Abstract
Gross primary production (GPP) is a fundamental ecosystem process that sequesters carbon dioxide (CO2) and forms the resource base for higher trophic levels. Still, the relative contribution of different controls on GPP at the whole-ecosystem scale is far from resolved. Here we show, by manipulating CO2 concentrations in large-scale experimental pond ecosystems, that CO2 availability is a key driver of whole-ecosystem GPP. This result suggests we need to reformulate past conceptual models describing controls of lake ecosystem productivity and include our findings when developing models used to predict future lake ecosystem responses to environmental change.
Subject terms: Ecology, Biogeochemistry
Introduction
The rate at which primary producers fix inorganic carbon controls the supply of organic matter to food webs and influences the biogeochemistry of aquatic ecosystems1,2. The role of inorganic nutrients and light in controlling GPP is indisputable and has been extensively studied3–5.
Data also suggest that CO2 availability can constrain GPP. Small scale experimental studies, and comparative studies of lakes, have shown that elevated CO2 concentration promotes phytoplankton biomass and GPP6,7. Similarly, CO2 concentration can limit phytoplankton growth in marine ecosystems8. However, aquatic ecosystems are comprised of both pelagic and benthic habitats, and recent studies emphasize the importance to include both habitats to understand whole-ecosystem productivity and function3,4. Presently, the role of CO2 availability for whole-ecosystem GPP, especially in shallow lakes where both pelagic and benthic GPP may contribute significantly to whole-ecosystem GPP, has never been tested9. Improved knowledge of the control of GPP is fundamental for understanding ecosystem function and impacts of environmental change.
Here we carried out a large-scale experiment to test the role of CO2 availability for whole-ecosystem GPP. We used a novel approach where part of the CO2 that accumulated under ice cover over winter was released to the atmosphere by manipulation of the spring ice cover. The aim was to decrease CO2 concentrations in the water column while having a limited effect on light, temperature, and other key environmental factors. In spring, the ice cover was decreased twice, by first removing 10% and then 50%, to impose a gradual but drastic difference in CO2 concentrations in the treatment ponds. Whole-ecosystem GPP was estimated using dissolved oxygen time series data, and potential abiotic and biotic drivers of GPP were monitored over the course of the experiment.
Results and Discussion
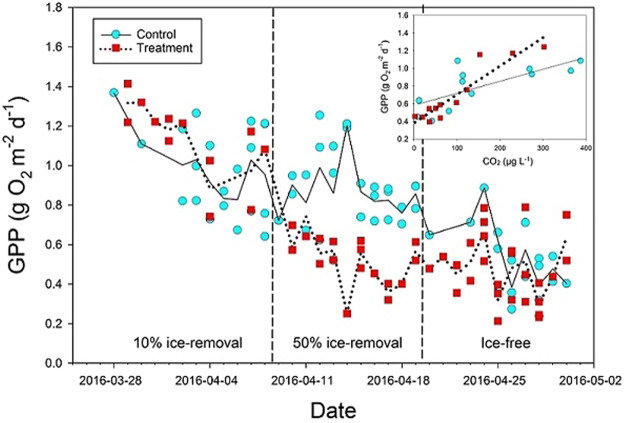
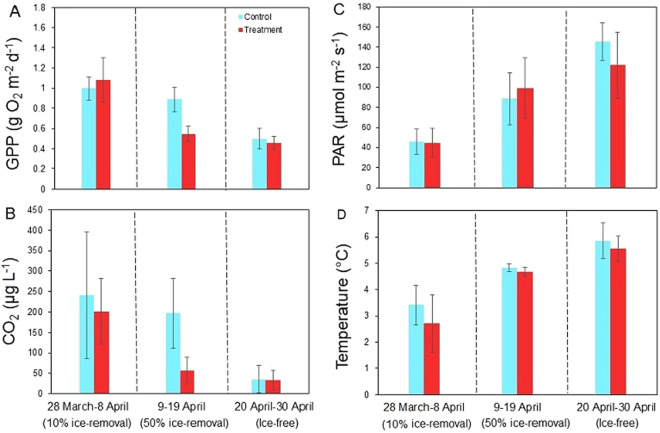
The GPP and CO2 concentration in control ponds were relatively high under ice compared to after ice break-up in spring (Figs 1, 2). The 10% ice-removal treatment did not change GPP or decrease CO2 concentration compared to control ponds (Fig. 2 and Table 1). In contrast, the 50% ice-removal treatment decreased both GPP and CO2 compared to control ponds (Fig. 2 and Table 1). Finally, after ice break-up, there were no differences in GPP and CO2 between control and treatment ponds (Figs 1, 2 and Table 1). There was a positive correlation between GPP and CO2 concentration in both control and treatment ponds (Fig. 1 and Table 1). These results show that the CO2 concentration was a key controlling factor for GPP in the ponds.
