Abstract
In 2013, the University of California, Biomedical Research, Acceleration, Integration, and Development (UC BRAID) convened a regional network of contracting directors from the five University of California (UC) health campuses to: (i) increase collaboration, (ii) operationalize and measure common metrics as a basis for performance improvement efforts, and (iii) identify and implement best practices to maintain a competitive edge in the field of biomedical research.
This article summarizes an 18-month examination of performance metrics across the five campuses, including methods for data collection and harmonization agreed upon by the UC contracting offices.
Some of the most striking, and previously unmeasured, results demonstrate that master agreements are a highly effective and successful strategy for significantly shortening the average time to completion of contract terms. Interestingly, clinical research organizations (CROs) significantly increase the time required to negotiate a contract. Results also point to the intra- and inter-organizational dependencies affecting time to contract execution.
Systematic and transparent data collection among UC stakeholders is necessary to continue improvements to the system. However, data collection is only one component of the larger need to develop shared technology both within and between institutions. This study demonstrates that collaboration and sharing of contract terms is an effective way to reduce study activation time, but the greatest improvements will be driven by the sharing of data and technology tools.
Introduction
Sponsored projects offices face increasing internal and external pressure to improve turnaround times for clinical trial contract execution a critical measurement when interfacing with industry sponsors and funding agencies. At the same time, the proliferation of multisite clinical trials provides opportunities for affiliated institutions to harmonize and align contract negotiations. The University of California Biomedical Research, Acceleration, Integration, and Development (UC BRAID) convened the Contracting Network (the Network), comprised of the contracting directors from five University of California (UC) academic medical campuses (UCs Davis, Irvine, Los Angeles, San Diego, and San Francisco) to create an adaptive system that shares terms and best practices and leverages resources.
The Network’s charter led to an 18-month effort to achieve several aims: (i) measure and improve industry-sponsored clinical trial contracting performance, (ii) increase collaboration for multisite clinical trial agreement (CTA) negotiations, (iii) mitigate redundant efforts and conserve resources, and (iv) share best practices and lessons learned with internal stakeholders and the larger research administration community. Affirming the importance of such examination, the National Center for Accelerating Translational Science (NCATS) recently launched a similar performance improvement initiative for the Clinical and Translational Science Award (CTSA) Program and views such improvement in administrative and clinical research processes among its top priorities (Dilts et al., 2012; NCATS, 2014; UC BRAID, 2012).
Led by the contracting directors, the Network also included BRAID leadership, CTSA evaluators, and a policy manager from the UC Office of the President. The directors agreed upon a common set of metrics applicable across sites and collected standardized data on more than 1,000 new clinical trial contracts and amendments. Although the contracting offices at each campus vary in size and scope of operations, electronic systems, and resources, the directors were able to develop a rich, metrics-based foundation and a performance improvement initiative worth pursuing by all members. Ultimately, this platform of close collaboration allows UC to quickly respond to a changing contracting landscape, identify and implement evidence-based best practices, and maintain a competitive edge in the field of biomedical research.
Methods
The objective of this study was to collect data on industry-sponsored clinical trials related to: (i) contract negotiation duration, (ii) the frequency of delays related to ancillary approval processes (e.g., IRB, coverage analysis, financial interest review), and (iii) overall duration: from the receipt of the minimum documents required to negotiate the contract to contract execution. Because the scope of responsibility varied for each contracting office, the directors each identified their essential minimum documents necessary to begin a negotiation and agreed that this would be the starting point for assessing the overall duration of contract negotiation. The group assigned the phrase “Minimum Documents” to this starting point, which generally included:
Campus intake forms
Study protocol
Draft budget and contract
Draft consent forms
University economic interest disclosures
Inclusion criteria for the contracts in this study were: (1) industry-sponsored trials; (2) meeting the 2006 UC Definition of Clinical Trials (http://www.ucbraid.org/ct); and (3) where “Minimum Documents” were received by the negotiating campus from July 1, 2013 through December 31, 2014. The Network collected data at all five sites using Vanderbilt University’s REDCap technology (https://projectredcap.org/) coupled with a unified survey form (available at: http://www.ucbraid.org/rmr). The Network then partnered with UCLA’s CTSA evaluation team for statistical analysis.
