Abstract
Background
Vascular stiffness is associated with aging and cognitive impairment in older populations without HIV. HIV has been linked to increased vascular stiffness. We examined whether vascular stiffness relates to cognitive decline at younger ages in women with or at risk for HIV.
Methods
We evaluated the association of carotid artery stiffness with decline in neuropsychological test performance among participants in the Women’s Interagency HIV Study, and assessed whether HIV modified the association. Baseline carotid stiffness, defined by the distensibility index, was determined at a single visit using carotid artery ultrasound. Longitudinal neuropsychological testing from 2004–2016 included Trail Making Tests A and B (TRLA, TRLB) and the Symbol Digit Modalities Test (SDMT). Relationships were assessed with linear mixed-effects models adjusted for demographic, behavioral, cardiometabolic, and neuropsychological factors.
Results
Among 1662 women (1192 [72%] HIV+), median baseline age was 41 years (IQR 34–47), with 60% non-Hispanic black and 28% Hispanic. Lower baseline distensibility (greater carotid stiffness) was associated with greater decline in neuropsychological test scores over 10-year follow-up as measured by SDMT (adjusted β=−0.06 per SD, p<0.001), TRLA (β=−0.08 per SD; p<0.001), and TRLB (β=−0.08 per SD; p<0.001). Changes in cognitive function did not differ by HIV serostatus, or HIV-related factors.
Conclusions
Higher carotid stiffness was independently associated with faster decline in executive functioning, information processing and psychomotor speed even in mostly middle-aged minority women and regardless of HIV serostatus. Our study highlights the need for cardiovascular risk factor modification to prevent premature cognitive deterioration in this at-risk population.
Keywords: HIV, Vascular Stiffness, Carotid Artery, Cognitive Aging, Women’s Health
Introduction
Arterial stiffness increases with advancing age, and is closely linked to hypertension, diabetes, and other cardiovascular risk factors.1–4 Increased stiffness of the central arteries leads to higher pulse pressure, exposing the brain, heart, and kidneys to damage from amplified blood pressure oscillation.5–9 Consistent with such end-organ damage, higher arterial stiffness assessed by aortic pulse wave velocity or carotid ultrasound has been associated with increased cardiovascular disease morbidity and mortality in general population samples of primarily older adults.10–15 In such cohorts, increased arterial stiffness has also been linked to cognitive decline, supporting the notion that increased pulsatile load is deleterious for cerebral vasculature.7–8
Human immunodeficiency virus (HIV) infection has a well-documented link to cardiovascular disease,16–17 with evidence not only of greater atherosclerosis,18–19 but also of increased vascular stiffness, reflecting arteriosclerosis.20 In particular, increased T-cell activation in a female cohort and low nadir CD4 T-cell count in a predominantly male cohort have been linked to vascular stiffness.21–22 HIV is also associated with cognitive impairment in both men and women, with deficits highlighted in various cognitive domains.23–24 Such deficits have been linked in cross-sectional studies to cardiovascular risk factors and measures of subclinical atherosclerosis in HIV-infected individuals of both sexes, underscoring a putative vascular basis.25–26 Although vascular stiffness offers a more direct link to cerebral vascular damage than clinical risk factors5–9, its relationship with cognitive function has scarcely been studied in people living with HIV, especially women.27 Nor has the prospective association between measures of vascular stiffness and cognitive decline been examined in such at-risk individuals. We leveraged availability of carotid stiffness measures and serial assessments of cognitive function in a longitudinal study of women with or at risk for HIV infection to study this relationship in a high-risk population at mid-life. We hypothesized that higher carotid stiffness at baseline relates to greater decline in executive functioning, information processing and psychomotor speed during long-term follow-up, and that HIV infection strengthens this association.
Methods
Study Population
The Women’s Interagency HIV Study (WIHS) is a prospective cohort study of women with or at risk for HIV infection.28–29 Participants were initially enrolled in the WIHS at six US study sites between 1994 and 2002. Women were recruited by referral from physician offices, HIV community and social service organizations, churches, and fellow participants. Women in the WIHS without HIV were selected only if they engaged in ≥1 high-risk behaviors, and are demographically similar to women with HIV. Participants are seen at semi-annual visits for detailed examinations, structured interviews, and laboratory testing.28–29 We used available baseline carotid stiffness measures and serial cognitive assessments obtained between 2004 and 2016. Inclusion criteria for the current analysis were completion of a carotid artery ultrasound examination at baseline (2004–2006), at least one assessment of cognitive function within one year of the baseline ultrasound visit, and the ability to complete neuropsychological testing in English. Exclusion criteria included self-reported history of stroke, transient ischemic attack, heart failure, or other heart disease. Given the high prevalence of mental health disorders in the population under study, we did not exclude women with such disorders in order to improve generalizability, instead adjusting for depressive symptoms. The study was approved by the Institutional Review Board at each study site.
Cognitive Outcome Measures
From 2004 through 2006, select neuropsychological assessments of executive function, information processing and psychomotor speed were performed yearly in the WIHS. Beginning in 2009, a more comprehensive neuropsychological assessment was added, with the participants completing testing every two years. The primary outcomes of our study were changes in score over time for the 3 neuropsychological tests available during the study period of 2004 to 2016 including the Trail Making Test Parts A and B (TRLA and TRLB)30 and the Symbol Digit Modalities Test (SDMT).31 These tests assess executive function (TRLB) and psychomotor and information processing speed (TRLA and SDMT) and are scored based on time to completion (Range: 0 to 5 minutes) for TRLA and TRLB and total correct (Range: 0 to 110) for SDMT. Testing was performed by trained psychometrists using standardized protocols.32 Standardized unadjusted Z-scores were derived for each outcome measure using the 470 HIV-uninfected women enrolled in the study as the normative sample. A Z-score was calculated as the difference between the individual neuropsychological test score and the normative sample mean neuropsychological test score at baseline divided by the normative sample standard deviation at baseline. The absolute value was taken so that positive Z-score indicates improved performance.
Carotid Artery Stiffness
The primary exposure was a one-time measure of carotid artery stiffness assessed beginning in 2004 using high-resolution B-mode ultrasound of the right common carotid artery, and interpreted at a central location.20 The baseline carotid stiffness visit was defined to be the closest visit within one year of the baseline neuropsychological testing visit.
The primary measure of carotid stiffness, a distensibility index,33 was calculated from ultrasound and contemporary seated brachial blood pressure measurements as:
Right common carotid artery diameter was assessed at maximum diameter at systole (RCCAmax) and minimum diameter at diastole (RCCAmin). Pulse pressure was calculated from the mean of five brachial artery measurements.
