Abstract
Background
Circadian misalignment may increase the risk of developing hepatocellular carcinoma (HCC). The aim of this study was to examine the association between distance from time zone meridian, a proxy for circadian misalignment, and HCC risk in the U.S. adjusting for known HCC risk factors.
Methods
Surveillance, Epidemiology, and End Results (SEER) provided information on 56,347 HCC cases diagnosed between 2000 and 2014 from 16 population-based cancer registries in the U.S. Distance from time zone meridian was estimated using the location of each SEER county’s Center of Population in a geographic information system (GIS). Poisson regression with robust variance estimation was used to calculate incidence rate ratios (IRRs) and 95% confidence intervals (CIs) for the association between distance from time zone meridian and HCC risk adjusting for individual-level age at diagnosis, sex, race/ethnicity, year of diagnosis, SEER registry, and county-level prevalence of health conditions, lifestyle factors, shift work occupation, socioeconomic status, and demographic and environmental factors.
Results
A 5 degree increase in longitude moving east to west within a time zone was associated with a statistically significant increased risk for HCC (IRR 1.07, 95% CI 1.01–1.14, p = 0.03). A statistically significant positive association was observed among those <65 years old, while no association was observed among individuals ≥65 years old (p for interaction < 0.01).
Conclusions
Circadian misalignment from residing in the western region of a time zone may impact hepatocarcinogenesis.
Impact
Circadian misalignment may be an independent risk factor for HCC.
Keywords: circadian misalignment, circadian disruption, social jetlag, liver cancer, hepatocellular carcinoma
Introduction
Circadian rhythms play critical roles in many biological processes, including the timing of sleep and wake, immune function, metabolism, hormone secretion, and gene regulation (1). A master circadian pacemaker located in the suprachiasmatic nucleus (SCN) in the hypothalamus controls the rhythms of biological clocks in central and peripheral tissues to maintain a period of approximately 24 hours. The SCN relies on exposure to light and other environmental signals (or Zeitgebers) to align these endogenous rhythms with our environment (2,3). Circadian misalignment (often referred to as circadian disruption) occurs when an endogenous circadian rhythm and environmental rhythms, such as the 24-hour light/dark cycle, are out of phase, and has been associated with adverse human health outcomes including cancer (4–8). Common habitual sources of chronic circadian misalignment include shift work and social jetlag, which occurs when work schedules and other social commitments require wakefulness at times during which sleep would otherwise occur (9,10). Although all individuals located in a time zone adhere to a common social time, as the sun progresses from east to west, the western region of a time zone is subject to less light exposure early in the morning and greater light exposure later in the day compared to those residing in the eastern region of a time zone (11,12). This differential exposure to light has been suggested to increase the likelihood of a later phase (i.e., time on the biological clock) of entrainment (i.e., daily resetting of the clock) to the 24-hour day in individuals in the western region of a time zone (who are still expected to awaken at the same local clock time for work or school obligations), leading to a chronic misalignment between biological time and daily social and occupational commitments (11). The mismatch between sun time and local clock time, as a proxy for circadian misalignment or disruption, increases with greater distance west within a time zone.
Experimental evidence suggests a potential role for circadian misalignment in the development of hepatocellular carcinoma (HCC). HCC, the most commonly occurring histological type of primary liver cancer, has been increasing in incidence and mortality in many countries across the world (13). In the U.S., a large proportion of HCC cases (40.5%) are not explained by established risk factors for HCC such as chronic hepatitis C virus (HCV), chronic hepatitis B virus (HBV), alcohol consumption, diabetes, and obesity (14). Circadian regulation plays a critical role in liver metabolism (15). An estimated 10% of the liver transcriptome is rhythmically expressed, including genes involved in bile acid synthesis and metabolism and glucose regulation (15). Bile acid synthesis in the liver is a tightly regulated process that mediates absorption of dietary fats, nutrients, and vitamins, and is under circadian regulation, synchronized with periods of feeding and fasting to maintain efficient nutrient use and storage (15). Chronic circadian disruption has been associated with increased bile acid levels, which have been shown to increase liver cancer risk (16,17). Chronic circadian disruption also promotes liver cancer in mice exposed to diethylnitrosamine, a hepatocarcinogen (18). Further, circadian disruption has been associated with glucose dysregulation and increased risk for type 2 diabetes, which is a risk factor for HCC (19). Gu et al. (2017) conducted an epidemiologic study demonstrating that western position in a time zone is associated with an increased risk for liver and intrahepatic bile duct cancer among whites, although the exposure assessment did not account for the Earth’s shape or for latitude-based differences in distance (20).
The objective of this study was to conduct the first prospective analysis examining the association between circadian misalignment as measured by residential distance from time zone meridian and HCC risk in the U.S. adjusting for HCC risk factors. An additional objective of this study was to further explore whether patterns of the association differed by specific factors such as age and shift work occupation.
Materials and Methods
Study population
Surveillance, Epidemiology, and End Results (SEER) is a U.S. National Cancer Institute (NCI) program that ascertains person-level information on cancer incidence, treatment, and survival from population-based cancer registries capturing 28% of the U.S. population (21). We included the following 16 cancer registries in the analysis: Atlanta (metropolitan), Greater California, Connecticut, Detroit (metropolitan), Greater Georgia, Iowa, Kentucky, Los Angeles, Louisiana (excluding July-December 2005 cases due to Hurricanes Katrina and Rita), New Jersey, New Mexico, Rural Georgia, San Francisco-Oakland, San Jose-Monterey, Seattle (Puget Sound), and Utah. We excluded registries that restricted coverage to specific subpopulations and registries located outside of the contiguous U.S. without available information on ultraviolet (UV) radiation. UV is a factor suspected to be associated with HCC risk (22,23) and was a priori determined to be included in modeling. The study area included all 607 counties located in the catchment areas captured by the 16 SEER registries included in the analysis. As SEER does not provide personal identifiers to protect patient confidentiality, this study was exempt from Institutional Review Board review.
Outcomes
We defined HCC cases using International Classification of Diseases for Oncology, Third Edition (ICD-O-3) topography code C22.0 for primary liver cancer and ICD-O-3 histology codes 8170-8175 (24). Cases were diagnostically confirmed through microscopic confirmation (e.g., positive histology), positive laboratory test/marker study, direct visualization, or radiography excluding clinical diagnosis only; with a sequence number of one primary only; diagnosed between 2000 and 2014; and not reported via autopsy or death certificate only (Supplementary Figure S1) (22,25). For each SEER county, the counts of HCC cases were stratified by age at diagnosis (<65 years, ≥65); sex (male, female); race/ethnicity (non-Hispanic white, non-Hispanic black, non-Hispanic Asian/Pacific Islander/American Indian/Alaska Native, Hispanic); year of diagnosis (2000–2007, 2008–2014); and SEER registry (22,26).