Data Cleaning and Normalization
Throughout and at the close of the collection period, the Network verified entries for accuracy and consistency, with a final sample of n=981 contracts included in the database. The sponsor and clinical research organization (CRO) names were then normalized to eliminate inconsistencies in data input. Incomplete or withdrawn contracts were excluded from this study, resulting in a sample size of n=827, including n=581 new contracts and n=246 amendments.
Results
Master Contracts Are Key to Reducing Negotiation Time
To date, the UC Office of the President (UCOP) has successfully negotiated over 30 master clinical trial agreements with industry sponsors. Such agreements were used in 38% of the trials in this study (n=218), but not in the remaining 62% of cases (n=362). While anecdotal reports suggested a positive effect when using these master agreements, the impact had not been quantified until the Network’s study. Figure 1 shows two metrics: (i) average days between minimum documents received and first comments provided, and (ii) days to contract terms finalized (n=581), both with and without master agreements in place.
Figure 1.
Average Time Between Minimum Documents Received and First Comments Provided and Contract Terms Finalized by UCOP Master Use
On average, using a UCOP master agreement significantly reduced the time to complete the contracting process. Finalizing contract terms took 39 days on average with a master, versus 73 days without. This represents a significant reduction of 47% (p<.001) in negotiation time when UCOP master agreements are used.
The evidence from this study strongly demonstrates that the UCOP approach to negotiating master agreements with industry sponsors is a highly effective and successful strategy. Having master contracts in place equates with a reduction of almost 50% in average days to finalize the contract. Such a reduction can speed the launch of clinical trials, resulting in decreased costs for sponsors and institutions alike (DiMasi et al., 2003; Rijswijk-Trompert, 2012). For patients, this reduction increases access and options to participate in clinical research, and can mean the difference between participating in a clinical trial versus receiving the standard therapy for their disease.
Industry Sponsors with/without UCOP Master Agreements
Figure 2 compares industry sponsors with (n=36 sponsors/216 contracts) and without (n=224 sponsors/349 contracts) UCOP master agreements. The scatterplot in Figure 2 graphically displays the relationship among volume of contracts per sponsor, average days to finalize contract terms, and having a UCOP master agreement. Unsurprisingly, industry sponsors with master agreements had significantly more contracts on average (6 compared to 2 contracts; p=0.0002), as well as significantly lower average days to terms finalized (46 compared to 76 days; p<0.0001).
Figure 2.
Industry Sponsors with/without UCOP Master Agreements
Simply put, leveraging one contract negotiation for multiple studies is most efficient. Having a master agreement in place is a strong positive factor when analyzing study feasibility for the site, especially when considering multiple, and competing, trials. For this reason, the Network plans to: (i) expand the number of master agreements through further collaboration with the UC Office of the President and major sponsors; (ii) continue to enhance collaborations in the contracting offices at the five UC CTSAs; and (iii) research benchmarks from other CTSA institutions to compare their results with those generated by the UC BRAID Network.
Even with master agreements in place, budget negotiations may introduce a significant time delay in finalizing contracts. Budget negotiation is a key process that intersects with contract execution and the UC campuses are looking internally and across their sister sites to identify approaches to streamline this step.
CROs Increase Negotiation Time
Contracting with Clinical Research Organizations
The Network examined the average time to finalize contract terms when a Clinical Research Organization (CRO) participated in the negotiations. Figure 3 shows CRO participation in 34% of new contracts (n=566), but the most notable results in Figure 3 indicate that CRO involvement in the contracting process significantly increases the time required to negotiate the contract 65 days compared to 57 days without a CRO (14% increase in duration; p< .01).
Figure 3.