An alternative measure of carotid stiffness, Young’s elastic modulus,33 was used in secondary analyses instead of distensibility. Young’s elastic modulus also accounts for carotid wall thickness, and was calculated as:
Carotid artery intima-media thickness (CIMT) was measured at the right distal common carotid artery18.
Baseline Covariates
All covariates were assessed at baseline. HIV serostatus was determined using ELISA with confirmation by Western blot. Educational achievement was determined by the Wide Range Achievement Test-3 (WRAT-3)34 and depressive symptoms by the Center for Epidemiological Studies Depression Scale (CES-D).35 Psychoactive medication use was defined as use of one or more antidepressant, antipsychotic, sedative or dementia medication. Diabetes was defined as fasting blood glucose ≥126 mg/dl, hemoglobin A1c ≥6.5%, or use of anti-hyperglycemic medication.36 Body mass index (BMI) was defined as the ratio of body weight in kilograms to height in meters squared. Hypertension was defined by systolic blood pressure ≥140 mm Hg, diastolic blood pressure ≥90 mm Hg or self-reported use of anti-hypertensive medication. History of coronary heart disease (CHD), stroke, heart failure, or other heart disease were assessed by self-report. History of hepatitis C virus (HCV) infection was determined by antibody status and RNA level. Estimated glomerular filtration rate (eGFR) was determined using the CKD-EPI equation.37 Framingham 10-year and the Data-collection on Adverse Effects of Anti-HIV Drugs (D:A:D) reduced 5-year risk scores for all cardiovascular disease were calculated using previously published regression equations.38–39
Statistical Analysis
Linear mixed-effects models were used to examine the association of baseline measures of carotid artery stiffness with change in neuropsychological testing performance over time, accounting for HIV status, with time operationalized as participant age. Z-scores for neuropsychological tests were analyzed as outcomes in separate regression models. Adjustments were made for baseline demographic, behavioral, cardiometabolic and neuropsychological covariates. Age was included as baseline age (time-constant) as well as current age (time-dependent) to capture its cross-sectional and longitudinal association with cognitive performance, respectively. This model is equivalent to widely used approaches for the analysis of longitudinal data that include baseline age and time since enrollment. Here we use current age instead of time since enrollment for ease of interpretation. Current age (for simplicity referred to as age hereafter) was included as a random effect in the mixed effect models (i.e. random slope) to capture heterogeneity in patterns of cognitive performance over time observed among participants. Further, cross-product terms of exposure measures with participant age were included to examine the relationships of these predictors with change in the slope of neuropsychological performance over time. For example, an interaction between distensibility and age represents how much the change in neuropsychologic performance over time is modified by distensibility. The statistical approach and assumption of linearity was modeled on previous longitudinal studies of cognitive function in general population cohorts without HIV.7–8
HIV-related covariates were assessed in separate linear mixed-effect models including baseline CD4 count, nadir CD4 count prior to antiretroviral therapy (ART) initiation, baseline plasma HIV RNA level, history and duration of ART, history of clinical AIDS, and persistent viral suppression, defined as both suppressed HIV RNA (<80 copies per ml) and consistent use of potent ART at every visit during the study period.
Potential effect modification by HIV status or by HIV-related factors of the relationship between baseline carotid artery stiffness and change in neuropsychological performance was assessed through the three-way interaction between HIV status or HIV-related factor, age, and each stiffness measure. Linear mixed effects models without longitudinal terms were used to assess cross-sectional associations between baseline covariates and neuropsychological performance at baseline.
Multiple imputation was performed for 146 (9%) women who had missing data for one or more adjustment variables (89% were HIV infected).40 In sensitivity analysis, excluding women with missing adjustment variables did not alter patterns of association. A two-sided p<0.05 was considered statistically significant. SAS 9.4 (SAS Institute, Cary, NC) was used for all analyses.
Results
Characteristics of Study Population
Of the 2301 WIHS eligible participants attending at least one visit during the study period, 1735 underwent carotid ultrasound, and 1723 of these had at least one neuropsychological assessment within 1 year of their single (baseline) carotid examination. Of these women, 85 had only one neuropsychological assessment; however excluding these women did not change longitudinal patterns of association. Another 61 women were excluded because of self-reported history of stroke, transient ischemic attack, heart failure, or other heart disease resulting in 1662 women (1192 HIV-infected, 72%) in the study sample (Figure 1). Compared with those excluded for not undergoing carotid ultrasound, included participants were younger (median 2 years less), and less likely to be post-menopausal, have hypertension or CHD, have AIDS history, or use psychoactive medications. Included participants were more likely to be non-Hispanic black or Hispanic and of low socioeconomic status and had marginally lower performance on some neuropsychological tests. Included and excluded women were otherwise similar including baseline carotid stiffness and the proportion HIV-infected (Supplementary Table 1). Women in the study sample had a median baseline age of 41 (IQR 34–47) years (Table 1). Most were non-Hispanic black (60%) or Hispanic (28%). Compared to HIV-uninfected women, HIV-infected women were older and more commonly post-menopausal, with higher prevalence of HCV infection and psychoactive medication use, and lower prevalence of smoking. Across lower carotid distensibility tertiles (higher arterial stiffness), women were older, more often non-Hispanic black, and exhibited higher BMI. They had a higher frequency of hypertension, diabetes, lipid-lowering or psychoactive medication use, HCV infection, and post-menopausal status, and lower eGFR. Correspondingly, women in lower carotid distensibility tertiles had higher Framingham and D:A:D cardiovascular risk scores. In addition, women in the lower and middle distensibility tertiles more often were HIV-infected and used illicit drugs. Performance on all three neuropsychological tests tended to be worse in the lower carotid distensibility tertiles. Among the HIV-infected group, women with lower carotid distensibility exhibited greater cumulative exposure to protease inhibitors and nucleoside reverse transcriptase inhibitors.
FIGURE 1. Study inclusion and exclusion flow diagram.
