of medications purchased in Finland. Although not confirmed, we assume that asthma medication purchase reliably correlates to the real use of medication, and thus serves well for the study purposes.
The limitations of this study are that from this registry data, we were unable to separate different asthma phenotypes such as allergic and non-allergic asthma or dosing of medication. Like none of the cohort studies so far, we were not able to control the severity of maternal asthma through pulmonary function testing or internationally validated test of asthma control [28].
The data on asthma diagnosis was based on reimbursement records of the national Drug Reimbursement Register. The criteria for the right for subsidised medication are a key element when interpreting our results (Table 1). The use of strict criteria through medical reimbursement guidelines imply that a significant number of mothers with some degree of asthma were not included in the group of treated confirmed asthma. The results that 37 398 (3.9%) of mothers had purchased asthma medications without the right for subsidised asthma medication support this assumption. These mothers were likely to have asthma symptoms but had either no objective verification of airway reversibility or the need for previous regular asthma medication for more than six months. The exclusion of mothers with intermittent asthma medication use made it possible to compare the fetal effects of asthma itself irrespective of asthma medication. The criteria for asthma used in Finland are according to international asthma guidelines, making the study generalizable [29–31].
Interpretation
Since asthma symptoms may vary over time, we assumed that most mothers with untreated asthma were currently asymptomatic, and therefore, without purchases of asthma medication despite previously diagnosed asthma. However, we cannot exclude poor adherence to asthma treatment. Nevertheless, we suggest that the observed association with the risk of perinatal mortality in this group is caused by the asthma process itself.
In the meta-analysis by Murphy and associates, maternal asthma increased the risk of neonatal death [10]. In the single large Canadian cohort, maternal asthma increased the risk of perinatal mortality in general, but this correlation vanished after adjustment for preterm birth and SGA [16]. These US and Canadian cohorts were not able to differentiate effects of asthma treatment on the risk analysis. Källén and associates showed in the Swedish cohort of 24 369 births to mothers with asthma, that the use of three or more asthma medications indicating severe asthma increased the risk of perinatal mortality [4]. The Swedish cohort was not able to differentiate untreated asthma or oral glucocorticoid treatment, and they did not have coverage of medication prescriptions for the whole pregnancy. In our cohort, unlike in the study of Källén, the use of increasing number of asthma medications did not significantly increase the risk of perinatal mortality [17].
Our data showed that asthma medication diminishing the risk of preterm birth but not the increased risks of SGA or perinatal death. This observation is supported by Rejnö and associates in newer Swedish cohorts [12,13].
According to the current asthma guidelines, asthma medications are recommended to be used in a stepwise manner [29–31]. The number of used asthma medications or the use of did not correlate with the increased risk of perinatal mortality or preterm birth but it did correlate with low birth weight and SGA. We suggest that these correlations with low birth weight and SGA reflect the severity of asthma more than the drug effect. Schatz and associates showed, that the severity of asthma during pregnancy determined by spirometry adjusted for the use of oral glucocorticoids increased the risk of preterm birth [32]. In the large Canadian cohort reported by Cossette and associates, the use of a combination of low to moderate doses of inhaled corticosteroids and long-acting β2 agonists adjusted for asthma severity did not associate with perinatal risks [33].
In the future, we will analyse the effect of intermittent asthma-medication use, dosage, the use of asthma medication in different trimesters of pregnancy, and consider of combining registry based data with other available information such as medical records.
Conclusion
Asthma is frequently encountered in pregnant women. Asthma is associated with increased risk of perinatal mortality, preterm birth, low birth weight, fetal growth restriction (SGA), and asphyxia. Asthma treatment reduces the increased risk for preterm delivery but it does not seem to reduce the risk of perinatal mortality and other complications. The mechanisms remain elusive. The entire causal pathway from maternal asthma to adverse perinatal outcomes especially growth retardation may not be targeted by current medications for asthma.
Supporting information
The study consisted of 962 405 singleton live and stillbirths in Finland between the years 1996 and 2012.
(DOC)
The study consisted of 962 405 live- and stillborn singletons in Finland between the years 1996 and 2012, Chi-square test between groups.
(DOC)
Unadjusted and adjusted odds ratios.
(DOC)
(DOC)
Data Availability
Because of the national legal restrictions, authors cannot make de-identified dataset available. Data contains confidential patient information for which access is strongly prohibited. An authorisation from the National Institute of Health and Welfare (THL) and Social Insurance Institution (KELA) is necessary for any use of similar confidential data for research purposes. The contact information for a data access committee in THL is kirjaamo@thl.fi and in KELA tietopyynnot@kela.fi.
Funding Statement
Drugs and Pregnancy database is a collaboration by THL National Institute for Health and Welfare, Kela Social Insurance Institution and Fimea Finnish Medicines Agency (main funder). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The author(s) received no specific funding for this work.