% Contracting with Clinical Research Organizations (CROs) and Average Time between Minimum Documents Received and Contract Terms Finalized with CROs
While billed as facilitating organizations, CROs can introduce complications in the negotiation process that may not exist with direct interaction between sponsors and contracting offices. The Network recognizes this as an area for concerted improvement and will devote resources to that end, such as exploration of CRO master agreements.
Local Factors Influence Turnaround Time
Contracting Compared among the Five UC Campuses
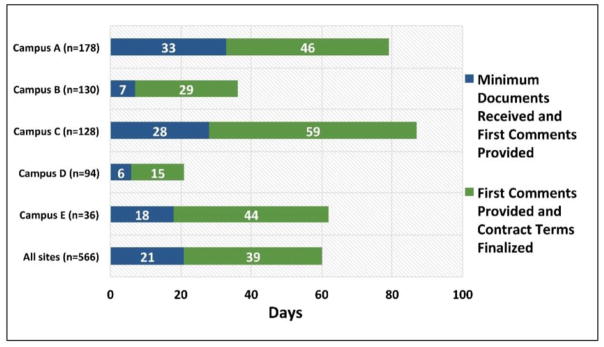
Owing to differences such as resources or the scope of responsibilities between sites, there was considerable variation in throughput for clinical trial contract execution. Time (in days) from minimum documents received to first comments provided was significantly different across the five sites (p<0.0001), ranging from a low of 6 days (campus D) to a high of 33 days (campus A). Days from first comments provided to contracting terms finalized was also significantly different (p<0.0001), ranging from a low of 15 days (campus D) to a high of 59 (campus C). In both cases, analysis of variance was used to test the mean difference in days across the five sites.
The Network’s performance improvement study started with examining minimum documents required for finalizing contract terms for each trial. It also distinguished between documents collected within the contract offices and those collected in other functional areas, within which the Network has no leadership authority or workflow control. The Network analyzed the variations in metrics among the campuses for informing the performance improvement priorities.
Other institutional factors that influence the Figure 4 results include volume and variability of contracts per campus, staffing resources (contracts per FTE), knowledge, experience, and training of staff, and useful and accessible reference guides for staff. Additionally, some contracting offices have other duties and obligations that affect performance, such as budget development and coverage analysis.
Figure 4.
Average Time between Minimum Contracts Recieved, First Comments Provided, and Contract Terms Finalized in New Contracts (n=566)
PI-initiated versus Sponsor-initiated Contracts
Historically, PI-initiated clinical trial agreements accounted for less than 5% of the industry-funded clinical trial agreements received by UC. A PI-initiated clinical trial is led by a UC faculty member who has authored the protocol and holds the Investigational New Drug Application (IND) or an Investigational Device Exemption (IDE). That number has been increasing in part because UC has been more involved in the development of treatments, making UC the sponsor of these projects.
Figure 5 shows that PI-initiated contracts (n=30) take longer to complete than sponsor-initiated contracts (n=536). PI-initiated contracts averaged 70 days to finalized terms, whereas sponsor-initiated contracts took an average of 59 days. Due to the paucity of PI-initiated contracts, this difference was not statistically significant. However, PI-initiated studies are frequently of greatest concern to faculty, despite their small number. In addition, members of the Network note that PI-initiated contracts are rapidly increasing in number and importance.
Figure 5.
Average Time between Minimum Documents Received and Contract Terms Finalized in PI-initiated versus Sponsor-initiated Contracts
PI-initiated trials require substantially more time to negotiate for several reasons. First, only a small handful of PI-initiated master agreements exist. Second, the increased intellectual contribution from UC changes the dynamic with the funding sponsor. This manifests in considerations such as inventions and intellectual property, data and publication rights, and more complicated liability and confidentiality negotiations. Finally, sponsors are typically less motivated to begin a PI-initiated study, as it is not theirs.
In some cases, and particularly with early phase studies, sponsors may be concerned about the openness of UC research, postponing the negotiation to conduct the study in a more confidential setting before making results public. Given that these types of projects are becoming more common, the Network is focused on developing resources, including master agreements, to assist contract offices in reducing the overall burden of these negotiations.