Table 1.
| a: Characteristics* of WIHS participants by tertiles of distensibility index
| ||||
|---|---|---|---|---|
| Distensibility index tertile | ||||
| Characteristic | Low Distensibility (N=541) |
Medium Distensibility (N=559) |
High Distensibility (N=562) |
P value |
| Distensibility index, 10−6 × Newtons−1 × meters2 | 10.1 (7.8–12.1) | 17.7 (16.0–19.8) | 28.3 (24.9–33.0) | N/A |
| HIV positive, n (%) | 406 (75) | 419 (75) | 367 (66) | 0.0002 |
| Demographics | ||||
| Age, years | 45 (39–51) | 42 (36–47) | 36 (30–42) | <0.0001 |
| Race/ethnicity, n (%) | ||||
| Non-Hispanic black | 380 (70) | 330 (59) | 287 (51) | <0.0001 |
| Hispanic | 111 (20) | 165 (29) | 194 (35) | <0.0001 |
| Non-Hispanic white | 41 (8) | 53 (10) | 65 (12) | 0.08 |
| WRAT-3 Reading Score | 93 (77–106) | 91 (77–103) | 93 (79–107) | 0.06 |
| Income <$30,000 per year, n (%) | 444 (82) | 461 (82) | 443 (79) | 0.23 |
| Behavior-related | ||||
| Current smoker, n (%) | 234 (43) | 255 (46) | 241 (43) | 0.61 |
| History of injection drug use, n (%) | 145 (27) | 155 (28) | 91 (16) | <0.0001 |
| Current crack/cocaine, n (%) | 45 (8) | 52 (9) | 42 (8) | 0.54 |
| Current alcohol use >7 drinks/week, n (%) | 34 (6) | 50 (9) | 42 (8) | 0.25 |
| History of hepatitis C virus infection, n (%) | 167 (31) | 167 (30) | 102 (18) | <0.0001 |
| Metabolic risk factors | ||||
| Body mass index, kg/m2 | 30 (25–34) | 28 (24–34) | 26 (23–31) | <0.0001 |
| Systolic blood pressure, mm Hg | 123 (113–138) | 117 (108–130) | 111 (104–118) | <0.0001 |
| Mean arterial pressure, mm Hg | 91 (83–101) | 87 (79–96) | 82 (76–88) | <0.0001 |
| Heart rate, beats per minute | 74 (68–83) | 71 (64–78) | 67 (61–73) | <0.0001 |
| Anti-hypertensive medication, n (%) | 179 (33) | 94 (17) | 32 (6) | <0.0001 |
| History of hypertension, n (%) | 243 (45) | 137 (25) | 65 (12) | <0.0001 |
| History of diabetes, n (%) | 170 (31) | 117 (21) | 64 (11) | <0.0001 |
| History of coronary heart disease, n (%) | 16 (3) | 29 (5) | 9 (2) | 0.003 |
| Total cholesterol, mg/dL | 175 (148–202) | 173 (149–201) | 166 (143–194) | 0.003 |
| HDL cholesterol, mg/dL | 49 (38–58) | 49 (40–60) | 49 (39–61) | 0.08 |
| Lipid-lowering medication, n (%) | 46 (8) | 41 (7) | 10 (2) | <0.0001 |
| eGFR, ml/min/1.73 m2 | 97 (80–114) | 100 (85–112) | 109 (93–120) | <0.0001 |
| Post-menopausal (self-report), n (%) | 171 (32) | 124 (22) | 54 (10) | <0.0001 |
| Framingham 10-year all-CVD risk % | 4.9 (2.5–9.9) | 3.3 (1.8–5.7) | 1.9 (1.2–3.2) | <0.0001 |
| D:A:D 5-year all-CVD risk % | 1.6 (0.0–3.1) | 1.1 (0.0–2.0) | 0.6 (0.0–1.1) | <0.0001 |
| Neuropsychological | ||||
| Trailmaking Test A Raw Score, seconds | 39 (30–49) | 36 (29–47) | 35 (27–45) | <0.0001 |
| Trailmaking Test B Raw Score, seconds | 87 (65–127) | 85 (63–114) | 77 (60–106) | <0.0001 |
| Symbol Digit Modalities Test Raw Score, total correct | 41 (32–49) | 42 (35–50) | 45 (36–52) | <0.0001 |
| Antipsychotic, antidepressant, sedative or Alzheimer dementia medication, n (%) | 140 (26) | 127 (23) | 93 (17) | 0.0006 |
| CES-D score | 11 (4–22) | 11 (4–19) | 10 (4–21) | 0.95 |
| HIV-specific (among HIV-infected subset) | ||||
| Baseline CD4+ T cell count, cells/mm3 | 453 (276–696) | 485 (308–696) | 456 (305–682) | 0.38 |
| Nadir CD4+ T cell count, cells/mm3 | 241 (136–379) | 283 (174–408) | 298 (191–415) | 0.0007 |
| Undetectable baseline viral load, n (%) | 186 (46) | 219 (54) | 167 (46) | 0.004 |
| History of AIDS, n (%) | 154 (38) | 162 (39) | 126 (34) | 0.02 |
| Potent ART use in past 6 months, n (%) | 261 (64) | 274 (66) | 225 (61) | 0.004 |
| Cumulative exposure of potent ART, years | 4.0 (1.5–6.5) | 4.0 (1.0–6.5) | 3.0 (1.0–6.0) | 0.07 |
| of PIs, years | 2.0 (0.0–5.0) | 1.5 (0.0–6.0) | 1.0 (0.0–4.0) | 0.01 |
| of NNRTIs, years | 0.5 (0.0–3.0) | 0.5 (0.0–3.0) | 0.5 (0.0–3.0) | 0.98 |
| of NRTIs, years | 6.0 (2.5–8.0) | 5.0 (1.5–8.0) | 3.5 (1.5–7.0) | 0.0001 |
| b: Characteristics* of WIHS participants by HIV serostatus
| |||
|---|---|---|---|
| HIV Serostatus | |||
| Characteristic | Positive (N=1192) |
Negative (N=470) |
P value |
| Distensibility index, 10−6 × Newtons−1 × meters2 | 17.6 (12.0–24.0) | 18.9 (12.4–27.2) | 0.006 |
| Demographics | |||
| Age, years | 42 (36–48) | 38 (31–46) | <0.0001 |
| Race/ethnicity, n (%) | |||
| Non-Hispanic black | 697 (59) | 300 (64) | 0.05 |
| Hispanic | 345 (29) | 125 (27) | 0.34 |
| Non-Hispanic white | 124 (10) | 35 (7) | 0.08 |
| WRAT-3 Reading Score | 93 (77–105) | 93 (79–107) | 0.04 |
| Income <$30,000 per year, n (%) | 984 (83) | 364 (77) | 0.02 |
| Behavior-related | |||
| Current smoker, n (%) | 493 (42) | 237 (51) | 0.001 |
| History of injection drug use, n (%) | 307 (26) | 84 (18) | 0.0007 |
| Current crack/cocaine, n (%) | 85 (7) | 54 (11) | 0.006 |
| Current alcohol use >7 drinks/week, n (%) | 75 (6) | 51 (11) | 0.003 |