References
- 1.Kwon HL, Triche EW, Belanger K, Bracken MB. The epidemiology of asthma during pregnancy: prevalence, diagnosis, and symptoms. Immunol Allergy Clin North Am. 2006;26: 29–62. doi: 10.1016/j.iac.2005.11.002 [DOI] [PubMed] [Google Scholar]
- 2.Kauppi P, Linna M, Martikainen J, Mäkelä MJ, Haahtela T. Follow-up of the Finnish Asthma Programme 2000–2010: reduction of hospital burden needs risk group rethinking. Thorax. 2013;68: 292–293. doi: 10.1136/thoraxjnl-2011-201028 [DOI] [PubMed] [Google Scholar]
- 3.Kainu A, Pallasaho P, Piirilä P, Lindqvist A, Sovijärvi A, Pietinalho A. Increase in prevalence of physician-diagnosed asthma in Helsinki during the Finnish Asthma Programme: improved recognition of asthma in primary care? A cross-sectional cohort study. Prim Care Respir J. 2013;22: 64–71. doi: 10.4104/pcrj.2013.00002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Källén B, Otterblad Olausson P. Use of anti-asthmatic drugs during pregnancy. 1. Maternal characteristics, pregnancy and delivery complications. Eur J Clin Pharmacol. 2007;63: 363–373. doi: 10.1007/s00228-006-0257-1 [DOI] [PubMed] [Google Scholar]
- 5.Bracken MB, Triche EW, Belanger K, Saftlas A, Beckett WS, Leaderer BP. Asthma symptoms, severity, and drug therapy: a prospective study of effects on 2205 pregnancies. Obstet Gynecol. 2003;102: 739–752. [DOI] [PubMed] [Google Scholar]
- 6.Alexander S, Dodds L, Armson BA. Perinatal outcomes in women with asthma during pregnancy. Obstet Gynecol. 1998;92: 435–440. [DOI] [PubMed] [Google Scholar]
- 7.Stenius-Aarniala BS, Hedman J, Teramo KA. Acute asthma during pregnancy. Thorax; 1996;51: 411–414. doi: 10.1136/thx.51.4.411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Perlow JH. Severity of asthma and perinatal outcome. 1992;167: 963–967. [DOI] [PubMed] [Google Scholar]
- 9.Olesen C, Thrane N, Nielsen GL, Sørensen HT, Olsen J, EuroMAP Group. A population-based prescription study of asthma drugs during pregnancy: changing the intensity of asthma therapy and perinatal outcomes. Respiration. 2001;68: 256–261. doi: 10.1159/000050507 [DOI] [PubMed] [Google Scholar]
- 10.Murphy VE, Wang G, Namazy JA, Powell H, Gibson PG, Chambers C, et al. The risk of congenital malformations, perinatal mortality and neonatal hospitalisation among pregnant women with asthma: a systematic review and meta-analysis. BJOG. 2013;120: 812–822. doi: 10.1111/1471-0528.12224 [DOI] [PubMed] [Google Scholar]
- 11.Murphy V. A meta-analysis of adverse perinatal outcomes in women with asthma. BJOG.; 2011;118: 1314–1323. doi: 10.1111/j.1471-0528.2011.03055.x [DOI] [PubMed] [Google Scholar]
- 12.Rejnö G, Lundholm C, Larsson K, Larsson H, Lichtenstein P, D’Onofrio BM, et al. Adverse Pregnancy Outcomes in Asthmatic Women: A Population-Based Family Design Study. J Allergy Clin Immunol Pract. 2017. doi: 10.1016/j.jaip.2017.07.036 [DOI] [PubMed] [Google Scholar]
- 13.Rejnö G, Lundholm C, Gong T, Larsson K, Saltvedt S, Almqvist C. Asthma during pregnancy in a population-based study—pregnancy complications and adverse perinatal outcomes. Laine K, editor. PLoS ONE. 2014;9: e104755 doi: 10.1371/journal.pone.0104755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mendola P, Männistö TI, Leishear K, Reddy UM, Chen Z, Laughon SK. Neonatal health of infants born to mothers with asthma. Journal of Allergy and Clinical Immunology. 2014;133: 85–90.e4. doi: 10.1016/j.jaci.2013.06.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mendola P, Laughon SK, Männistö TI, Leishear K, Reddy UM, Chen Z, et al. Obstetric complications among US women with asthma. Am J Obstet Gynecol. 2013;208: 127.e1–8. doi: 10.1016/j.ajog.2012.11.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Breton M-C, Beauchesne M-F, Lemière C, Rey E, Forget A, Blais L. Risk of perinatal mortality associated with asthma during pregnancy. Thorax. 2009;64: 101–106. doi: 10.1136/thx.2008.102970 [DOI] [PubMed] [Google Scholar]
- 17.Källén B, Otterblad Olausson P. Use of anti-asthmatic drugs during pregnancy. 2. Infant characteristics excluding congenital malformations. Eur J Clin Pharmacol. 2007;63: 375–381. doi: 10.1007/s00228-006-0258-0 [DOI] [PubMed] [Google Scholar]
- 18.Demissie K, Marcella SW, Breckenridge MB, Rhoads GG. Maternal asthma and transient tachypnea of the newborn. Pediatrics. 1998;102: 84–90. [DOI] [PubMed] [Google Scholar]
