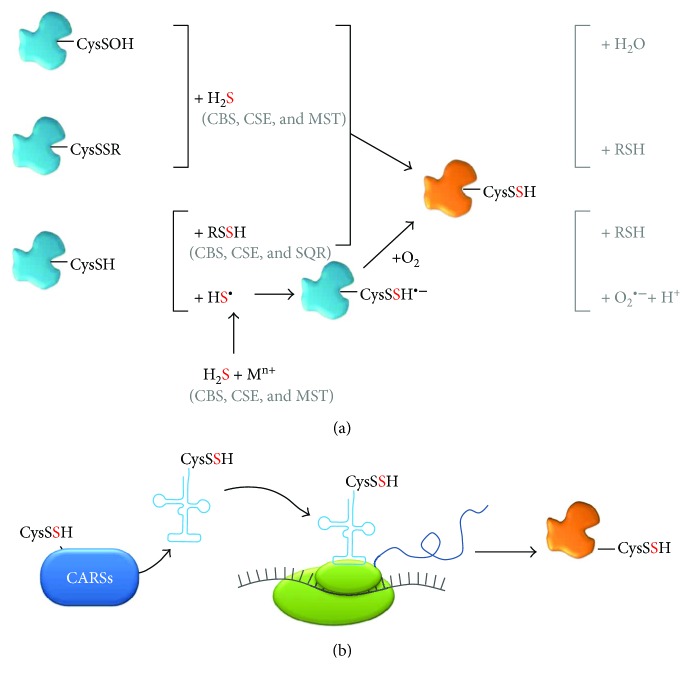
Figure 11.
Reactions leading to protein persulfidation. Scheme depicting the most plausible in vivo reactions leading to cysteine persulfidation in target proteins, through (a) posttranslational modification of cysteine residues or (b) cotranslational incorporation of CysSSH via CysSSH-bound tRNA derived from cysteinyl-tRNA synthetases (CARSs). (a) Proteins with oxidized cysteine residues (CysSSR and CysSOH) can react directly with H2S (derived from its synthesizing enzymes CBS, CSE, and MST) to yield a protein cysteine persulfide (CysSSH). Proteins with reduced cysteine residues (CysSH) can react with free LMW persulfides (RSSH, such as glutathione persulfide and cysteine persulfide, products of H2S biosynthetic—CBS, CSE, and MST—or catabolic—SQR—enzymes) or with the HS· radical formed by reaction of H2S with metal centers (Mn+). The resulting protein-bound cysteine perthiyl radical (CysSSH·−) can then react with oxygen to yield CysSSH and superoxide anion. (b) Generation of protein cysteine persulfides through cotranslational incorporation of CysSSH. Mammalian cysteinyl-tRNA synthetases (CARSs, dark blue box, which also synthesize free CysSSH from cysteine) catalyze the synthesis of tRNA-bound cysteine persulfides (light blue line), which incorporate CysSSH into the nascent polypeptide (dark blue line) at cysteine sites upon translation (ribosome depicted in green and mRNA in grey).