| History of hepatitis C virus infection, n (%) | 351 (29) | 85 (18) | <0.0001 |
| Metabolic risk factors | |||
| Body mass index, kg/m2 | 27 (24–32) | 30 (25–35) | <0.0001 |
| Systolic blood pressure, mm Hg | 116 (107–128) | 117 (109–129) | 0.05 |
| Mean arterial pressure, mm Hg | 86 (79–95) | 86 (78–96) | 0.99 |
| Heart rate, beats per minute | 71 (64–78) | 69 (63–76) | 0.003 |
| Anti-hypertensive medication, n (%) | 228 (19) | 77 (16) | 0.22 |
| History of hypertension, n (%) | 316 (27) | 129 (27) | 0.70 |
| History of diabetes, n (%) | 253 (21) | 98 (21) | 0.92 |
| History of coronary heart disease, n (%) | 38 (3) | 16 (3) | 0.93 |
| Total cholesterol, mg/dL | 170 (145–200) | 171 (149–198) | 0.31 |
| HDL cholesterol, mg/dL | 47 (37–58) | 54 (44–64) | <0.0001 |
| Lipid-lowering medication, n (%) | 88 (7) | 9 (2) | <0.0001 |
| eGFR, ml/min/1.73 m2 | 101 (85–117) | 102 (89–117) | 0.20 |
| Post-menopausal (self-report), n (%) | 282 (24) | 67 (14) | <0.0001 |
| Framingham 10-year all-CVD risk % | 3.2 (1.7–6.2) | 3.1 (1.6–6.4) | 0.99 |
| D:A:D 5-year all-CVD risk % | 1.0 (0.5–2.1) | 0.9 (0.4–17) | 0.07 |
| Neuropsychological | |||
| Trailmaking Test A Raw Score, seconds | 37 (30–48) | 34 (27–45) | <0.0001 |
| Trailmaking Test B Raw Score, seconds | 85 (63–119) | 78 (60–103) | 0.0002 |
| Symbol Digit Modalities Test Raw Score, total correct | 42 (33–50) | 45 (37–52) | <0.0001 |
| Antipsychotic, antidepressant, sedative or Alzheimer dementia medication, n (%) | 294 (25) | 66 (14) | <0.0001 |
| CES-D score | 11 (4–22) | 9 (4–18) | 0.12 |
| HIV-specific (among HIV-infected subset) | |||
| Baseline CD4+ T cell count, cells/mm3 | 433 (277- 630) | N/A | |
| Nadir CD4+ T cell count, cells/mm3 | 275 (164–400) | N/A | |
| Undetectable baseline viral load, n (%) | 605 (51) | N/A | |
| History of AIDS, n (%) | 442 (37) | N/A | |
| Potent ART use in past 6 months, n (%) | 760 (64) | N/A | |
| Cumulative exposure of potent ART, years | 3.5 (1.0–6.5) | N/A | |
| of PIs, years | 1.5 (0.0–5.0) | N/A | |
| of NNRTIs, years | 0.5 (0.0–3.0) | N/A | |
| of NRTIs, years | 5.0 (2.0–8.0) | N/A | |
Continuous variables are presented as medians (interquartile ranges), comparisons by the Wilcoxon rank-sum test for medians or Fisher’s exact for counts
AIDS: Acquired immunodeficiency syndrome; ART: Anti-retroviral therapy; CES-D: Center for Epidemiological Studies Depression Scale (range 0–60); WRAT-3 : Wide Range Achievement Test 3 (standard average 90–109); D:A:D: Data-collection on Adverse Effects of Anti-HIV Drugs; eGFR: Estimated glomerular filtration rate; NNRTI: Non-nucleoside reverse transcriptase inhibitor; NRTI: Nucleoside reverse transcriptase inhibitor; PI: Protease inhibitor
Cross-sectional Associations with Cognitive Function at Baseline
Some factors in adjusted models were associated with cross-sectional differences in Z-scores at the baseline visit across the three neuropsychological tests (Supplementary Table 2). Higher baseline age, HIV infection, Hispanic ethnicity, Black race, higher CES-D score, hypertension and post-menopausal status were associated with lower Z-scores indicating worse neuropsychological test performance. Greater distensibility at baseline, income of >$30,000 per year, higher WRAT-3 score, and higher BMI were associated with higher Z-scores indicating better neuropsychological test performance.
Longitudinal Association of Carotid Distensibility with Cognitive Decline
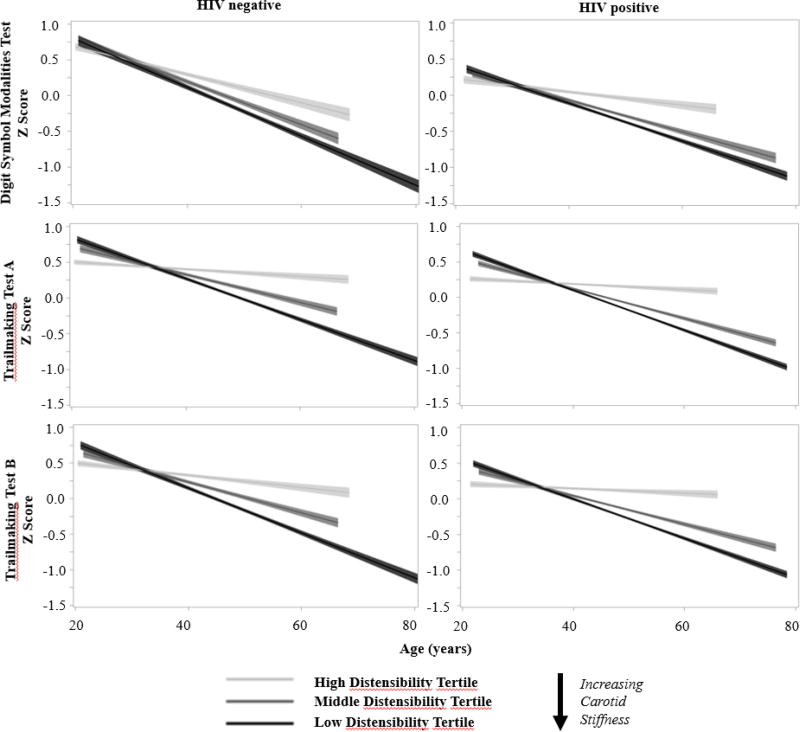
Serial cognitive assessments were performed at a median (range) of 6 (1–7) visits over 8.5 (0–12) years. Participants experienced an unadjusted rate of decline in Z-score over ten years of age in SDMT, TRLA, and TRLB of −0.10 (95% CI −0.13 to −0.07), −0.12 (−0.16 to −0.09), and −0.18 (−0.21 to −0.14). In fully adjusted linear mixed-effects models, lower baseline carotid distensibility (greater arterial stiffness) was associated with greater cognitive decline with increasing age (Table 2 and Figure 2). Interactions of distensibility with age were significant for all three neuropsychological outcome measures, indicating a significant corresponding decline in executive function, information processing and psychomotor speed over time. The β coefficients in Table 2 represent the increase in rate of cognitive decline, measured as the difference in Z-score per 10-year increase in age, for one standard deviation (SD) decrement in distensibility. Additional adjustment for mean arterial pressure and heart rate did not alter these longitudinal relationships. Associations were similar when examining elastic modulus (Supplementary Table 3 and Supplementary Figure 1). Examination of spaghetti plots showed that no one single pattern could characterize the trend in all individuals, supporting linearity that incorporates the random slope as a reasonable approximation.