Exposure assessment
The county of residence at diagnosis was available for each case from SEER. For each county in the study area, distance from time zone meridian was calculated as the geodesic distance in degrees of longitude between the 2000 U.S. Census Bureau county Center of Population (https://www2.census.gov/geo/pdfs/reference/cenpop2010/COP2010_documentation.pdf) and the time zone meridian applying the Vincenty formula using a geographic information system (GIS) (20). The Vincenty formula accounts for the Earth’s shape as an ellipsoid (i.e., oblate spheroid) rather than a sphere (27), as well as for latitude-based (i.e., north-south) differences in distance, where the distance for 1 degree of longitude decreases moving north or south of the Equator towards the poles. The county Center of Population is the latitude and longitude of the point location in the county at which the population would balance if equally weighting the location of each person in the decennial census (https://www2.census.gov/geo/pdfs/reference/cenpop2010/COP2010_documentation.pdf). Centers of Population more realistically represent where the majority of the population in a geographic variable (e.g., county) resides compared to geographic centroids. The time zone meridian for the Pacific, Mountain, Central, and Eastern time zones are −120 degrees, −105 degrees, −90 degrees, and −75 degrees, respectively. Centers of Population located west of a time zone meridian were assigned positive distance values and Centers of Population located east of a time zone meridian were assigned negative distance values. The U.S. Geological Survey Small-Scale Dataset of U.S. Cities and Towns was used to estimate distances from cities to the time zone meridian (https://www.sciencebase.gov/catalog/item/581d0517e4b08da350d52386). All spatial analyses were conducted using ArcGIS 10.5.1 (Esri, Redlands, CA).
Additional covariates
The following variables were assessed as potential confounders as they are known or suspected HCC risk factors or associated with the exposure. Individual-level age at diagnosis, sex, race/ethnicity, year of diagnosis, and SEER registry were available for each case from the SEER database. County-level socioeconomic variables for median household income, educational attainment (percentage with a Bachelor’s degree or higher), percentage unemployed, poverty (percentage of individuals below the poverty level), and percentage foreign born (used as a proxy for HBV prevalence because HBV is endemic in parts of Asia and Africa (28)) were acquired from the 2000 U.S. Census Bureau Summary Files provided through SEER. Urbanicity, defined as U.S. Department of Agriculture Rural-Urban Continuum Codes 1-7 for urban and 8-9 for rural, was available from SEER. The Institute for Health Metrics and Evaluation (IHME) provided county-level data on known and suspected HCC risk factors estimated using the Behavioral Risk Factor Surveillance System (BRFSS) and National Health and Nutrition Examination Survey (NHANES) (22). IHME data included county-level sex-specific age-adjusted prevalence of alcohol consumption in 2005 (an average of >1 drink per day for women or >2 drinks per day for men in the past 30 days); total diagnosed and undiagnosed diabetes in 2000 (adults aged ≥20 years who reported a previous diabetes diagnosis and/or have a fasting plasma glucose ≥126 mg/dL and/or hemoglobin A1c ≥6.5%); any physical activity in 2001 (participation during the past month in any physical activities or exercises outside of work); obesity in 2001 (body mass index [BMI] ≥30 kg/m2); and current smoking in 2000 (currently smoking cigarettes some days).
Shift work is a source of circadian misalignment and a Group 2A probable human carcinogen according to the International Agency for Research on Cancer (IARC) (29). Shift work occupation was defined as the county-level sex-specific percentage employed in occupations in which at least 10% of that workforce regularly works night shifts, rotating shifts, or between midnight and 6 a.m. according to the U.S. Bureau of Labor Statistics (https://www.bls.gov/tus/tables/a4_1115.htm). Using the 2000 U.S. Census Bureau Summary File 3, we summed the percentages of the employed civilian population aged 16 years and over in each county working in the following shift work occupations satisfying this criterion: healthcare practitioners and technical; healthcare support; protective service; food preparation and serving-related; production, transportation, and material moving; and farmers and farm managers.
The following county-level environmental variables were estimated using GIS. Ambient UV exposure (mW/m2) in 2000 was estimated by averaging a spatiotemporal exposure model of UV created using established UV predictors (e.g., ozone) to the county level (30). UV is the primary source of circulating vitamin D levels in humans, and higher levels of UV as well as serum vitamin D have been associated with decreased risk for HCC (22,23). Ambient particulate matter air pollution <2.5 microns in diameter (PM2.5), a suggested liver cancer risk factor (31), was estimated by averaging an inverse distance-weighted interpolated raster surface of PM2.5 concentrations (μg/m3) across each county (22). The PM2.5 model was created using the U.S. Environmental Protection Agency Air Quality System database annual summary file in 2000 (22). Using the Zonal Statistics as Table tool in ArcGIS, average outdoor light at night in 2000 was estimated for each county using cloud-free composite images provided by the U.S. National Oceanic and Atmospheric Administration Global Radiance Calibrated Nighttime Lights product from the Defense Meteorological Satellite Program Operational Linescan System (https://ngdc.noaa.gov/eog/archive.html) (32). All county-level data were compiled using Federal Information Processing Standard (FIPS) codes.
Statistical analysis
We used Poisson regression with robust variance estimation to calculate incidence rate ratios (IRRs) and 95% confidence intervals (CIs) for the association between distance from time zone meridian and HCC risk. Distance from time zone meridian was examined continuously per 5 degree increase in longitude (20), where 1 degree of longitude corresponds to approximately 69 mi or 111 km at the equator, and 49 mi or 79 km at 45 degrees latitude. Restricted cubic regression splines showed no evidence of deviations from linearity. We used the natural logarithm of the county population size from the 2000 U.S. Census Bureau Summary File 3 as the offset in all models. Basic models included adjustment for age at diagnosis, sex, race/ethnicity, year of diagnosis, and SEER registry. Full multivariable models further adjusted for the following county-level variables, which are known or suspected HCC risk factors: alcohol consumption, smoking, obesity, diabetes, shift work occupation, socioeconomic factors (median household income, Bachelor’s degree education or higher, unemployment, and poverty), foreign born, urbanicity, outdoor light at night, and UV. We also evaluated potential confounding by any physical activity and PM2.5. As adjustment for these variables did not change the effect estimate for distance from time zone meridian and HCC risk (<10% change in the IRR), they were not included in the final model.