Figure 1.
Daily gross primary production (GPP) and correlations between GPP and carbon dioxide (CO2) concentrations (inserted) in control (full line) and treatment (dashed line) ponds. The trend lines are moving averages of daily GPP.
Figure 2.
Average (±1 SD) gross primary production (GPP) (A), carbon dioxide (CO2) (B), photosynthetically active radiation (PAR) (C) and temperature (D) for the different periods during the experiment.
Table 1.
Statistical Analysis results.
| Test | Ponds | Treatment | Variables | Statistics | p-values | n | df |
|---|---|---|---|---|---|---|---|
| T-test | t-value | ||||||
| C vs. T | 10% | GPP | −0.77 | 0.49 | 4 | 3 | |
| C vs. T | 10% | CO2 | 0.49 | 0.56 | 4 | 3 | |
| C vs. T | 10% | PAR | 0.23 | 0.82 | 4 | 3 | |
| C vs. T | 10% | Temp. | 2.6 | 0.07 | 4 | 3 | |
| C vs. T | 50% | GPP | 12.21 | <0.01 | 4 | 3 | |
| C vs. T | 50% | CO2 | 3.64 | <0.05 | 4 | 3 | |
| C vs. T | 50% | PAR | −1.30 | 0.28 | 4 | 3 | |
| C vs. T | 50% | Temp. | 0.97 | 0.40 | 4 | 3 | |
| C vs. T | Ice-free | GPP | 1.18 | 0.32 | 4 | 3 | |
| C vs. T | Ice-free | CO2 | 0.13 | 0.90 | 4 | 3 | |
| C vs. T | Ice-free | PAR | 0.92 | 0.42 | 4 | 3 | |
| C vs. T | Ice-free | Temp. | 0.80 | 0.47 | 4 | 3 | |
| Pearson’s r | r-value | ||||||
| C | GPP vs. CO2 | 0.73 | <0.01 | ||||
| T | GPP vs. CO2 | 0.93 | <0.001 | ||||
| C | GPP vs. PAR | −0.86 | <0.001 | ||||
| T | GPP vs. PAR | −0.69 | <0.05 | ||||
| C | GPP vs. Temp. | −0.80 | <0.01 | ||||
| T | GPP vs. Temp. | −0.73 | <0.01 | ||||
| ANOVA | F-value | ||||||
| C vs. T | GPP * time | 6.92 | <0.05 | 2, 12 | |||
| C vs. T | CO2 * time | 3.95 | <0.05 | 2, 12 | |||
| C vs. T | PAR * time | 2.16 | 0.15 | 2, 12 | |||
| C vs. T | Temp.* time | 0.3 | 0.74 | 2, 12 | |||
Control ponds, treatment ponds, number of replicates, degrees of freedom, 10% ice-cover removing treatment, 50% ice-cover removing treatment, Ice-free period, and linear correlation are abbreviated as C, T, n, df, 10%, 50%, Ice-free and r, respectively.