Table 2.
Association of baseline carotid distensibility index or HIV serostatus with longitudinal change in neuropsychological testing score (SD/per 10-year increase)
| Increase in rate of decline per SD decrement in distensibility |
Increase in rate of decline for HIV positive women (versus HIV negative women) |
|||
|---|---|---|---|---|
| Cognitive Test | β* (95% CI) | P | β† (95% CI) | P |
| Symbol Digit Modalities Test, Z Score | −0.06 (−0.03 to −0.10) | 0.0002 | −0.03 (−0.10 to 0.03) | 0.33 |
| Trail Making Test A, Z Score | −0.08 (−0.04 to −0.09) | <0.0001 | −0.07 (−0.10 to 0.02) | 0.11 |
| Trail Making Test B, Z Score | −0.08 (−0.06 to −0.09) | <0.0001 | −0.05 (−0.10 to 0.02) | 0.18 |
Models adjusted for baseline age, HIV serostatus, carotid distensibility index, race-ethnicity, income, Wide Range Achievement Test (WRAT)-3 reading score, Center for Epidemiologic Studies Depression Scale (CES-D), current smoking, current alcohol use, history of intravenous drug use, current crack/cocaine, history hepatitis C virus, body mass index, hypertension, diabetes, menopausal status, total cholesterol, HDL cholesterol, use of lipid-lowering medications, psychoactive medication use, history of coronary heart disease, and eGFR.
β is the distensibility × age interaction term, and represents the increase in rate of cognitive decline (measured as difference in Z-score per 10-year increase in age) for one standard deviation (9.4 × 10−6 × Newtons−1 × meters2) decrease in distensibility
β is the HIV serostatus × age interaction term, and represents the increase in rate of cognitive decline (measured as difference in Z-score per 10-year increase in age) for an HIV-positive woman versus an HIV-negative woman
FIGURE 2. Multivariable adjusted longitudinal change in neuropsychological performance as a function of distensibility index (tertiles).
Models adjusted for baseline age, HIV serostatus, carotid distensibility index, race-ethnicity, income, Wide Range Achievement Test (WRAT)-3 reading score, Center for Epidemiologic Studies Depression Scale (CES-D), current smoking, current alcohol use, history of intravenous drug use, current crack/cocaine, history hepatitis C virus, body mass index, hypertension, diabetes, menopausal status, total cholesterol, HDL cholesterol, use of lipid-lowering medications, psychoactive medication use, history of coronary heart disease, and eGFR
Longitudinal Associations of HIV infection and HIV-related Factors with Cognitive Decline
Participants with HIV had fewer serial cognitive assessments than those without HIV, with medians (ranges) of 5 (1–7) and 6 (1–7) assessments respectively (p<0.001). As calculated from the beta coefficients for the time-varying age terms, the unadjusted rates of neuropsychological testing score decline over 10 years of age in HIV-infected participants were similar to those of uninfected participants (SDMT: −0.09 [95% CI −0.13 to −0.05] vs −0.10 [-0.15 to −0.05], p=0.95; TRLA: −0.12 [-0.17 to −0.07] vs −0.11 [-0.17 to −0.05], p=0.35; TRLB: −0.18 [-0.22 to −0.14] vs −0.16 [-0.21 to −0.11], p=0.47). In a fully adjusted model controlling for carotid stiffness, the rate of decline in neuropsychological testing scores was not different in HIV-infected compared to HIV-uninfected women (see Table 2 and Supplementary Table 3). Associations of a variety of HIV-related factors with longitudinal decline in neuropsychological testing performance were also analyzed in separate fully adjusted linear mixed-effects models (Supplementary Table 4). The following covariates were significantly associated with the longitudinal slope in neuropsychological test scores: history of AIDS (versus AIDS-free, SDMT: β=−0.09 SD units over 10 years increasing age, p=0.03 and TRLA: β=−0.20, p=0.002), persistent viral suppression (versus participants with 1+ viremic or untreated visit during follow-up, TRLA: β=−0.20, p=0.03), and duration of protease inhibitor use at baseline (per year, TRLA: β=−0.02, p=0.009 and TRLB: β =−0.01; p=0.03).
The longitudinal association of carotid stiffness measures with cognitive performance did not differ by HIV status (all p interaction values nonsignificant), as assessed by the three-way interaction between HIV status, age, and carotid stiffness measures. Additionally all three-way interactions between HIV-related factors, age and carotid stiffness measures were not statistically significant.
Discussion
Among middle-aged women with or at risk for HIV infection, we found that baseline carotid stiffness was associated with substantial decline in executive function, information processing and psychomotor speed over time, independent of potential confounders. In adjusted models, HIV serostatus was not associated with decline in neuropsychological test performance over time, nor did it modify the association of carotid stiffness with longitudinal decline in neuropsychological performance. By contrast, certain HIV-related factors were associated with significantly steeper rates of decline in neuropsychological performance, including a history of AIDS and duration of protease inhibitor use, although these were likewise not found to modify the relationship between carotid stiffness and longitudinal decline in neuropsychological performance.
The women under study were predominantly a low-income, minority, urban cohort with high prevalence of smoking, substance use, diabetes, hypertension, and HCV infection. This placed these women at greater cardiovascular risk41 than reflected by their age or captured by their Framingham or D:A:D risk scores. Indeed, these women with or at risk for HIV were younger than participants in previous longitudinal studies of aging and cognition in the general population. Despite their relative youth, we identified an association between vascular stiffness and cognitive decline. As these women age, cognitive performance for those with greater vascular stiffness is expected to continue to decline at a faster rate, increasing risk for cognitive impairment and dementia.
A strength of our study is its longitudinal nature, mitigating the risk of bias inherent in the comparison to external norms by comparing individuals to their own performance over time. An additional strength is the use of HIV-uninfected women who are demographically and behaviorally similar to their HIV-infected counterparts. Both HIV-infected and HIV-uninfected women in this study are cognitively impaired compared to community-based general population samples, with mean TRLA, TRLB, and SDMT scores below the general population mean scores at each age group.42–43 The relationship of carotid artery stiffness with cognitive function in this high-risk cohort of women cannot be directly compared to different measures of vascular stiffness and neuropsychological assessments in the general population. Yet, the magnitude of associations proved substantial, with each SD decrement in carotid artery distensibility showing 10-year declines in neuropsychological outcome measures that were a substantial proportion of the average 10-year rate of decline in the entire sample. Such associations highlight the potential importance of intervening more aggressively in these high-risk individuals to modify risk factors influencing vascular and cognitive health.