We explored potential effect modification by age, shift work occupation, alcohol consumption, urbanicity, race/ethnicity, sex, obesity, diabetes, UV, and residential mobility using stratified analyses. We performed tests for interaction between each of these factors and distance from time zone meridian by adding an interaction term to the model and using likelihood ratio tests to determine statistical significance. We hypothesized that the relationship between circadian misalignment and HCC risk may vary by these factors for the following reasons: aging is associated with earlier circadian phase and shorter sleep duration, and subsequently could reduce susceptibility to circadian misalignment from western location in a time zone (33); circadian misalignment may be elevated in areas with a higher prevalence of shift work occupation as biological adaptation to shift work schedules is rare; heavy alcohol consumption disrupts circadian rhythms that regulate neuroendocrine function (1); noise and light pollution, which disrupt circadian rhythms, are prevalent in urban areas; and there are observed racial/ethnic and gender disparities in liver cancer incidence in the U.S. Circadian disruption adversely impacts glucose metabolism through multiple mechanisms, including reduced glucose tolerance, thereby potentially increasing the risk of diabetes and obesity (34). UV exposure is also related to circadian disruption, as those who experience misalignment between their biological clock and the light/dark cycle tend to be exposed to less daytime sunlight (35). To assess potential exposure misclassification due to residential migration, we conducted stratified analyses by county-level residential mobility. Using data from the 2000 U.S. Census Bureau Summary File 1 in SEER, non-movers were defined as those residing in a county at diagnosis in which ≥51.9% (20th percentile of all counties) of the population did not migrate between 1995 and 2000, and movers were defined as those residing in a county where <51.9% did not migrate. Similar results were observed when using Poisson regression models with a random intercept for county to assess potential county-level clustering and Pearson- and deviance-based scaled Poisson models accounting for overdispersion. All statistical tests were two-sided and p < 0.05 was considered statistically significant. All statistical analyses were conducted using SAS 9.4 (SAS Institute, Cary, NC).
Results
There were 56,347 HCC cases diagnosed between 2000 and 2014 included in the analysis. As shown in Table 1, HCC cases were on average 62.4 years of age at diagnosis, predominantly non-Hispanic white, male, and/or resided in the Pacific time zone. Using the county at diagnosis for each case provided by SEER and county-level measures for known and suspected HCC risk factors, we report the average values for these variables according to the counties in which HCC cases resided. HCC cases mostly resided in counties located west of their respective time zone meridian (1.5 degrees ± 3.9) where an average of 27.8% of the population engaged in shift work occupation. HCC cases resided in counties that were predominantly urban in which an average of 8.3% of the population consumed alcohol, 23.9% smoked cigarettes, 25.7% were obese, and 11.4% had diabetes. The average median household income was $47,100 and an average of 26.1% of the population had a Bachelor’s degree education or higher, 6.5% were unemployed, and 17.9% were foreign born. Average ambient UV and PM2.5 levels were 215.0 mW/m2 and 14.6 μg/m3, respectively.
Table 1.
Characteristics of hepatocellular carcinoma cases in the U.S. diagnosed from 2000-2014
| Characteristic | Cases (N = 56,347) |
|---|---|
| Individual level | |
| Mean (SD) | |
| Age at diagnosis | 62.4 (11.6) |
| N (%) | |
| Sex | |
| Male | 43,435 (77.1) |
| Female | 12,912 (22.9) |
| Race/ethnicity | |
| Non-Hispanic white | 27,808 (49.4) |
| Non-Hispanic black | 7,623 (13.5) |
| Non-Hispanic Asian or Pacific Islander | 9,217 (16.4) |
| Non-Hispanic American Indian or Alaskan Native | 595 (1.1) |
| Hispanic | 11,104 (19.7) |
| Time zone | |
| Eastern | 16,870 (29.9) |
| Central | 4,850 (8.6) |
| Mountain | 2,300 (4.1) |
| Pacific | 32,327 (57.4) |
| Year of diagnosis | |
| 2000–2007 | 23,613 (41.9) |
| 2008–2014 | 32,734 (58.1) |
| County levela | |
| Mean (SD) | |
| Distance from time zone meridian (degrees)b | 1.5 (3.9) |
| Percent shift work occupationc | 27.8 (6.0) |
| Percent alcohol consumption | 8.3 (2.2) |
| Percent smoking status | 23.9 (4.8) |
| Percent any physical activityd | 76.9 (5.8) |
| Percent obesity (≥30 kg/m2)d | 25.7 (4.1) |
| Percent diabetes | 11.4 (1.7) |
| Median household income (per $10,000) | 47.1 (11.1) |
| Percent bachelor’s degree or higher | 26.1 (9.2) |
| Percent unemployed | 6.5 (2.3) |
| Percent poverty | 13.1 (5.3) |
| PM2.5 (μg/m3) | 14.6 (3.1) |
| UV (mW/m2) | 215.0 (39.1) |
| Percent foreign born | 17.9 (12.1) |
| N (%) | |
| Urbanicity | |
| Rural | 460 (0.8) |
| Urban | 55,887 (99.2) |
Abbreviations: kg/m2, kilogram per square meter; mW/m2, milliwatt per square meter; PM2.5, particulate matter <2.5 microns in diameter; SD, standard deviation; μg/m3, microgram per cubic meter; UV, ultraviolet radiation.
We used the county at diagnosis for cases from SEER to determine characteristics regarding the counties in which each case resided. We used GIS to estimate distance from time zone meridian, PM2.5 air pollution, and UV. We used county-level measures provided by SEER, U.S. Census Bureau, and the Institute for Health Metrics and Evaluation to estimate values for the variables listed under “County level” (e.g., occupation, lifestyle, health conditions, socioeconomic status, demographics). Please refer to the Methods section for a detailed description of each of these variables.
Positive distance values indicate the SEER county Center of Population is located west of the time zone meridian. Negative values indicate the Center of Population is located east of the meridian.
Occupations where ≥10% of the U.S. workforce regularly works night shifts, rotating shifts, or between midnight and 6 a.m.
Sex-specific any physical activity and obesity prevalence rates were averaged to estimate a total prevalence.
Figure 1 shows distance from time zone meridian (degrees) for each of the 607 counties included in the study. The inset map on the bottom left of Figure 1 shows the time zones to which each county belongs and the respective time zone meridians. The majority of the counties, such as those in the California, Utah, Iowa, Louisiana, and Georgia registries, are located west of their time zone meridian, shown in red. Some counties in the California, New Mexico, Kentucky, New Jersey, and Connecticut registries are located east of their time zone meridian, shown in blue.