No other abiotic or biotic factors explain the patterns in GPP over time and between treatments. There were no differences in nutrient concentrations (NO3−, PO43− and NH4+, Supplementary Table S1), photosynthetically active radiation (PAR) or water temperature between control and treatment ponds (Fig. 2 and Table 1). Interestingly, GPP in control and treatment ponds was negatively correlated to both PAR and water temperature (Table 1). The consumer biomass (zooplankton and zoobenthos) did not differ between control and treatment ponds (Supplementary Table S1), suggesting that top-down effects on primary producers, if any, were similar and therefore should have no effect on the main patterns in GPP observed between treatment and control. As expected, pH of the water increased with decreasing CO2 concentrations in the ponds (Supplementary Table S1). The change in pH and carbonate system is part of the change associated with changes in CO2 saturation and cannot be easily separated from each other. Yet, previous studies on the effect of CO2 on GPP have not found any effect of pH on GPP and attributed GPP responses to CO2 availability and not to pH per se6,7,10.
This is the first experimental study on the role of CO2 in controlling whole-ecosystem GPP. Although CO2 has a key role as substrate for photosynthetic enzymes11, the CO2 supply is generally regarded as sufficient to meet the primary producer’s demands and that other factors are limiting photosynthetic rates. However, CO2 may often be at suboptimal levels for maximum photosynthetic efficiency7. Previous work has shown positive effects of CO2 on pelagic GPP6,7, but here we show that this effect applies also at the whole-ecosystem scale. This is important since lake habitats are not isolated units and both benthic and pelagic habitats are important for understanding whole-lake ecosystem food web dynamics and productivity3,4.
CO2 limitation effect on lake productivity is likely a general characteristic that needs to be taken into account in food web models for lakes. The CO2 concentrations of lakes vary largely across systems but also within systems, both spatially and temporally12, implying that CO2 availability can help to explain GPP at multiple scales. Given that lake CO2 supersaturation is common, including in abundant northern lakes13, we suggest that CO2 stimulation of GPP is a common but often overlooked phenomenon.
Although not explicitly studied in this experiment, these results also add important insight to the role of terrestrial organic matter for lake GPP. Export of dissolved organic matter from land is regarded to have two main effects on GPP in recipient lake ecosystems: (i) stimulating GPP by supplying nutrients14 and (ii) repressing GPP by supplying light absorbing substances4. Terrestrial organic matter is also mineralized in lakes, resulting in supersaturation of CO215,16, and our results suggest this will stimulate GPP. It is likely that CO2 stimulates GPP in ecosystems with low to moderate terrestrial organic matter inputs, up to a point when light becomes suboptimal, after which further increase in terrestrial organic matter inputs will have an overall negative effect on GPP due to light limitation. Future research should assess the role of CO2 for GPP across various type of lakes.
The results from this study provide fundamental knowledge of the effects of CO2 dynamics on biomass production in lake food webs. More broadly, these results have major implications for a greater understanding of the effects of climate change on lake productivity, as CO2 dynamics in lakes are controlled by climate-dependent factors such as terrestrial carbon export, hydrologic residence time, metabolic process rates, mixing regimes and ice conditions12,17,18. We stress the need for future research efforts, where the effects of CO2 are incorporated in both experiments and models of lake ecosystem function to generate patterns at the whole-ecosystem scale.
Methods
Study site
The study was performed in the Umeå University Experimental Ecosystem Facility (EXEF) in northern Sweden (63°48′N, 20°14′E). The pond is divided into 20 enclosures (12.5 * 7.3 m, average depth 1.48 m) by thick non-permeable tarpaulins and each pond has a 7.3 m long natural shoreline and a bottom substrate of soft sediment. Each pond has a natural food web including basal producers (algae and bacteria), consumers (insect larvae and zooplankton) and a reproducing top-consumer population (nine-spine Sticklebacks), where benthic GPP constitutes approximately 50% of whole-ecosystem GPP19. For further details see19,20. In this study we used eight enclosures: we manipulated the CO2 concentration in four enclosures by ice cover removal (treatment), and four enclosures served as controls. Ice formed naturally on the ponds in mid-November 2015, and the experiment was carried out in March-April 2016. The ice cover removal was preformed twice in 2016 on the treatment ponds; 10% removal from 28th March to 8th April and 50% from 9th to 19th April, i.e. ten days for each treatment. The four control ponds experienced a natural and gradual ice melting. The treatment and control ponds became ice-free between 20th of April and 1st of May. The ice-removal treatment was executed by making vertical bores in the ice cover with an ice drill. Pieces of ice were cut out in-between the bores by a large-ice saw and then pushed in under the remaining ice.