Intra- and Inter-Organizational Dependencies
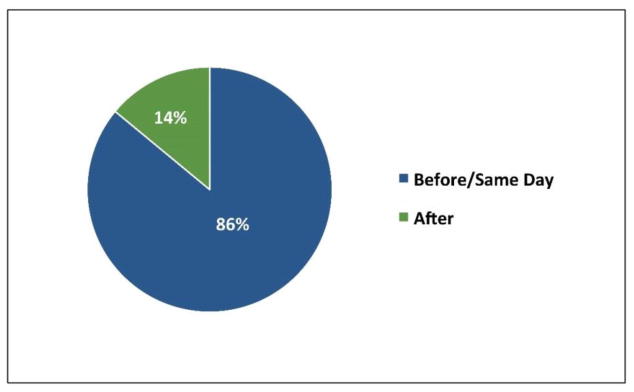
A common (mis)perception at research institutions is that contracting processes cause delays in study activation. Despite those perceptions, the study revealed that this is rarely the case within UC. In fact, of the 581 new contracts executed, 86% completed negotiation of the terms (i.e., confidentiality, publication, intellectual property) on the same day, or even before, approvals were secured for IRB, conflict of interest (COI), and/or completion of budget negotiation and coverage analysis. These results indicate that contract negotiation is not the prevalent rate-limiting step.
This is not to suggest that other institutional approvals are the lone bottleneck in the complex and interconnected march toward study activation. Rather, these results highlight the need for parallel processing, information sharing, and supportive technology. As evidenced by Anderson’s Project “Zero Delay” (Kurzrock et al., 2009), dissecting interdependent processes can liberate them from sequential or serial processing, allowing each functional unit (e.g., IRB, coverage analysis, COI, etc.) to move forward unrestrained. The gold standard is clearly parallel processing (Logan, 2002), and to increase its application, UC must work collaboratively to bring historically isolated departments together. The Network sees this as an opportunity to test and implement more efficient parallel processing procedures while breaking down silos.
The Need for Shared Technology
Realizing the full benefits of parallel processing and mitigating redundant effort requires a range of technological capabilities to share information both within and between campuses.
One real-world example of such a shared technology is UC TrialQuest, a UC-wide database of clinical trials in the process of, or having received, IRB approval over the past three years. Originally deployed in 2014, TrialQuest gives a window into the UC clinical trials portfolio with up-to-date information from the five campus IRB systems. The tool also allows contracting offices to see if a given clinical trial is underway at a sister campus, providing the opportunity to share previously approved contract terms.
UC TrialQuest is but a first step toward developing a technology platform for sharing actionable clinical trial information. Stakeholders could use this information to prevent unnecessary delays and accelerate business and regulatory processes within the clinical research enterprise. Future technology could also measure turnaround times for other functional areas, providing evidence to target specific areas for improvement.
Ultimately, accelerating clinical trial contract execution is a shared ownership of multiple stakeholders deserving of investment in human and technological resources.
Putting Data to Work
Results from this study had wide and varied impact across the UCs. Some campuses used the conclusions as a basis for local process improvements, including better integration of research administration units, for example. Others used this study as justification for hiring additional staff or restructuring teams to dedicate more officers to the negotiation process.
In particular, two campuses embarked on “Lean” process improvements building upon the success of this project. The data points and workflow mapping developed by the Network served as the foundation for these projects, and continue to be used to inform campus leadership and other stakeholders.
Future Focus
Recognizing the efficiencies gained by using master agreements, the Network will continue to work with UCOP to increase the number of agreements and broaden their use. In addition, it will explore the use and impact of the widely adopted Accelerated Clinical Trial Agreement (ACTA).
Improving the processes with respect to CROs will become a higher priority for the Network. For example, it is investigating the feasibility and benefit of CRO master agreements. The intent is to have a single agreement with each CRO that addresses their typical concerns while promoting the use of template language, including UCOP master agreements. The hope is that CRO master agreements will reduce negotiation time and cost, but further examination is required.