Figure 1.
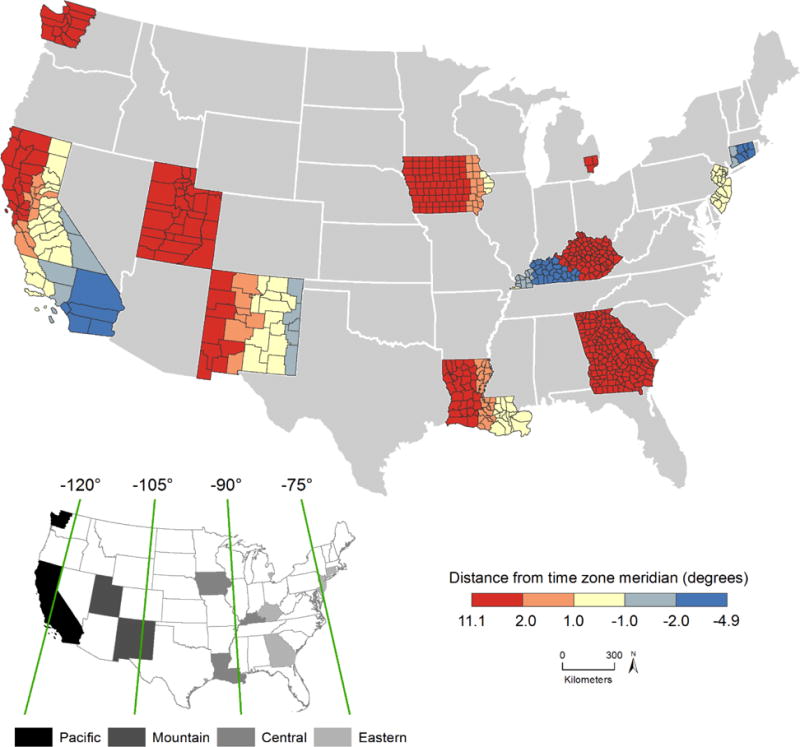
Distance from time zone meridian for the Centers of Population of 607 counties (16 SEER registries). Increasing distance from time zone meridian towards the west is shown in red, and increasing distance from time zone meridian towards the east is shown in blue. The inset map shows the time zone meridians in degrees of longitude and counties within each of the four time zones in the contiguous U.S.
In basic models adjusting for age, sex, race/ethnicity, year, and SEER registry, distance from time zone meridian was positively associated with HCC risk (IRR per 5 degree increase in longitude moving east to west within a time zone: 1.10, 95% CI 1.02-1.18, p = 0.01) (Table 2). Figure 2 shows 5 degree increases across all time zones in the contiguous U.S. After additional adjustment for county-level alcohol consumption, smoking, obesity, diabetes, shift work occupation, median household income, education, unemployment, poverty, foreign born, urbanicity, outdoor light at night, and UV, the positive association between distance from time zone meridian and HCC risk was attenuated but remained statistically significant. A 5 degree increase in longitude moving east to west within a time zone was associated with a 7% increase in HCC risk (adjusted IRR 1.07, 95% CI 1.01-1.14, p = 0.03) (Table 2). When comparing risk estimates for cities that are located in the same time zone but west and east of the meridian, residence in Seattle, WA vs. San Diego, CA is associated with a 10% increase in HCC risk, and residence in Lexington, KY vs. Hartford, CT is associated with a 10% increase in HCC risk. Model building is shown in Supplementary Table S1, and results using distance from time zone meridian moving west to east within a time zone are shown in Supplementary Table S2.
Table 2.
Associations between distance from time zone meridian and HCC incidence (SEER 2000-2014)
| Distance from time zone meridian (per 5 degree increase)a | Cases (N) | Basicb IRR (95% CI) |
P | Fully adjustedc IRR (95% CI) |
P |
|---|---|---|---|---|---|
| Main analyses | |||||
| 56,347 | 1.10 (1.02-1.18) | 0.01 | 1.07 (1.01-1.14) | 0.03 | |
| Stratified analyses | |||||
| Age (years) | <0.01 | ||||
| <65 | 33,767 | 1.11 (1.04-1.20) | |||
| ≥65 | 22,580 | 1.01 (0.94-1.10) | |||
| Race/ethnicity | <0.01 | ||||
| Non-Hispanic white | 27,808 | 1.11 (1.04–1.17) | |||
| Non-Hispanic black | 7,623 | 1.47 (1.07–2.02) | |||
| Non-Hispanic Asian, Pacific Islander, American Indian, or Alaskan Native | 9,812 | 1.50 (1.02–2.22) | |||
| Hispanic | 11,104 | 0.75 (0.68–0.84) |
Abbreviations: CI, confidence interval; HCC, hepatocellular carcinoma; IRR, incidence rate ratio; SEER, Surveillance, Epidemiology, and End Results.
Per 5 degrees in longitude moving east to west within a time zone.
Models are adjusted for individual-level age at diagnosis, sex, race/ethnicity, year of diagnosis, and SEER registry.
Models are additionally adjusted for the following county-level variables: alcohol consumption; smoking; obesity; diabetes; shift work occupation; median household income; Bachelor’s degree or higher; unemployed; poverty; foreign born; urbanicity; outdoor light at night; UV.
Figure 2.
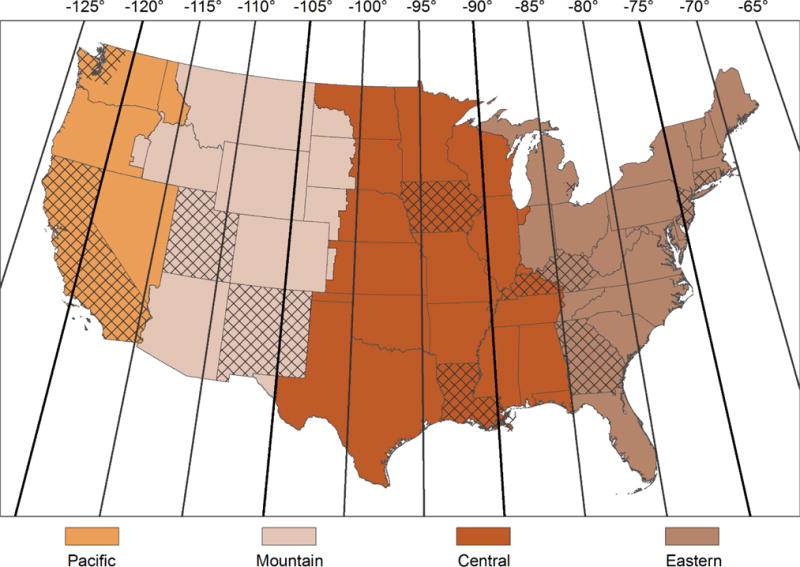
Time zones in the U.S. and 5 degree longitude meridians. The four time zones in the contiguous U.S. are shown: Pacific, Mountain, Central, and Eastern. Meridians running north to south are shown for every 5 degrees of longitude, corresponding to the units of the incidence rate ratios presented in the study. The 607 counties included in the study are shown in cross-hatch.
