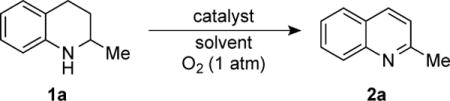
Table 1.
Low Temperature Dehydrogenation of 1,2,3,4-Tetrahydroquinaldine
| ||||
|---|---|---|---|---|
|
| ||||
| entry | catalyst (mol %) | solvent (concn [M]) | temp | yield (conv)b |
| 1 | Pd(TFA)2 (5)/2-NMe2-py (10)/pTsOH (20) | DMSO (1) | 80 | 52 (88) |
| 2 | Pd(TFA)2 (5)/DMSO (10) | AcOH (0.2) | 80 | 55 (100) |
| 3 | Pd(TFA)2 (5)/4,5-diazafluoren-9-one (5) | DMSO (0.2) | 80 | 49 (61) |
| 4a,c | CoOx-phen/AB | MeOH (0.125) | 60 | 92 (94) |
| 5a | CoOx-phen/AB | MeOH (0.125) | 60 | 43 (43) |
| 6a,c | CoOx-bpy/AB | MeOH (0.125) | 60 | 0 (1) |
| 7a,c | CoOx-terpy/AB | MeOH (0.125) | 60 | 82 (100) |
| 8a,c | CoOx-phd/AB | MeOH (0.125) | 60 | 67 (74) |
| 9a,c | MnOx-phen/AB | MeOH (0.125) | 60 | 16 (32) |
| 10a,c | FeOx-phen/AB | MeOH (0.125) | 60 | 0 (52) |
| 11a,c | NiOx-phen/AB | MeOH (0.125) | 60 | 0 (13) |
| 12a,c | CoOx-phen/Vulcan XC72R | MeOH (0.125) | 60 | 92 (100) |
| 13a | Pd/C | Xylene (0.2) | 60 | 0 (0) |
| 14a | Pd/C | MeOH (0.125) | 60 | 13 (30) |
| 15a,d,e | Activated C | Xylene (0.2) | 60 | 29 (66) |
| 16a,d,f | Co(salen) | EtOH (0.066) | 60 | 28 (63) |
|
| ||||
1a (0.10 mmol), solvent, 2.5 mol % [cat.], 1 atm O2, orbital mixing, 60 °C, 4 h.
% Yields and conv determined by 1H NMR.
1 equiv K2CO3 added.
Magnetic stirring was employed.
100 wt % catalyst, 24 h.
1 mol % catalyst was used, 24 h. C = carbon; AB = Acetylene Black.