HIV-associated Neurocognitive Disorders (HAND) are common, but their severity is waning in the era of potent ART. Age, cardiovascular risk factors, use of illicit drugs, and psychiatric disorders have all been identified as determinants.44 In previous cross-sectional analyses in the present cohort, HIV has been associated with lower performance in multiple cognitive domains including executive function, complex attention, learning, and memory.24, 32 Previous findings that HIV infection, and in particular increased CD4+ T cell activation, is associated with increased carotid stiffness21–22 suggest that one contribution to poor performance in HIV-infected women could be increased vascular stiffness. However, neither the current study nor a previous longitudinal study in the same cohort that included a more comprehensive battery of neuropsychological assessments45 found a longitudinal association of HIV infection with greater decline in performance in most cognitive domains. The current study did find that HIV-infected women with high-risk features including AIDS history and longer protease inhibitor use had faster decline in neuropsychological performance compared with HIV-infected women without these features. These findings are consistent with prior data linking protease inhibitors with atherosclerosis and increased vascular stiffness.46 A link between persistent viral suppression and greater slope of decline in neuropsychological test performance runs counter to expectation, but could be explained by deleterious effects of long-term potent ART use on central and cerebral vasculature. This finding was observed in only one neuropsychological performance measure however. The premise of HIV-related vascular stiffness being associated with deficits in cognition will require further investigation in larger samples.
The impact of vascular stiffness on cognition is thought to be mediated by a downstream influence on small vessels, including endothelial dysfunction and exposure to high pulsatility.5–6 White matter lesions can be an imaging sign of such vascular damage occurring in younger individuals47 and have been correlated with deficits in cognition.48 Neuroimaging would further elucidate the pathophysiology behind vascular stiffness-associated cognitive decline and how this process might differ in people living with HIV.
Several limitations of the present study bear mention. The neuropsychological tests utilized primarily assess executive function and psychomotor speed. Deficits in learning, memory, and attention have also been identified in women with HIV, and there may be a differential effect of HIV on the longitudinal association of vascular stiffness found with a more comprehensive cognitive battery. Additionally, carotid stiffness measured by ultrasound is a less validated measure for vascular stiffness than aortic pulse wave velocity. We suspect that using carotid stiffness underestimated the association of vascular stiffness with cognitive function based on previous studies finding that the aorta stiffens more than the carotid artery with age and increasing cardiovascular risk factors.49 Moreover, pulse pressure was measured peripherally, and not centrally, which may have underestimated true arterial stiffness and biased the results toward the null. Unexpectedly we did not find that HIV infection or HIV-related factors modified longitudinal associations of carotid stiffness with cognitive performance, possibly because of sample size limitations, limited follow-up period, or because almost two-thirds of women were taking potent ART at baseline attenuating the HIV effect. Women with HIV also had significantly fewer follow-up neuropsychological tests than those without HIV. Although this differential loss to follow-up was modest, it may have diminished the study’s ability to detect differences by HIV serostatus. Furthermore, we only included carotid stiffness measures at or within one year of the baseline neuropsychological assessment, so we cannot account for change in carotid stiffness over time. Whether HIV infection accentuates longitudinal changes in carotid stiffness differentially impacting cognition will require future study. We had a limited number of women (N=43) who were age 60 or older limiting our ability to extrapolate findings to older adults with HIV. Finally, the study only included women, most of whom were non-Hispanic black or Hispanic, so the findings are not necessarily generalized to men or non-Hispanic white women.
Conclusions
The findings of this study underscore the impact of adverse socioeconomic, behavioral and clinical factors on vascular health in women, manifesting even in early middle age, and the importance of loss of such vascular health to premature deterioration in cognitive function. Preventive interventions in this population are important to lessen not only cardiovascular risk, but also premature cognitive decline.
Supplementary Material
Acknowledgments
DMH designed the study and statistical analysis plan, performed the statistical analysis, and drafted and revised the paper. DBH designed the study and statistical analysis plan, and revised the draft paper. LHR designed the study and statistical analysis plan, and revised the draft paper. PM designed the study, and revised the draft paper. VV designed the study, and revised the draft paper. GS designed the study and statistical analysis plan, and revised the draft paper. XX designed the study and statistical analysis plan, and revised the draft paper. JL advised on the design, and revised the draft paper. HH advised on the design, and revised the draft paper. KA designed the study, and revised the draft paper. RCK designed the study and statistical analysis plan, and revised the draft paper. JRK designed the study and statistical analysis plan, and revised the draft paper.
DBH was supported by K01-HL-137557. RCK was supported by NIH grant R01HL126543, R01HL132794, R01HL095140, 1R01HL083760. JRK was supported by R01 HL132794 and K24 HL135413. VV was supported by K24MH098759. LHR was supported by NIMH K01MH098798.
Data in this manuscript were collected by the Women’s Interagency HIV Study (WIHS). The contents of this publication are solely the responsibility of the authors and do not represent the official views of the National Institutes of Health (NIH). WIHS (Principal Investigators): UAB-MS WIHS (Michael Saag, Mirjam-Colette Kempf, and Deborah Konkle-Parker), U01-AI-103401; Atlanta WIHS (Ighovwerha Ofotokun and Gina Wingood), U01-AI-103408; Bronx WIHS (Kathryn Anastos), U01-AI-035004; Brooklyn WIHS (Howard Minkoff and Deborah Gustafson), U01-AI-031834; Chicago WIHS (Mardge Cohen and Audrey French), U01-AI-034993; Metropolitan Washington WIHS (Seble Kassaye), U01-AI-034994; Miami WIHS (Margaret Fischl and Lisa Metsch), U01-AI-103397; UNC WIHS (Adaora Adimora), U01-AI-103390; Connie Wofsy Women’s HIV Study, Northern California (Ruth Greenblatt, Bradley Aouizerat, and Phyllis Tien), U01-AI-034989; WIHS Data Management and Analysis Center (Stephen Gange and Elizabeth Golub), U01-AI-042590; Southern California WIHS (Joel Milam), U01-HD-032632 (WIHS I – WIHS IV). The WIHS is funded primarily by the National Institute of Allergy and Infectious Diseases (NIAID), with additional co-funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), the National Cancer Institute (NCI), the National Institute on Drug Abuse (NIDA), and the National Institute on Mental Health (NIMH). Targeted supplemental funding for specific projects is also provided by the National Institute of Dental and Craniofacial Research (NIDCR), the National Institute on Alcohol Abuse and Alcoholism (NIAAA), the National Institute on Deafness and other Communication Disorders (NIDCD), and the NIH Office of Research on Women’s Health. WIHS data collection is also supported by UL1-TR000004 (UCSF CTSA) and UL1-TR000454 (Atlanta CTSA).