We observed statistically significant interactions between distance from time zone meridian and age (p for interaction < 0.01), race/ethnicity (p for interaction < 0.01), and obesity (p for interaction = 0.01) (Table 2 and Supplementary Table S3). Increasing distance from east to west within a time zone was associated with a 11% increase in HCC risk among individuals aged <65 years old (adjusted IRR 1.11, 95% CI 1.04-1.20), and was not associated with HCC risk among those aged ≥65 years old (adjusted IRR 1.01, 95% CI 0.94-1.10). Circadian misalignment was positively associated with HCC risk among non-Hispanic whites, non-Hispanic blacks, and non-Hispanic Asians, Pacific Islanders, American Indians, and Alaskan Natives; there was an inverse association among Hispanics (Table 2). Although circadian misalignment was positively associated with residing in areas with low obesity prevalence, there was no association among those residing in areas with high obesity prevalence (Supplementary Table S3). The association between distance from time zone meridian and HCC risk did not differ by sex, shift work occupation, alcohol consumption, urbanicity, diabetes, UV, or residential mobility (p for interaction > 0.05) (Supplementary Table S3).
Discussion
In this prospective analysis in the U.S., we observed a statistically significant positive association between increasing distance in longitude moving east to west within a time zone, as a measure of circadian misalignment or disruption, and HCC risk. This association remained statistically significant after adjustment for individual-level information on age at diagnosis, sex, race/ethnicity, year of diagnosis, and SEER registry, as well as county-level information on known and suspected HCC risk factors, including health behavior and lifestyle factors, shift work occupation, socioeconomic status, demographic variables, and environmental exposures. The association between distance from time zone meridian and HCC risk varied by age, where a positive association was observed among individuals aged less than 65 years while no association was observed among those aged 65 years and older. We also observed statistically significant interactions with race/ethnicity and obesity prevalence. To the best of our knowledge, this is the first epidemiologic study examining circadian misalignment and HCC risk with adjustment for HCC risk factors.
HCC incidence has been rising in some parts of the world including in the U.S. (13,36). Contrary to our habits throughout evolution, our 24/7 society does not conform to activities which align with sunrise and sunset. This, along with technological advances, such as the proliferation of artificial light, have made circadian disruption commonplace (37). Light is the most important Zeitgeber or cue that serves to synchronize human circadian timing systems (38). Although time zones demarcate boundaries created to address the varying solar exposure across the population due to the Earth’s rotation and orbit, the sun rises and sets at different social times within a given time zone (39). Due to the east-to-west movement of the sun, increasing distance west within a time zone may be a source of circadian misalignment through light exposure delaying the endogenous circadian phase and leading to misalignment between biological time and social time. One degree of longitude corresponds to approximately 4 minutes; thus, a city located 5 degrees (longitude) west of another city in a given time zone would experience a sunrise approximately 20 minutes after the city located in the east. Circadian misalignment has been associated with chronic sleep loss and adverse health conditions such as obesity, which is a risk factor for HCC (37). Further, circadian misalignment may be carcinogenic in general, as shift work is classified as an IARC probable human carcinogen (Group 2A) (40). It is noteworthy that circadian disruption may be an issue affecting many individuals in the population not limited to shift workers, and represents a modifiable risk factor. Regardless of location in a time zone, behavioral and environmental changes to promote circadian alignment and healthy sleep include limiting exposure to light at night and optimizing the timing, duration, and regularity of sleep (41). Evening light exposure can be limited by using window shades to create a dark bedroom environment. Devices which emit blue-enriched light may increase alertness and delay circadian phase, and thus should be avoided prior to bed (42). Schedules designed to align work with biological time are feasible to implement and have been shown to reduce circadian disruption (43). Policies that enable sufficient sleep, such as delayed school start times among adolescents, may also minimize disruption and benefit health (44).
Previous experimental studies have demonstrated biological plausibility for the association between circadian misalignment and HCC risk. Chronic jet lag exposure among mice induced nonalcoholic fatty liver disease and HCC and was also associated with suppression of farnesoid X receptor (FXR) and induction of constitutive androstane receptor (CAR) nuclear receptors, which are both involved in liver bile acid metabolism (16). Jet-lagged mice had higher bile acid levels, which have been associated with higher risk for HCC (16,17). Further, circadian disruption observed when meals are consumed during the biological night has been associated with changes in metabolic pathways and nutrition, and might thereby affect risk for liver cancer (45).
In the only published epidemiologic study to date (also based on SEER data), Gu et al. (2017) restricted analyses to whites only and reported a positive association between increasing distance from the time zone meridian towards the west and liver and intrahepatic bile duct cancer risk among white males adjusting for age, poverty percentage, cigarette smoking prevalence, state, and latitude (adjusted RR 1.11, 95% CI 1.05-1.18) (20). Similar results were observed among white females (adjusted RR 1.10, 95% CI 1.03-1.18) (20). Results did not change after adjustment for obesity or urbanicity, or when restricting to non-Hispanic whites. Our findings are generally in accordance with these results. We observed a statistically significant positive association between increasing distance moving east to west within a time zone and HCC risk (IRR 1.07, 95% CI 1.01-1.14). However, although we observed a statistically significant positive association among non-Hispanic white males (IRR 1.13, 95% CI 1.06-1.20), in comparison to Gu et al. (2017), we did not observe an association among non-Hispanic white females (IRR 1.03, 95% CI 0.95-1.12) (20). These two studies differed in both exposure assessment and adjustment for potential confounders. Specifically, in contrast to the earlier report (20), our exposure metric accounted for the Earth’s ellipsoidal shape and consequent latitude-based differences in the calculations for distance in longitude between the county Center of Population and time zone meridian. We also adjusted for known and suspected HCC risk factors, including age, race/ethnicity, county-level information on the prevalence of alcohol consumption, smoking, obesity, diabetes, foreign born as a proxy for HBV prevalence, urbanicity as it has been associated with healthcare access as well as higher HCV prevalence (46), and socioeconomic status. Our study results were also adjusted for shift work occupation (a major source of circadian disruption) (40), outdoor light at night, and ambient UV exposure rather than latitude as a proxy used by Gu et al. (2017) (20). Further, we examined HCC only, the most common histological subtype of primary liver cancer, while Gu et al. (2017) included liver and intrahepatic bile duct cancers (20); risk factor associations have varied according to histology (47).
