Abstract
The effects of diatomite, activated clay and acticarbon on the decolorization of orange essential oil were investigated. Single factor and orthogonal tests were performed to determine the optimum discoloring conditions. The results showed that the activated clay exhibited the most satisfactory effect on discoloring. Then it was used as the decolorizer for the decolorization of orange essential oil. The highest decolorization rate (84.5%) was obtained using 10% activated clay at 60 °C for 30 min. The contents of oxygenated compounds (linalool and citral) increased from 1.4 to 3.1% after decolorization. Sensory assessment revealed that the orange essential oil after decolorization using activated clay had a mellow and characteristic orange aroma. Chromaticity analysis showed that it had excellent transparency and yellow color under the optimized condition. Thus, decolorization with activated clay could maintain the quality and prolong the storage of orange essential oil.
Keywords: Orange essential oil, Decolorizer, Aromatic composition, Chromaticity value, Sensory evaluation
Introduction
Orange, also known as yellow fruit, golden globe, and golden orange, is a citrus fruit that belongs to the Rutaceae subfamily. Oranges, by virtue of their nutritional value and desirable flavor, are accepted widely by consumers. They are mainly utilized as fresh fruit, juice, salads and etc. In addition, they provide valuable essential oils. Essential oils of Citrus spp. are widely used worldwide; these oils provide antimicrobial effect against bacteria and fungi [2, 12]. Natural orange essential oil, which exhibits a fresh orange flavor, is used to alleviate insomnia caused by anxiety and relieve tension and pressure. This essential oil is also used to prevent colds, promote collagen formation, and balance the pH of the skin. Citrus essential oils are obtained through mechanical extraction and extensively used in food (beverage and sweets), cosmetics, and pharmaceutical products because of their fragrance and flavor [1].
Essential oils contain abundant monoterpenes, and they are used as food preservatives and functions as natural antioxidants against certain cancers. Dietary monoterpenes exhibit antitumor activity and thus prevent the formation and progression of cancer. Limonene and perillyl alcohol confer protection against different cancer types [13]. Limonene is unstable under light and heat and can be easily oxidized, thereby reducing the quality and aroma of orange essential oil. Moreover, oils contain 1–10% oxygenated compounds which are the main aroma source of orange essential oil [6, 8, 16, 18].
Most non-decolorized essential oils in China exhibit the original deep yellow color. In this regard, the color quality of orange essential oil must be improved to satisfy global requirements. Decolorization can decrease the amount of color pigments produced by compounds such as carotene, carotenoid, xanthophyll, and chlorophyll in oil to the acceptable levels [27]. Discoloration and purification of orange essential oil are mainly conducted to: (1) decrease the deterioration rate of essential oil and prolong its storage by removing terpene hydrocarbons; (2) increase the proportion of oxygenated compounds and enhance the intensity of orange essential oil aroma; and (3) improve the color quality of the essential oil. This study aimed to investigate the effects of diatomite, activated clay and acticarbon on the decolorization of orange essential oil. Single factor and orthogonal tests were performed to determine the optimum discoloring conditions. Gas chromatography-mass spectrometry (GC–MS), chromaticity analysis, and sensory evaluation were used to assess the decolorization effects.
Materials and methods
Materials
Orange essential oil used in this work was provided by Qu Gu Food Co., Ltd (Zigui, China). It was extracted by cold grinding method, and then it was treated with a three-phase separation centrifuge in the factory. After the essential oil was sent to the laboratory, it was dried over using anhydrous sodium sulfate and was stored at 4 °C. Diatomite, activated clay, and acticarbon were purchased from Xuyi Xinyuan Technology Co., Ltd (Nanjing, China). Table 1 shows the physicochemical properties of activated clay and acticarbon. Diatomite, which consists essentially of amorphous silica (SiO2·nH2O) has highly porous structure, low density and high surface area. And ethanol was purchased from Chinese Medicine Group Chemical Reagent Co., Ltd (Xuyi, China). Standard solutions of linalool, citral, and d-limonene were obtained from Sigma Chemical Company (Saint Louis, MO, USA). Tartaric acid and glucose syrup were acquired from Hualin Chemical Co., Ltd. (Nanjing, China) and Wanda Food Additive Co., Ltd (Beijing, China), respectively.
Table 1.
Physicochemical properties of activated clay and acticarbon
| Physicochemical properties of activated clay | Technical index of acticarbon | ||
|---|---|---|---|
| pH | 3.3 | The methylene blue decolorizing ability (mL) | 14 |
| SiO2 (wt%) | 83.5 | ||
| Al2O3 (wt%) | 11.3 | Loss on drying (%) | 7 |
| Fe2O3 (wt%) | 1.5 | ||
| MgO (wt%) | 2.5 | Iron content (%) | 0.04 |
| CaO (wt%) | 0.4 | ||
| Na2O (wt%) | 0.4 | Acid soluble (%) | 1.8 |
| K2O (wt%) | 0.6 | ||
| TiO2 (wt%) | 0.2 | The content of calcium and magnesium (%) | 0.2 |
| BETsurface (m2/g) | 240 | ||
| Pore value (cc/g) | 0.38 | pH | 5.7 |
Decolorizer and single-factor test
The adsorption and decolorization effects of diatomite, activated clay, and acticarbon on orange essential oil were compared to select the most suitable decolorizer. A certain amount of the decolorizer was added to 20 mL of orange oil, and the mixture was placed in a thermostatic water bath at 70 °C for 20 min. The sample was then filtered to obtain a pellucid discoloring solution of orange essential oil. Decolorization rate was calculated under ultraviolet spectrophotometer after filtering, and the decolorization effect was then assessed.