- 19.Stenius-Aarniala B, Piirila P, Teramo K. Asthma and pregnancy: a prospective study of 198 pregnancies. Thorax. 1988;43: 12–18. doi: 10.1136/thx.43.1.12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Powell H, Murphy VE, Taylor DR, Hensley MJ, McCaffery K, Giles W, et al. Management of asthma in pregnancy guided by measurement of fraction of exhaled nitric oxide: a double-blind, randomised controlled trial. Lancet. 2011;378: 983–990. doi: 10.1016/S0140-6736(11)60971-9 [DOI] [PubMed] [Google Scholar]
- 21.Tamási L, Bohács A, Horváth I, Losonczy G. Asthma in pregnancy—from immunology to clinical management. Multidiscip Respir Med. BioMed Central Ltd; 2010;5(4):259–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gissler M, Teperi J, Hemminki E, Meriläinen J. Data quality after restructuring a national medical registry. Scand J Soc Med. 1995;23: 75–80. [DOI] [PubMed] [Google Scholar]
- 23.Artama M, Gissler M, Malm H, Ritvanen A, Drugs and Pregnancy Study Group. Nationwide register-based surveillance system on drugs and pregnancy in Finland 1996–2006. Pharmacoepidemiol Drug Saf. 2011;20: 729–738. doi: 10.1002/pds.2159 [DOI] [PubMed] [Google Scholar]
- 24.Sankilampi U, Hannila M-L, Saari A, Gissler M, Dunkel L. New population-based references for birth weight, length, and head circumference in singletons and twins from 23 to 43 gestation weeks. Ann Med. 2013;45: 446–454. doi: 10.3109/07853890.2013.803739 [DOI] [PubMed] [Google Scholar]
- 25.Meher S, Alfirevic Z. Choice of primary outcomes in randomised trials and systematic reviews evaluating interventions for preterm birth prevention: a systematic review. BJOG. 2014;121: 1188–94– discussion 1195–6. doi: 10.1111/1471-0528.12593 [DOI] [PubMed] [Google Scholar]
- 26.Källén B, Otterblad Olausson P. Use of anti-asthmatic drugs during pregnancy. 3. Congenital malformations in the infants. Eur J Clin Pharmacol. 2007;63: 383–388. doi: 10.1007/s00228-006-0259-z [DOI] [PubMed] [Google Scholar]
- 27.Zhang J, Troendle J, Reddy UM, Laughon SK, Branch DW, Burkman R, et al. Contemporary cesarean delivery practice in the United States. Am J Obstet Gynecol. 2010;203: 326.e1–326.e10. doi: 10.1016/j.ajog.2010.06.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Palmsten K, Schatz M, Chan PH, Johnson DL, Chambers CD. Validation of the Pregnancy Asthma Control Test. J Allergy Clin Immunol Pract. 2016;4: 310–5.e1. doi: 10.1016/j.jaip.2015.11.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Society BT. British guideline on the management of asthma https://www.brit-thoracic.org.uk/standards-of-care/guidelines/btssign-british-guideline-on-the-management-of-asthma/ Visitide at March 2018. In: https://www.brit-thoracic.org.uk/standards-of-care/guidelines/btssign-british-guideline-on-the-management-of-asthma/. Sep 2016 p. 214.
- 30.Chung KF, Wenzel SE, Brozek JL, Bush A, Castro M, Sterk PJ, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014;43: 343–373. doi: 10.1183/09031936.00202013 [DOI] [PubMed] [Google Scholar]
- 31.Asthma GIF. Global strategy for asthma management and prevention, update 2017 http://ginasthma.org/2017-gina-report-global-strategy-for-asthma-management-and-prevention/. In: http://ginasthma.org/2017-gina-report-global-strategy-for-asthma-management-and-prevention/.
- 32.Schatz M, Dombrowski MP, Wise R, Momirova V, Landon M, Mabie W, et al. Spirometry is related to perinatal outcomes in pregnant women with asthma. Am J Obstet Gynecol. 2006;194: 120–126. doi: 10.1016/j.ajog.2005.06.028 [DOI] [PubMed] [Google Scholar]
- 33.Cossette B, Forget A, Beauchesne M-F, Rey É, Lemière C, Larivée P, et al. Impact of maternal use of asthma-controller therapy on perinatal outcomes. Thorax. 2013;68: thoraxjnl–2012–203122–730. doi: 10.1136/thoraxjnl-2012-203122 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The study consisted of 962 405 singleton live and stillbirths in Finland between the years 1996 and 2012.
(DOC)
The study consisted of 962 405 live- and stillborn singletons in Finland between the years 1996 and 2012, Chi-square test between groups.
(DOC)
Unadjusted and adjusted odds ratios.
(DOC)
(DOC)
Data Availability Statement
Because of the national legal restrictions, authors cannot make de-identified dataset available. Data contains confidential patient information for which access is strongly prohibited. An authorisation from the National Institute of Health and Welfare (THL) and Social Insurance Institution (KELA) is necessary for any use of similar confidential data for research purposes. The contact information for a data access committee in THL is kirjaamo@thl.fi and in KELA tietopyynnot@kela.fi.