Although there was little evidence of confounding in the analyses, UV was the strongest confounder in the relationship between distance from time zone meridian and HCC in our study. UV is the primary source of vitamin D for most people and has been associated with a decreased risk for HCC (22,23). In this analysis, UV was inversely associated with increasing westward distance from the time zone meridian, indicating that the counties in which HCC cases resided at diagnosis that were located west of the time zone meridian were characterized by relatively lower UV levels compared to counties east of the time zone meridian. Given the large number of HCC cases in our study, we were well-powered to detect an association. The effect size for circadian misalignment observed in this study is modest compared to other important risk factors for HCC such as chronic HCV infection. However, over 40% of HCC cases are diagnosed in individuals with no known risk factors (14). Given biological plausibility for the relationship between circadian misalignment and HCC based on experimental evidence suggesting that chronic circadian disruption is associated with hepatocarcinogenic effects (16,18), these findings provide support for a novel etiologic factor for HCC.
There was a statistically significant interaction between distance from time zone meridian and age on HCC risk. Increasing distance moving east to west within a time zone was associated with an increased risk for HCC among those <65 years old, but was not associated with HCC risk among those ≥65 years old. With increasing age, the proportion of early chronotypes increases, characterized by an early bedtime and early wake time (33,39). Thus, circadian disruption due to residence in a western region of a time zone may differentially impact late chronotypes more than early chronotypes. Further, younger individuals may be more likely to have and comply with school and work obligations associated with early start/local clock times. In addition, we observed statistically significant interactions between distance from time zone meridian and race/ethnicity as well as obesity prevalence on HCC risk. Positive associations were observed across all racial/ethnic categories, although an inverse association was observed among Hispanics. This finding among Hispanics was unexpected and warrants further study. Further, results stratified by obesity prevalence showed generally similar positive associations; the sample size for HCC cases residing in high obesity prevalence areas was relatively smaller than for those living in low obesity prevalence areas.
This study includes several limitations. We did not have information on individual-level measures of circadian misalignment such as chronotype and work schedule. However, we adjusted for county-level prevalence of shift work occupation, which did not change the study results. We also adjusted for urbanicity, which is associated with light pollution, noise, and other factors related to sleep disturbances. The counties included in our study observe Daylight Saving Time, which could be a transient source of annual circadian disruption. We used the county at diagnosis to estimate circadian misalignment exposure, assuming that cases resided in these counties prior to diagnosis. Although we did not have information on geocoded residential history, we used the county Center of Population to approximate the location of each case at diagnosis. Center of Population locations are determined using information on where the majority of residents live within a county. Further, stratification by county-level residential mobility showed similar results. The study results may be subject to the ecological fallacy as we used the county at diagnosis to determine distance from time zone meridian. There may be residual confounding due to lack of adjustment for individual-level risk factors for HCC, including chronic HBV infection, chronic HCV infection, obesity, diabetes, and alcohol consumption. For example, obesity is a risk factor for HCC that has also been associated with circadian disruption (37). Further, percentage foreign born is not a perfect proxy for HBV infection status, as HBV prevalence rates vary across different countries. Information on country of birth is not included in publicly available SEER datasets (48). However, we adjusted for county-level information from objective data sources, such as SEER and IHME data created using BRFSS and NHANES, on factors known or suspected to be associated with HCC, including proxies for HBV (percentage foreign born) and HCV (urbanicity), alcohol consumption, smoking, obesity, and diabetes; no variables substantially changed the effect estimates. We also adjusted for ambient UV exposure using a validated high-resolution spatiotemporal UV model (30). Future research should also consider adjusting for individual-level information regarding prediabetes as well as BMI to account for individuals who are overweight or obese, as incidence of nonalcoholic fatty liver disease has been increasing, which is an important risk factor for HCC in the U.S.
Strengths of our study include an objective exposure assessment estimating distance from time zone meridian using the Centers of Population for counties at diagnosis provided by SEER, which was conducted similarly across all counties in the study area. Importantly, our distance from time zone meridian calculations used an accurate representation of the Earth’s shape as an ellipsoid and accounted for latitude. We were able to examine many counties across the U.S. that are characterized by a wide range of distances from their respective time zone meridians. We examined a large number of confirmed incident primary HCC cases using information from the SEER database, the most common histological type of primary liver cancer across most parts of the world including the U.S. We also evaluated potential confounding and effect modification by known and suspected HCC risk factors, including individual-level information on age, and county-level data on lifestyle and health behaviors, health conditions, shift work occupation, socioeconomic status, and environmental factors.
In conclusion, increasing distance from east to west within a time zone was associated with a statistically significant positive association with HCC risk in the U.S. This association was observed among individuals aged <65 years old, but not among those aged ≥65 years old, suggesting that circadian misalignment associated with western residence in a time zone may differentially affect early vs. late chronotypes. Alternatively, younger individuals may be more likely to have school and/or work obligations that require early start/local clock times. Both circadian misalignment and HCC have been increasing across many regions in the world. Circadian misalignment is a modifiable risk factor. Future research should be conducted to examine this association using individual-level information on circadian misalignment and HCC risk factors.
Supplementary Material
Acknowledgments
T. VoPham was supported by the National Institutes of Health (NIH) National Cancer Institute (NCI) Training Program in Cancer Epidemiology (T32 CA009001). M. Weaver was supported by the NIH National Heart, Lung, and Blood Institute (NHLBI) Training Program in Sleep, Circadian and Respiratory Neurobiology (T32 HL007901) and NHLBI award number F32 HL134249. The authors would like to acknowledge the NCI Surveillance, Epidemiology, and End Results (SEER) program, Institute for Health Metrics and Evaluation (IHME), Environmental Protection Agency (EPA), U.S. Census Bureau, U.S. Geological Survey, Minnesota Population Center, and Centers for Disease Control and Prevention for providing the publicly available data used in this study.
Footnotes
Conflicts of interest: The authors declare no potential conflicts of interest.