Data collection
CO2 concentrations were estimated every second day by using a headspace equilibration technique21 and analysis by gas chromatograph (Perkin Elmer Clarus 500). At the same time, nutrients (NO3−, PO43−, and NH4+) were sampled by filtering water through burnt (550 °C, 4 h) 0.45 μm GF/F filters to 50 ml Falcon tubes and stored in the freezer until analyzed with photometric flow injection analysis (FIA) methods22.
Dissolved oxygen (DO) and water temperature were measured during the experiment period at ten-minute intervals by logging sensors (MiniDOT, PME, Vista, CA, USA) which were deployed at 0.5 m below the water surface in the center of each pond. Photosynthetic available radiation (PAR) was measured at ten-minute intervals by light sensors (SQ-110, Apogee USA) deployed at 0.8 m below the water surface in the center of each pond. Temperature and PAR data were converted to daily means for each enclosure based on 144 measures (6 per hour × 24 hours) and then a mean for all days within each treatment period during the experiment and finally we got a mean for each group (control and treatment) that contains four enclosures. Wind speed was recorded every ten minutes by a climate station next to the pond.
Whole-ecosystem gross primary production (GPP) estimates
From the oxygen sensor data whole-ecosystem GPP, integrating GPP in both benthic and pelagic habitats in these non-stratified ponds, was calculated with inverse modeling and Bayesian parameter estimation using a similar parameter estimation approach as for diel DO in streams23, but modified for pond ecosystems “equation (1)”:
| 1 |
where mOi is modeled DO at time i (g O2 m−3) given parameter estimates of GPP and ecosystem respiration (ER; g O2 m−2 d−1). Because changes in O2 are a function of GPP, ER, and gas exchange, we calculated daily air-water O2 fluxes based on O2 saturation and the temperature-corrected gas exchange velocity for O2 (Ki, d−1). Ki was estimated from K600 derived from wind speed24. The emission flux of O2 () was corrected for changes in pond area open to the atmosphere (0–100% with changing ice cover) and zmix (mean of mixing depth; m), where zmix varied daily according to changes in water column depth depending on ice cover thickness. The metabolism model used a “random walk” metropolis algorithm and Markov Chain Monte Carlo (MCMC) sampling from the “metrop” function in the “mcmc” package of the statistical program R25 to find the best fit between measured and modelled O2 data given model estimates of GPP and ER. Each parameter estimate was derived from 10000 model iterations after removing an initial 1000 iterations of “burn-in” from parameter starting values. We checked for convergence of parameter estimates and removed days with negative GPP and with poor fits between measured and modeled O2 before assessing the response of GPP to changes in CO2.
Invertebrate sampling
Zooplankton were sampled by a zooplankton net (diameter 20 cm, 100 µm mesh size) drawn vertically trough whole water column and preserved in Lugol’s solution. Zoobenthos were sampled with a net (30 cm wide, 1 mm mesh size), drawn at the bottom substrate for a distance of 1 m, and preserved in ethanol. Zooplankton and zoobenthos lengths were measured to obtain dry biomass using length-weight regressions26,27.
Statistical analyses
Statistics (SPSS 20 and R v3.2.3) are based on individual pond means of measured response variables. Results were tested for time effects between treatments periods by using repeated measures ANOVA, for differences between the control and treatment within treatment periods by using standard t-tests, and for correlations between selected variables by using Pearson correlation coefficient.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on request.