Resource-intensive negotiations for investigator-initiated studies require better tools and techniques to expedite contract execution. The Network will pursue solutions to this growing proportion of the clinical trials portfolio.
Multisite trials provide many opportunities to strengthen cross-campus collaboration. While outside the scope of this study, areas such as budgeting and coverage analysis have components that may be shared with sister campuses to reduce redundant efforts. Further research on the appropriate approach to enable this exchange of information will be crucial.
Finally, the Network plans to compare results with benchmarks from other institutions and the NCATS performance improvement initiative currently underway with the CTSAs.
Conclusions
The five UC academic medical centers are complex adaptive systems (McDaniel et al., 2009) that design and deploy clinical trials involving functional areas that reach well beyond the contracting offices internally. Interfacing with industry sponsors and CROs is a critical contracting function, and ripe for improvements in support of swift clinical trial activation.
Through effective collaboration and collection of metrics, the Network improved processes and identified best practices. Although characteristics of the contracting offices varied widely, the analysis revealed significant trends shared by all five UCs, such as the positive impact of master agreements, longer turnaround times when working with CROs, and delays involving other campus offices (e.g., coverage analysis, IRB, conflict of interest).
Now, with a rich metrics-based foundation, the Network will address performance gaps and develop further infrastructure supporting multisite research. Developing IT tools and resources to handle the increasing volume of complex studies will be a critical step toward reducing time to study activation. However, the human element must not be ignored. Such tools and resources allow the UC to quickly respond to a changing contracting landscape, identify and implement best practices, and maintain a competitive edge in the field of biomedical research.
Figure 6.
% of Contract Terms Finalized Same Day/Before Final Documents Approved (n=581)
Acknowledgments
1. National Center for Advancing Translational Sciences (NCATS), Clinical and Translation Science Award (CTSA) grants for the five University of California, Biomedical Research, Acceleration, Integration, and Development (UC BRAID) CTSAs. The content is solely the responsibility of the co-authors and does not necessarily represent the official views of the National Institutes of Health (NIH).
UC Davis, Clinical and Translational Science Center funded through NCATS grant number UL1 TR001860; http://www.ucdmc.ucdavis.edu/ctsc/audience/investigators/ctsc_publication_citing.html,
UC Irvine, Institute for Clinical and Translational Science funded through NCATS grant number UL1 TR001414, http://www.icts.uci.edu/citing-the-icts.asp,
UC Los Angeles, Clinical and Translational Science Institute funded through NCATS grant number UL1 TR001881, https://ctsi.ucla.edu/pages/nih-acknowledgement,
UC San Diego, Clinical and Translational Research Institute funded through NCATS grant numbers UL1 TR000100 and UL1 TR001442, https://ctri.ucsd.edu/about/Grant-support/Pages/about-publications.aspx,
UC San Francisco, Clinical and Translational Science Institute funded through NCATS grant numbers UL1 TR000004 and UL1 TR001872, http://accelerate.ucsf.edu/cite.
2. The article is based on a presentation provided at the National Council of University Research Administrators (NCURA) 58th Annual Meeting, “Collaboration in Action: Improving Contracting Performance Across the University of California”, Washington, DC, August 7–10, 2016.
Biographies
LeeAnna Bowman-Carpio is a research associate in the Evaluation Core at the UCLA Clinical and Translational Science Institute. She graduated from UCLA, receiving her B.A. in anthropology with a focus on qualitative research and analysis. As a research associate in the UCLA Clinical and Translational Science Institute, she collaborates with two UC BRAID teams to: (i) provide research support to the Contracting Network, and (ii) analyze survey data to inform the design of a UC-wide virtual biobank.
Nate Buscher is the program manager for UC BRAID, a network of the five University of California academic medical centers. With an undergraduate degree from the University of California, Los Angeles in human psychology, he manages initiatives to improve research administration across the five UCs, including the BRAID Contracting Network and regulatory efforts. Nate comes to this role with administration experience in academia, government, and non-profit sectors. He also has a strong background in information technology, operations, and fundraising. Nate is a member of NCURA and PRIM&R, and presents at numerous nationwide conferences.