References
- 1.Voigt RM, Forsyth CB, Keshavarzian A. Circadian disruption: potential implications in inflammatory and metabolic diseases associated with alcohol. Alcohol Res. 2013;35(1):87. [PMC free article] [PubMed] [Google Scholar]
- 2.Berson DM, Dunn FA, Takao M. Phototransduction by retinal ganglion cells that set the circadian clock. Science. 2002;295(5557):1070–3. doi: 10.1126/science.1067262. [DOI] [PubMed] [Google Scholar]
- 3.Czeisler CA, Allan JS, Strogatz SH, Ronda JM, Sanchez R, Rios CD, et al. Bright light resets the human circadian pacemaker independent of the timing of the sleep-wake cycle. Science. 1986;233:667–72. doi: 10.1126/science.3726555. [DOI] [PubMed] [Google Scholar]
- 4.Yang WS, Deng Q, Fan WY, Wang WY, Wang X. Light exposure at night, sleep duration, melatonin, and breast cancer: a dose-response analysis of observational studies. Eur J Cancer Prev. 2014;23(4):269–76. doi: 10.1097/CEJ.0000000000000030. [DOI] [PubMed] [Google Scholar]
- 5.Savvidis C, Koutsilieris M. Circadian rhythm disruption in cancer biology. Mol Med. 2012;18(1):1249. doi: 10.2119/molmed.2012.00077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Reid KJ, Zee PC. Circadian disorders of the sleep-wake cycle. Philadelphia, PA: WB Saunders; 2005. pp. 691–701. [Google Scholar]
- 7.Evans JA, Davidson AJ. Health consequences of circadian disruption in humans and animal models. Prog Mol Biol Transl Sci. 2013;119:283–323. doi: 10.1016/B978-0-12-396971-2.00010-5. [DOI] [PubMed] [Google Scholar]
- 8.Roenneberg T, Merrow M. The circadian clock and human health. Curr Biol. 2016;26(10):R432–R43. doi: 10.1016/j.cub.2016.04.011. [DOI] [PubMed] [Google Scholar]
- 9.Potter GD, Skene DJ, Arendt J, Cade JE, Grant PJ, Hardie LJ. Circadian rhythm and sleep disruption: causes, metabolic consequences, and countermeasures. Endocr Rev. 2016;37(6):584–608. doi: 10.1210/er.2016-1083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wittmann M, Dinich J, Merrow M, Roenneberg T. Social jetlag: misalignment of biological and social time. Chronobiol Int. 2006;23(1–2):497–509. doi: 10.1080/07420520500545979. [DOI] [PubMed] [Google Scholar]
- 11.Roenneberg T, Kumar CJ, Merrow M. The human circadian clock entrains to sun time. Curr Biol. 2007;17(2):R44–R5. doi: 10.1016/j.cub.2006.12.011. [DOI] [PubMed] [Google Scholar]
- 12.Borisenkov MF. Latitude of residence and position in time zone are predictors of cancer incidence, cancer mortality, and life expectancy at birth. Chronobiol Int. 2011;28(2):155–62. doi: 10.3109/07420528.2010.541312. [DOI] [PubMed] [Google Scholar]
- 13.Wong MC, Jiang JY, Goggins WB, Liang M, Fang Y, Fung FD, et al. International incidence and mortality trends of liver cancer: a global profile. Sci Rep. 2017;7 doi: 10.1038/srep45846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Makarova-Rusher OV, Altekruse SF, McNeel TS, Ulahannan S, Duffy AG, Graubard BI, et al. Population attributable fractions of risk factors for hepatocellular carcinoma in the United States. Cancer. 2016;122(11):1757–65. doi: 10.1002/cncr.29971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ferrell JM, Chiang JY. Circadian rhythms in liver metabolism and disease. Acta Pharm Sin B. 2015;5(2):113–22. doi: 10.1016/j.apsb.2015.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kettner NM, Voicu H, Finegold MJ, Coarfa C, Sreekumar A, Putluri N, et al. Circadian homeostasis of liver metabolism suppresses hepatocarcinogenesis. Cancer Cell. 2016;30(6):909–24. doi: 10.1016/j.ccell.2016.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang X, Fu X, Van Ness C, Meng Z, Ma X, Huang W. Bile acid receptors and liver cancer. Curr Pathobiol Rep. 2013;1(1):29–35. doi: 10.1007/s40139-012-0003-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Filipski E, Subramanian P, Carriere J, Guettier C, Barbason H, Levi F. Circadian disruption accelerates liver carcinogenesis in mice. Mutat Res Genet Toxicol Environ Mutagen. 2009;680(1):95–105. doi: 10.1016/j.mrgentox.2009.10.002. [DOI] [PubMed] [Google Scholar]
- 19.Shi S-Q, Ansari TS, McGuinness OP, Wasserman DH, Johnson CH. Circadian disruption leads to insulin resistance and obesity. Curr Biol. 2013;23(5):372–81. doi: 10.1016/j.cub.2013.01.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gu F, Xu S, Devesa SS, Zhang F, Klerman EB, Graubard BI, et al. Longitude Position in a Timezone and Cancer Risk in the United States. Cancer Epidemiol Biomarkers Prev. 2017;26(8):1306–11. doi: 10.1158/1055-9965.EPI-16-1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Surveillance, Epidemiology, and End Results (SEER) Program SEER*Stat Database: Incidence - SEER 18 Regs Research Data + Hurricane Katrina Impacted Louisiana Cases, Nov 2016 Sub (1973–2014 varying) - Linked To County Attributes - Total US, 1969–2015 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, Surveillance Systems Branch. released April 2017 based on the November 2016 submission. [Google Scholar]
- 22.VoPham T, Bertrand KA, Yuan JM, Tamimi RM, Hart JE, Laden F. Ambient ultraviolet radiation exposure and hepatocellular carcinoma incidence in the United States. Environ Health. 2017;16(1):89. doi: 10.1186/s12940-017-0299-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fedirko V, Duarte‐Salles T, Bamia C, Trichopoulou A, Aleksandrova K, Trichopoulos D, et al. Prediagnostic circulating vitamin D levels and risk of hepatocellular carcinoma in European populations: A nested case‐control study. Hepatology. 2014;60(4):1222–30. doi: 10.1002/hep.27079. [DOI] [PubMed] [Google Scholar]
- 24.World Health Organization. International Classification of Diseases for Oncology. Geneva, Switzerland: World Health Organization; 2013. First revision. [Google Scholar]