Electronic supplementary material
Acknowledgements
Thanks to Anders Jonsson and Erik Geibrink for their help in the laboratory and in the field. This study was financially supported by the Ministry of Higher Education and Scientific Research in Iraq (18603-2014-12-06) and by the Swedish Research Council (621-2011-3908). This study is contribution no. 8 from Umeå University Experimental Ecosystem Facility (EXEF).
Author Contributions
M.H., J.K. and P.B. designed the study with contribution from J.A. M.H. and M.J.A. performed the field and lab work. E.H. wrote the metabolism codes. M.H. analyzed the data with contribution from E.H. and P.B. M.H. wrote the manuscript and all co-authors revised the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
6/12/2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-29166-3.
References
- 1.Persson L, Diehl S, Johansson L, Andersson G, Hamrin SF. Trophic interactions in temperate lake ecosystems: a test of food chain theory. Am. Nat. 1992;140:59–84. doi: 10.1086/285403. [DOI] [Google Scholar]
- 2.Falkowski PG, Fenche T, Delong EF. The microbial engines that drive earth’s biogeochemical cycles. Science. 2008;320:1034–1039. doi: 10.1126/science.1153213. [DOI] [PubMed] [Google Scholar]
- 3.Vadeboncoeur Y, Lodge DM, Carpenter SR. Whole-lake fertilization effects on distribution of primary production between benthic and pelagic habitats. Ecology. 2001;82:1065–1077. doi: 10.1890/0012-9658(2001)082[1065:WLFEOD]2.0.CO;2. [DOI] [Google Scholar]
- 4.Karlsson J, et al. Light limitation of nutrient-poor lake ecosystems. Nature. 2009;460:506–509. doi: 10.1038/nature08179. [DOI] [PubMed] [Google Scholar]
- 5.Elser JJ, et al. Shifts in lake N: P stoichiometry and nutrient limitation driven by atmospheric nitrogen deposition. Science. 2009;326:835–837. doi: 10.1126/science.1176199. [DOI] [PubMed] [Google Scholar]
- 6.Jansson M, Karlsson J, Jonsson A. Carbon dioxide supersaturation promotes primary production in lakes. Ecol. Lett. 2012;15:527–532. doi: 10.1111/j.1461-0248.2012.01762.x. [DOI] [PubMed] [Google Scholar]
- 7.Vogt RJ, St-Gelais NF, Bogard MJ, Beisner BE, del Giorgio PA. Surface water CO2 concentration influences phytoplankton production but not community composition across boreal lakes. Ecol. Lett. 2017;20:1395–1404. doi: 10.1111/ele.12835. [DOI] [PubMed] [Google Scholar]
- 8.Riebesell U, Wolf-Gladrow DA, Smetacek V. Carbon dioxide limitation of marine phytoplankton growth rates. Nature. 1993;361:249–251. doi: 10.1038/361249a0. [DOI] [Google Scholar]
- 9.Hasler CT, Butman D, Jeffrey JD, Suski CD. Freshwater biota and rising pCO2? Ecol. Lett. 2016;19:98–108. doi: 10.1111/ele.12549. [DOI] [PubMed] [Google Scholar]
- 10.Shapiro J. The role of carbon dioxide in the initiation and maintenance of blue-green dominance in lakes. Freshwat. Biol. 1997;37:307–323. doi: 10.1046/j.1365-2427.1997.00164.x. [DOI] [Google Scholar]
- 11.Badger MR, et al. The diversity and coevolution of rubisco, plastids, pyrenoids, and chloroplast-based CO2-concentrating mechanisms in algae. Can. J. Bot. 1998;76:1052–1071. [Google Scholar]
- 12.Cole JJ, Caraco NF, Kling GW, Kratz TK. Carbon dioxide supersaturation in the surface waters of lakes. Science. 1994;265:1568–1570. doi: 10.1126/science.265.5178.1568. [DOI] [PubMed] [Google Scholar]