Footnotes
Conflicts of Interest and Sources of Funding
No conflicts of interest.
Conferences Work Presented At
AHA Epi Lifestyle, Portland OR, March 10 2017
References
- 1.Safar ME, Blacher J, Mourad JJ, et al. Stiffness of Carotid Artery Wall Material and Blood Pressure in Humans. Stroke. 2000;31:782–790. doi: 10.1161/01.str.31.3.782. [DOI] [PubMed] [Google Scholar]
- 2.Laurent S, Boutouyrie P. The Structural Factor of Hypertension: Large and Small Artery Alterations. Circ Res. 2015;116:1007–1021. doi: 10.1161/CIRCRESAHA.116.303596. [DOI] [PubMed] [Google Scholar]
- 3.Mitchell GF. Effects of central arterial aging on the structure and function of the peripheral vasculature: implications for end-organ damage. J Appl Physiol. 2008;105:1652–1660. doi: 10.1152/japplphysiol.90549.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Najjar SS, Scuteri A, Shetty V, et al. Pulse wave velocity is an independent predictor of the longitudinal increase in systolic blood pressure and of incident hypertension in the Baltimore Longitudinal Study of Aging. Journal of the American College of Cardiology. 2008;51:1377–1383. doi: 10.1016/j.jacc.2007.10.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.O’Rourke MF, Safar ME. Relationship Between Aortic Stiffening and Microvascular Disease in Brain and Kidney. Hypertension. 2005;46:200–204. doi: 10.1161/01.HYP.0000168052.00426.65. [DOI] [PubMed] [Google Scholar]
- 6.Tzourio C, Laurent S, Debette S. Is Hypertension Associated With an Accelerated Aging of the Brain? Hypertension. 2013;63:894–903. doi: 10.1161/HYPERTENSIONAHA.113.00147. [DOI] [PubMed] [Google Scholar]
- 7.Zeki Al Hazzouri A, Newman AB, Simonsick E, et al. Pulse wave velocity and cognitive decline in elders: the Health, Aging, and Body Composition Study. Stroke. 2013;44(2):388–93. doi: 10.1161/STROKEAHA.112.673533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Waldstein SR, Rice SC, Thayer JF, et al. Pulse pressure and pulse wave velocity are related to cognitive decline in the Baltimore Longitudinal Study of Aging. Hypertension. 2008;51(1):99–104. doi: 10.1161/HYPERTENSIONAHA.107.093674. [DOI] [PubMed] [Google Scholar]
- 9.Briet M, Collin C, Karras A, et al. for the Nephrotest Study Group. Arterial Remodeling Associates with CKD Progression. J Am Soc Nephrol. 2011;22(5):967–974. doi: 10.1681/ASN.2010080863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ben-Shlomo Y, Spears M, Boustred C, et al. Aortic pulse wave velocity improves cardiovascular event prediction: an individual participant meta-analysis of prospective observational data from 17.635 subjects. J Am Coll Cardiol. 2014;63(7):636–46. doi: 10.1016/j.jacc.2013.09.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vlachopoulos C, Aznaouridis K, O’Rourke MF, et al. Prediction of cardiovascular events and all-cause mortality with central haemodynamics: a systematic review and meta-analysis. Eur Heart J. 2010;31(15):1865–71. doi: 10.1093/eurheartj/ehq024. [DOI] [PubMed] [Google Scholar]
- 12.Sutton-Tyrrell K, Najjar SS, Boudreau RM, et al. Health ABC Study. Elevated aortic pulse wave velocity, a marker of arterial stiffness, predicts cardiovascular events in well-functioning older adults. Circulation. 2005;111:3384–3390. doi: 10.1161/CIRCULATIONAHA.104.483628. [DOI] [PubMed] [Google Scholar]
- 13.Mattace-Raso FUS, Van der Cammen TJM, Hofman A, et al. Arterial stiffness and risk of coronary heart disease and stroke - The Rotterdam Study. Circulation. 2006;113:657–663. doi: 10.1161/CIRCULATIONAHA.105.555235. [DOI] [PubMed] [Google Scholar]
- 14.Van Sloten TT, Sedaghat S, Laurent S, et al. Carotid stiffness is associated with incident stroke: a systematic review and individual participant data meta-analysis. J Am Coll Cardiol. 2015;66(19):2116–2125. doi: 10.1016/j.jacc.2015.08.888. [DOI] [PubMed] [Google Scholar]
- 15.Yuan C, Wang J, Ying M. Predictive Value of Carotid Distensibility Coefficient for Cardiovascular Disease and All-Cause Mortality: A Meta Analysis. PLOS One. 2016;11(4) doi: 10.1371/journal.pone.0152799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zanni MV, Schouten J, Grinspoon SK, et al. Risk of coronary heart disease in patients with HIV infection. Nat. Rev. Cardiol. 2014;11:728–741. doi: 10.1038/nrcardio.2014.167. [DOI] [PubMed] [Google Scholar]
- 17.Marcus JL, Leyden WA, Chao CR, et al. HIV infection and incidence of ischemic stroke. AIDS. 2014;28:1911–1919. doi: 10.1097/QAD.0000000000000352. [DOI] [PubMed] [Google Scholar]
- 18.Hanna DB, Post WS, Deal JA, et al. HIV Infection Is Associated With Progression of Subclinical Carotid Atherosclerosis. Clinical Infectious Diseases. 2015;61:640–650. doi: 10.1093/cid/civ325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Post WS, Budoff M, Kingsley L, et al. Associations between HIV infection and subclinical coronary atherosclerosis. Ann Intern Med. 2014;160(7):458–67. doi: 10.7326/M13-1754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Seaberg EC, Benning L, Sharrett R, et al. Association Between Human Immunodeficiency Virus Infection and Stiffness of the Common Carotid Artery. Stroke. 2010;41:2163–2170. doi: 10.1161/STROKEAHA.110.583856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kaplan RC, Sinclair E, Landay AL, et al. T cell activation predicts carotid artery stiffness among HIV-infected women. Atherosclerosis. 2011;217:207–213. doi: 10.1016/j.atherosclerosis.2011.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Maia-Leite LH, Catez E, Boyd A, et al. Aortic stiffness aging is influenced by past profound immunodeficiency in HIV-infected individuals; results from the ESAS-HIV (EValuation of Aortic Stiffness in HIV-infected individuals) J Hypertens. 2016;34(7):1338–1346. doi: 10.1097/HJH.0000000000000957. [DOI] [PubMed] [Google Scholar]