- 25.VoPham T, Brooks MM, Yuan JM, Talbott EO, Ruddell D, Hart JE, et al. Pesticide exposure and hepatocellular carcinoma risk: A case-control study using a geographic information system (GIS) to link SEER-Medicare and California pesticide data. Environ Res. 2015;143(Pt A):68–82. doi: 10.1016/j.envres.2015.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cahoon EK, Pfeiffer RM, Wheeler DC, Arhancet J, Lin SW, Alexander BH, et al. Relationship between ambient ultraviolet radiation and non‐Hodgkin lymphoma subtypes: A US population‐based study of racial and ethnic groups. Int J Cancer. 2015;136(5):E432–E41. doi: 10.1002/ijc.29237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thomas C, Featherstone W. Validation of Vincenty’s formulas for the geodesic using a new fourth-order extension of Kivioja’s formula. J Surv Eng. 2005;131(1):20–6. [Google Scholar]
- 28.Carr BI. Hepatocellular Carcinoma: Diagnosis and Treatment. Cham, Switzerland: Springer International Publishing; 2016. [Google Scholar]
- 29.International Agency for Research on Cancer. Outdoor Air Pollution. Geneva, Switzerland: WHO Press; 2016. [Google Scholar]
- 30.VoPham T, Hart JE, Bertrand KA, Sun Z, Tamimi RM, Laden F. Spatiotemporal exposure modeling of ambient erythemal ultraviolet radiation. Environ Health. 2016;15(1):111. doi: 10.1186/s12940-016-0197-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pan W-C, Wu C-D, Chen M-J, Huang Y-T, Chen C-J, Su H-J, et al. Fine particle pollution, alanine transaminase, and liver cancer: A Taiwanese prospective cohort study (REVEAL-HBV) J Natl Cancer Inst. 2016;108(3):djv341. doi: 10.1093/jnci/djv341. [DOI] [PubMed] [Google Scholar]
- 32.James P, Bertrand KA, Hart JE, Schernhammer ES, Tamimi RM, Laden F. Outdoor Light at Night and Breast Cancer Incidence in the Nurses’ Health Study II. Environ Health Perspect. 2017;125(8):087010. doi: 10.1289/EHP935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fischer D, Lombardi DA, Marucci-Wellman H, Roenneberg T. Chronotypes in the US–Influence of age and sex. PLOS ONE. 2017;12(6):e0178782. doi: 10.1371/journal.pone.0178782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Qian J, Scheer FA. Circadian System and Glucose Metabolism: Implications for Physiology and Disease. Trends in endocrinology and metabolism: TEM. 2016;27(5):282–93. doi: 10.1016/j.tem.2016.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Smolensky MH, Hermida RC, Reinberg A, Sackett-Lundeen L, Portaluppi F. Circadian disruption: New clinical perspective of disease pathology and basis for chronotherapeutic intervention. Chronobiol Int. 2016;33(8):1101–19. doi: 10.1080/07420528.2016.1184678. [DOI] [PubMed] [Google Scholar]
- 36.Ryerson AB, Eheman CR, Altekruse SF, Ward JW, Jemal A, Sherman RL, et al. Annual Report to the Nation on the Status of Cancer, 1975‐2012, featuring the increasing incidence of liver cancer. Cancer. 2016;122(9):1312–37. doi: 10.1002/cncr.29936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Roenneberg T, Allebrandt KV, Merrow M, Vetter C. Social jetlag and obesity. Curr Biol. 2012;22(10):939–43. doi: 10.1016/j.cub.2012.03.038. [DOI] [PubMed] [Google Scholar]
- 38.Roenneberg T, Kantermann T, Juda M, Vetter C, Allebrandt KV. Light and the human circadian clock Circadian Clocks. First. Berlin, Germany: Springer; 2013. pp. 311–31. [DOI] [PubMed] [Google Scholar]
- 39.Roenneberg T, Kuehnle T, Juda M, Kantermann T, Allebrandt K, Gordijn M, et al. Epidemiology of the human circadian clock. Sleep Med Rev. 2007;11(6):429–38. doi: 10.1016/j.smrv.2007.07.005. [DOI] [PubMed] [Google Scholar]
- 40.IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Painting, firefighting, and shiftwork. IARC monographs on the evaluation of carcinogenic risks to humans. 2010;98:9. [PMC free article] [PubMed] [Google Scholar]
- 41.Irish LA, Kline CE, Gunn HE, Buysse DJ, Hall MH. The role of sleep hygiene in promoting public health: A review of empirical evidence. Sleep Med Rev. 2015;22:23–36. doi: 10.1016/j.smrv.2014.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chang AM, Aeschbach D, Duffy JF, Czeisler CA. Evening use of light-emitting eReaders negatively affects sleep, circadian timing, and next-morning alertness. Proceedings of the National Academy of Sciences of the United States of America. 2015;112(4):1232–7. doi: 10.1073/pnas.1418490112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Vetter C, Fischer D, Matera JL, Roenneberg T. Aligning work and circadian time in shift workers improves sleep and reduces circadian disruption. Curr Biol. 2015;25(7):907–11. doi: 10.1016/j.cub.2015.01.064. [DOI] [PubMed] [Google Scholar]
- 44.Owens JA, Belon K, Moss P. Impact of delaying school start time on adolescent sleep, mood, and behavior. Arch Pediatr Adolesc Med. 2010;164(7):608–14. doi: 10.1001/archpediatrics.2010.96. [DOI] [PubMed] [Google Scholar]
- 45.Asher G, Sassone-Corsi P. Time for food: the intimate interplay between nutrition, metabolism, and the circadian clock. Cell. 2015;161(1):84–92. doi: 10.1016/j.cell.2015.03.015. [DOI] [PubMed] [Google Scholar]
- 46.Kauhl B, Heil J, Hoebe CJ, Schweikart J, Krafft T, Dukers-Muijrers NH. The spatial distribution of hepatitis C virus infections and associated determinants—An application of a geographically weighted poisson regression for evidence-based screening interventions in hotspots. PLOS ONE. 2015;10(9):e0135656. doi: 10.1371/journal.pone.0135656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Tyson GL, El-Serag HB. Risk factors for cholangiocarcinoma. Hepatology. 2011;54(1):173–84. doi: 10.1002/hep.24351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yang JD, Altekruse SF, Nguyen MH, Gores GJ, Roberts LR. Impact of country of birth on age at the time of diagnosis of hepatocellular carcinoma in the United States. Cancer. 2017;123(1):81–9. doi: 10.1002/cncr.30246. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