Pamela Davidson is the leader of the Evaluation Core for the UCLA Clinical and Translational Science Institute. Dr. Davidson’s expertise is in evaluation research design and methods, strategic planning, and leadership, team, and organizational performance in health research organizations. Her professional effort is shared as leader of the Evaluation Core for the UCLA Clinical and Translational Science Institute, and co-leader of the NIH-sponsored Diversity Program Consortium Center for Coordination and Evaluation at UCLA. Additionally, she is on the faculty of the Department of Health Policy and Management, UCLA Fielding School of Public Health, where she teaches courses in evaluation and health systems and organizations.
Jennifer J. Ford is the director of the Office of Clinical Trials Administration (OCTA) at the University of California, San Diego whose purview is industry sponsor-initiated clinical trials. She has been the director of OCTA since 2012 and has been working at UC San Diego for 16 years in various research administration roles. Many of those years as a contract manager for a central contract and grant office, which handles federal, non-profit, and for-profit research and clinical research. Since she has been the director of OCTA, she has worked on many process analysis and improvement initiatives like the UC BRAID Contracting Network. She received her B.A. in business and master’s degree in business administration.
Erick Jenkins is the manager of Health System Contracts, Clinical Trials and Health Affairs at the University of California, Davis. Erick received a B.S. in biological psychology, an M.S. in psychology and a JD from UC Davis. He began work at UC Davis as a research contracts officer in the Sponsored Programs Office and then accepted a position at the Health System as a contracts officer in charge of health affairs and business agreements for the health system. Soon after, industry clinical trial contract negotiation was moved to the UC Davis Health System and Erick became manager of health affairs and clinical trial contracts for the UC Davis Health System.
Hillary Noll Kalay is counsel for health law for the University of California. She received her JD from New York University and her B.A. and MPP from the University of California at Berkeley. Hillary advises university stakeholders on clinical research matters, as well as procurement for the university’s health system. Hillary previously served as research policy manager at the University of California Office of the President, where she developed and advised on university clinical research policies and negotiated master clinical trial agreements for the university.
Terry Nakazono is the senior researcher/data analyst at the David Geffen School of Medicine at UCLA and the UCLA Clinical and Translational Science Institute. His expertise is in research methods and data analysis applied to multiple research and evaluation initiatives in the Center for the Health Sciences at UCLA. Among his appointments, he is a senior researcher with the UCLA Clinical and Translational Science Institute and provides data-analytic support to the UC BRAID initiatives.
Helene Orescan is the director of Clinical Trials Contracts & Strategic Relations at the University of California, Los Angeles School of Medicine. She works with sponsors and CROs negotiating complex clinical trial agreements of all phases for both sponsor- and investigator-initiated trials, as well as strategic agreements with industry partners to foster collaborative clinical research trials at UCLA. Prior to joining UCLA, she was the contracts counsel for the First Advantage Corporation, and the senior associate counsel for the Research Foundation of CUNY. She holds a B.A. in liberal arts from SUNY Stony Brook and a JD from New York Law School.
Rachael Sak is the director of the University of California Biomedical Research Acceleration, Integration, and Development (UC BRAID) consortium. Rachael received her MPH at the University of Michigan with a focus in health management and policy and researched the use of health information technology in healthcare delivery systems. She has 16 years of experience working in clinical research in the academic setting and served as associate director of the UC, San Francisco Pancreas Cancer Program at the Helen Diller Family Comprehensive Cancer Center. Prior to joining UC BRAID, Rachael was the assistant director for strategic initiatives in the Clinical & Translational Science Institute (CTSI) at UCSF.
Irene Shin is the interim director for the University of California, San Francisco Industry Contracts Division, Office of Innovation, Technology & Alliances. She received her JD from the University of California, Davis, King Hall School of Law, and her B.A. in philosophy from the University of California, Santa Cruz. She performs a leadership and advisory role at UCSF in support of the development and negotiation of basic and translational research agreements with industry. She has extensive experience in contract negotiation, developed over her twenty years as an attorney representing corporate, non-profit, and government clients.