- 23.Heaton RK, Franklin DR, Ellis RJ, et al. HIV-associated neurocognitive disorders before and during the era of combination antiretroviral therapy: differences in rates, nature, and predictors. J Neurovirol. 2011;17(1):3–16. doi: 10.1007/s13365-010-0006-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Maki PM, Rubin LH, Valcour V, et al. Cognitive Function in Women with HIV: Findings from the Women’s Interagency HIV Study. Neurology. 2015;84(3):231–240. doi: 10.1212/WNL.0000000000001151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Crystal HA, Weedon J, Holman S, et al. Associations of cardiovascular variables and HAART with cognition in middle-aged HIV-infected and uninfected women. J Neurovirol. 2011;17(5):469–476. doi: 10.1007/s13365-011-0052-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fabbiani M, Ciccarelli N, Tana M, et al. Cardiovascular risk factors and carotid intima-media thickness are associated with lower cognitive performance in HIV-infected patients. HIV Med. 2013;14:136–144. doi: 10.1111/j.1468-1293.2012.01044.x. [DOI] [PubMed] [Google Scholar]
- 27.Valcour VV, Shikuma CM, Watters MR, Sacktor NC. Cognitive impairment in older HIV-1-seropositive individuals: prevalence and potential mechanisms. AIDS. 2004;18:S79–86. doi: 10.1097/00002030-200401001-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Barkan SE, Melnick SL, Preston-Martin S, et al. The Women’s Interagency HIV Study. WIHS Collaborative Study Group. Epidemiology. 1998;9:117–125. [PubMed] [Google Scholar]
- 29.Bacon MC, von Wyl V, Alden C, et al. The Women’s Interagency HIV Study: an observational cohort brings clinical sciences to the bench. Clin Diagn Lab Immunol. 2005;12:1013–1019. doi: 10.1128/CDLI.12.9.1013-1019.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Reitan R. Manual for Administration of Neuropsychological Test Batteries for Adults and Children. Tuscon, AZ: Reitan Neuropsychology Laboratories, Inc; 1978. [Google Scholar]
- 31.Smith A. The Symbol-Digit Modalities Test: A neuropsychologic test for economic screening of learning and other cerebral disorders. Learning Disorders. 1968;3:83–9. [Google Scholar]
- 32.Manly JJ, Smith C, Crystal HA, et al. Relationship of ethnicity, age, education and reading level to speed and executive function among HIV+ and HIV− women. J Clin Exp Neuropsychol. 2011;33(8):853–863. doi: 10.1080/13803395.2010.547662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Laurent S, Cockcroft J, Van Bortel L, et al. Expert consensus document on arterial stiffness: methodological issues and clinical applications. European Heart Journal. 2006;21(1):2588–2605. doi: 10.1093/eurheartj/ehl254. [DOI] [PubMed] [Google Scholar]
- 34.Wilkinson GS. Wide Range Achievement Test 3 - Administration Manual. Wilimington, DE: Jastak Associates, Inc; 1993. [Google Scholar]
- 35.Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. Applied Psychological Measurement. 1977;1:385–401. [Google Scholar]
- 36.Tien PC, Schneider MF, Cox C, et al. Association of HIV infection with incident diabetes mellitus: impact of using hemoglobin A1C as a criterion for diabetes. J Acquir Immune Defic Syndr. 2012;61(3):334–340. doi: 10.1097/QAI.0b013e31826bfc32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Levey AS, Stevens LA, Schmid CH, et al. CKD-EPI. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–12. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.D’Agostino RB, Vasan RS, Pencina MJ, et al. General Cardiovascular Risk Profile for Use in Primary Care: The Framingham Heart Study. Circulation. 2008;117:743–753. doi: 10.1161/CIRCULATIONAHA.107.699579. [DOI] [PubMed] [Google Scholar]
- 39.Friis-Moller N, Ryom L, Smith C, et al. An updated prediction model of the global risk of cardiovascular disease in HIV-positive persons: The Data-collection on Adverse Effects of Anti-HIV Drugs (D:A:D) study. Eur J Prev Cardiol. 2016;23(2):214–223. doi: 10.1177/2047487315579291. [DOI] [PubMed] [Google Scholar]
- 40.Raghunathan TE, Lepkowski JM, Hoewyk JV. A Multivariate Technique for Multiply Imputing Missing Values Using a Sequence of Regression Models. Survey Methodology. 2001;27(1):85–95. [Google Scholar]
- 41.Kaplan RC, Hanna DB, Kizer JR. Recent Insights Into Cardiovascular Disease (CVD) Risk Among HIV-Infected Adults. Current HIV/AIDS Reports. 2016;13(1):44–52. doi: 10.1007/s11904-016-0301-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sheridan LK, Fitzgerald HE, Adams KM, et al. Normative Symbol Digit Modalities Test performance in a community-based sample. Archives of Clinical Neuropsychology. 2006;21(1):23–28. doi: 10.1016/j.acn.2005.07.003. [DOI] [PubMed] [Google Scholar]
- 43.Tombaugh TN. Trail Making Test A and B: Normative data stratified by age and education. Archive of Clinical Neuropsychology. 2004;19(2):203–214. doi: 10.1016/S0887-6177(03)00039-8. [DOI] [PubMed] [Google Scholar]
- 44.Saylor D, Dickens AM, Sacktor N, et al. HIV-associated neurocognitive disorder – pathogenesis and prospects for treatment. Nat Rev Neurol. 2016;12(4):234–248. doi: 10.1038/nrneurol.2016.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rubin LH, Springer G, Maki PM, et al. Womens Interagency HIV Study Neurocognitive Working Group. Cognitive trajectories over 4 years among HIV-infected women with optimal viral suppression. CROI Conference Abstracts. 2017 doi: 10.1212/WNL.0000000000004491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sun D, Wu Y, Yuan Y, et al. Is the atherosclerotic process accentuated under conditions of HIV infection, antiretroviral therapy, and protease inhibitor exposure? Meta-analysis of the markers of arterial structure and function. Atherosclerosis. 2015;242(1):109–116. doi: 10.1016/j.atherosclerosis.2015.06.059. [DOI] [PubMed] [Google Scholar]
- 47.Gorelick PB, Scuteri A, Black SE, et al. Vascular Contributions to Cognitive Impairment and Dementia: A Statement for Healthcare Professionals from the American Heart Association/American Stroke Association. Stroke. 2011;42:2672–2713. doi: 10.1161/STR.0b013e3182299496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Smith EE, Salat DH, Jeng J, et al. Correlations between MRI white matter lesion location and executive function and episodic memory. Neurology. 2011;76(17):1492–1499. doi: 10.1212/WNL.0b013e318217e7c8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Paini A, Boutouyrie P, Calvet D, et al. Carotid and Aortic Stiffness: Determinants of Discrepancies. Hypertension. 2006;47:371–376. doi: 10.1161/01.HYP.0000202052.25238.68. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.