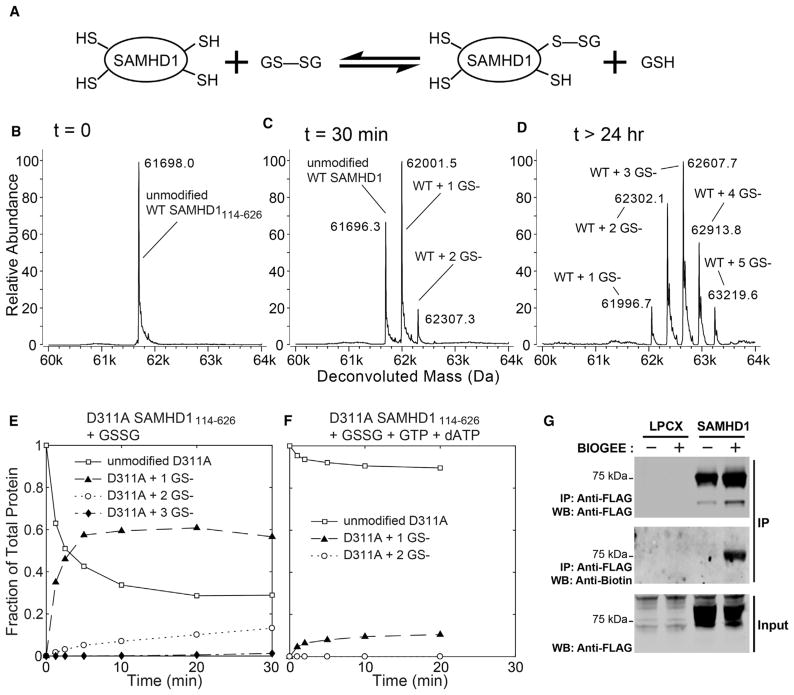
Figure 1. SAMHD1 S-Glutathionylation In Vitro and in Cultured U937 Cells.
(A) Disulfide exchange reactions with oxidized glutathione can be used to identify redox-active cysteines in proteins.
(B–D) Analysis of SAMHD1 S-glutathionylation by intact-mass ESI-TOF mass spectrometry. Deconvoluted mass spectra of the purified HD domain construct of SAMHD1114–626 before (B) and after (C) a 30-min incubation with 5 mM oxidized glutathione reveal formation of covalent glutathione conjugates. (D) Overnight incubation with oxidized glutathione results in modification of the protein with up to five glutathione molecules per SAMHD1 monomer.
(E and F) Time course experiments reveal that SAMHD1 glutathionylation depends on its oligomerization state. Rapid monoglutathionylation of the catalytically inactive mutant D311A (E) is abolished upon addition of GTP and dATP nucleotides, which promote SAMHD1 tetramerization (F).
(G) Glutathionylation of immunopurified full-length SAMHD1-FLAG can be detected by western blot analysis after incubation of SAMHD1-expressing, PMA-treated human U937 cells with biotinylated glutathione (BIOGEE).
See also Figures S1 and S3.