Tam Tran is the assistant director of sponsored projects for the University of California, Irvine. Her office has responsibility for the leadership and operations management of the clinical trial contracting unit at UC Irvine. She has 15 years of experience in contracts and grants research administration with experience in both pre- and post-award (non-financial) functions. She received both her B.S. in biology and M.P.H. in public health with a focus in health services administration from the University of California, Los Angeles and has attained the designation of a certified research administrator.
Contributor Information
Tam Tran, University of California at Irvine.
LeeAnna Bowman-Carpio, University of California at Los Angeles.
Nate Buscher, University of California Biomedical Research Acceleration, Integration, and Development.
Jennifer J. Ford, University of California at San Diego
Erick Jenkins, University of California at Davis.
Hillary Noll Kalay, University of California Office of the President.
Terry Nakazono, University of California at Los Angeles.
Helene Orescan, University of California at Los Angeles.
Rachael Sak, University of California Biomedical Research Acceleration, Integration, and Development.
Irene Shin, University of California at San Francisco.
Pamela Davidson, University of California at Los Angeles.
References
- Accelerated Clinical Trial Agreement. ( https://www.ara4us.org)
- Dilts MD, Rosenblum D, Trochim WM. A virtual national laboratory for reengineering clinical translational science. Science Translational Medicine. 2012;4(118) doi: 10.1126/scitranslmed.3002951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiMasi JA, Hansen RW, Grabowski HG. The price of innovation: New estimates of drug development costs. Journal of Health Economics. 2003;22(2):151–185. doi: 10.1016/S0167-6296(02)00126-1. [DOI] [PubMed] [Google Scholar]
- Kurzrock R, Pilat S, Bartolazzi M, Sanders D, Van Wart Hood J, Tucker SD, Webster K, Mallamaci MA, Strand S, Babcock E, Bast RC. Project Zero Delay: A process for accelerating the activation of cancer clinical trials. Journal of Clinical Oncology. 2009;27(26):4433–4440. doi: 10.1200/JCO.2008.21.6093. [DOI] [PubMed] [Google Scholar]
- Logan GD. Parallel and serial processing. Chapter 7 in Steven’s handbook of experimental psychology. In: Pashler H Ed.-in-Chief, Wixted J, editors. Volume 4: Methodology in experimental psychology. 3. New York: John Wiley & Sons, Inc; 2002. [Google Scholar]
- McDaniel RR, Lanham HJ, Anderson RJ. Implications of complex adaptive systems theory for the design of research on health care organizations. Health Care Management Review. 2009;34(2):191–199. doi: 10.1097/HMR.0b013e31819c8b38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National Center for Accelerating Clinical and Translational Science (NCATS) Advisory Council Working Group on the Institute of Medicine (IOM) Report: The CTSA program at NIH. A Working Group of the NCATS Advisory Council to the Director. 2014 Retrieved from: http://www.ncats.nih.gov/files/CTSA-IOM-WG-Draft-Report.pdf.
- REDCap. n.d ( https://projectredcap.org/)
- Rijswijk-Trompert M. Clinical trial agreement negotiations. Applied clinical trials. 2012 Retrieved from: http://www.appliedclinicaltrialsonline.com/clinical-trial-agreement-negotiations.
- UC BRAID: University of California, Biomedical Research Acceleration Integration and Development. 2015 http://www.ucbraid.org/2015.
- UC BRAID Executive Committee, UC BRAID Evaluation Network, Clinical and Translational Science Awards (CTSA), Evaluation Key Function Committee. UC BRAID: Co-creating and evaluating performance in a regional laboratory for conducting translational science. Bethesda, MD: UC BRAID: Biomedical Research Acceleration, Integration and Development; 2012. [Google Scholar]
- University of California. Clinical trials definition. n.d Retrieved from: http://www.ucbraid.org/ct.