- 13.Raymond PA, et al. Global carbon dioxide emissions from inland waters. Nature. 2013;50:355–359. doi: 10.1038/nature12760. [DOI] [PubMed] [Google Scholar]
- 14.Hessen DO, Håll JP, Thrane JE, Andersen T. Coupling dissolved organic carbon, CO2 and productivity in boreal lakes. Freshw. Biol. 2017;62:945–953. doi: 10.1111/fwb.12914. [DOI] [Google Scholar]
- 15.del Giorgio PA, Peters RH. Patterns in planktonic P:R ratios in lakes: Influence of lake trophy and dissolved organic carbon. Limnol. Oceanogr. 1994;39:772–787. doi: 10.4319/lo.1994.39.4.0772. [DOI] [Google Scholar]
- 16.Karlsson J, Jansson M, Jonsson A. Respiration of allochthonous organic carbon in unproductive forest lakes determined by the Keeling plot method. Limnol. Oceanogr. 2007;52:603–608. doi: 10.4319/lo.2007.52.2.0603. [DOI] [Google Scholar]
- 17.Sobek S, Algesten G, Bergström A-K, Jansson M, Tranvik LJ. The catchment and climate regulation of pCO2 in boreal lakes. Glob. Chang. Biol. 2003;9:630–641. doi: 10.1046/j.1365-2486.2003.00619.x. [DOI] [Google Scholar]
- 18.Denfeld BA, Baulch HM, del Giorgio PA, Hampton SE, Karlsson J. A synthesis of carbon dioxide and methane dynamics during the ice-covered period of northern lakes. Limnol. Oceanogr. Lett. 2018;3:117–131. doi: 10.1002/lol2.10079. [DOI] [Google Scholar]
- 19.Rodríguez P, et al. Do warming and humic river runoff alter the metabolic balance of lake ecosystems? Aquat. Sci. 2016;78:717–725. doi: 10.1007/s00027-015-0463-y. [DOI] [Google Scholar]
- 20.Jonsson M, et al. Climate change modifies the size structure of assemblages of emerging aquatic insects. Freshw. Biol. 2015;60:78–88. doi: 10.1111/fwb.12468. [DOI] [Google Scholar]
- 21.Lundin EJ, Giesler R, Persson A, Thompson MS, Karlsson J. Integrating carbon emissions from lakes and streams in a subarctic catchment. Geophys. Res. Biogeosci. 2013;118:1200–1207. doi: 10.1002/jgrg.20092. [DOI] [Google Scholar]
- 22.Gray S, et al. Flow analysis techniques for spatial and temporal measurement of nutrients in aquatic systems. Environ. Chem. 2006;3:3–18. doi: 10.1071/EN05059. [DOI] [Google Scholar]
- 23.Hotchkiss ER, Hall ROJ. High rates of daytime respiration in three streams: Use of δ18O O2 and O2 to model diel ecosystem metabolism. Limnol. Oceanogr. 2014;59:798–810. doi: 10.4319/lo.2014.59.3.0798. [DOI] [Google Scholar]
- 24.Vachon D, Prairie YT. The ecosystem size and shape dependence of gas transfer velocity versus wind speed relationships in lakes. Can. J. Fish. Aquat. Sci. 2013;70:1757–1764. doi: 10.1139/cjfas-2013-0241. [DOI] [Google Scholar]
- 25.Geyer, C. J. & Johnson, L. T. mcmc: Markov Chain Monte Carlo. R package version 0.9-2. Available from http://cran.r-project.org/package=mcmc (2013).
- 26.Botrell HH, et al. A review of some problems in zooplankton production studies. Norwegian J. Zool. 1976;24:419–456. [Google Scholar]
- 27.Persson L, Andersson J, Wahlstrom E, Eklov P. Size-specific interactions in lake systems: predator gape limitation and prey growth rate and mortality. Ecology. 1996;77:900–911. doi: 10.2307/2265510. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on request.