A single-factor experiment was conducted using three parameters: amount of decolorizer, discoloring time, and discoloring temperature. Each parameter was tested in triplicate.
Optimization of decolorization conditions
Decolorizer amount, decolorization temperature, and decolorization time were determined through orthogonal test, which was based on the single-factor experiment. Decolorization rate was considered as the evaluation index. The discoloring conditions for orange oil were then optimized.
Calculation of decolorization rate
Absorption at 424 nm was determined in the preliminary experiment through UV spectrophotometer (Pharmaspec UV-1700, Shimadzu, Kyoto, Japan) of orange oil. Absorbance of the orange oil diluent at 424 nm was compared before and after the discoloration. Decolorization rate was then calculated using changes in the maximum absorbance (A) with the following formula:
where A, was the absorbance of orange essential oil at 424 nm before the discoloration; B, was the absorbance of orange essential oil at 424 nm after the discoloration.
Gas chromatography–mass spectrometry analysis
Essential oils were subjected to an Agilent 6890N GC coupled with an Agilent 5975B mass spectrometer (Agilent Technologies, Palo Alto, CA, USA) and equipped with a J&W HP-5MS fused silica capillary column (30 m × 0.25 mm i.d., 0.25 µm-thick film). The mass spectrometer was operated in the electron ionization mode at a voltage of 70 eV. The flow rate of helium in the HP-5 column was 1.2 mL/min. A 0.75 mm liner was used, and analysis was performed in the splitless mode and the injector temperature was 250 °C. The column was held at 40° for 1 min, increased from 40 to 220 °C at 6 °C/min, held at 220 °C for 1 min, and increased to 250 °C at a rate of 30 °C/min.
Quantitative determination was conducted using the external standard method. Standard curves of linalool, citral and d-limonene were determined and used to calculate their contents from the GC-peak areas. The standard curves of the aroma compounds were established by the linear relationship between a series of concentration gradients of aroma compounds (n × 1/6, n × 2/6, n × 3/6, n × 4/6, n × 5/6, n × 6/6, n × 7/6 (n = 50 μg/mL)) and their GC peak areas. The equations of the standard curves of 3 aroma compounds were: d-limonene (y = 2 × 108x + 9 × 108, R2 = 0.995), citral (y = 1 × 109x + 3 × 108, R2 = 0.999), linalool (y = 1 × 108x + 7 × 107, R2 = 0.996). The contents of linalool, citral and d-limonene were calculated according to the standard curves and then it was expressed as % in order to obtain the percentages of these three aroma compounds in the oil samples.
Color analysis
The parameter a*, which ranges from − 80 to + 80, takes positive and negative values for reddish and greenish colors, respectively. Whereas b*, which ranges from − 80 to + 80, takes positive and negative values for yellowish and bluish colors, respectively. L* is the estimated value of the relative luminosity, which ranges from black (L* = 0) to white (L* = 100) [7, 28]. L*, a* and b* were determined using a Hunterlab Ultrascan XE colorimeter. A glass utensil containing the same quantity of samples was placed at the light port (50 mm in diameter). The values were read directly by the colorimeter.
Sensory evaluation
Sensory analysis was conducted by 11 assessors from the College of Food Science and Technology, Huazhong Agricultural University who were appropriately trained before the evaluation according to the guidelines of the ISO 8586-1 (1993) and have passed the screening tests, in order to familiarize the sensory attributes of the essential oil and to improve their ability to recognize, identify and quantify the appearance, aroma, color, and taste of the essential oil samples.
Each sample (5 mL) was placed in a 10 mL coded flask for aroma testing. Approximately 2 cm of the extremity of the fragrance blotter paper (142 mm × 6 mm) was immersed in the respective juices for 0.5 min and then presented to the assessors.
A taste medium (sugar–acid–water solution) were prepared by mixing 0.3 mL of 50% citric acid solution with 250 mL of 68° Brix Ce syrup and 750 mL of water. The medium was then mixed with the crude oil and orange oil discolored by activated clay, acticarbon, and diatomite, respectively. The assessors were asked to taste each sample and identify those with satisfactory and unsatisfactory taste. The assessors should use warm water for rinsing after tasting a sample to prevent any effect on the taste of the next sample.
All sensory tests were conducted in triplicate at room temperature. Intensity was evaluated on a scale of 1–9 (1 = very weak intensity, 3 = weak intensity, 5 = moderate intensity, 7 = strong intensity, and 9 = very strong intensity). The results of the sensory profile analysis were averaged for each parameter and then plotted in a spider web diagram.
Statistical analysis
The difference among groups was subjected to one-way ANOVA using the SPSS 19.0 for Windows (SPSS Inc., Chicago, USA) to determine statistical significance. The mean and standard deviation of the parallel experiments and the external standard method were determined using Excel 2010 (Microsoft Inc., City of Seattle, USA).
Results and discussion
Effect of different decolorizers on the decolorization effect on orange essential oil
The results showed that activated clay exhibited the best effect on decolorization with a decolorization rate of 74.3%, whereas the effect of acticarbon on the decolorization was the worst among these three decolorizers with a decolorization rate of 27.4%. And the decolorization rate of diatomite was 54.8%. In this study, activated carbon presented the optimal decolorization performance; and previous study also showed that acid-activated bentonites were the most effective in removing color from refined oils [15]. Activated clay contains bentonite clay, and it exhibits high adsorption capacity for carotenoids and other pigments of orange essential oil, thereby it could improve the oxidation parameters and the sensory quality of the oil [19, 22, 23]. Activated clay is also an efficient material used for the decolorization of crude oil [3, 20].
Effect of different decolorizers on the aroma compositions of orange essential oil
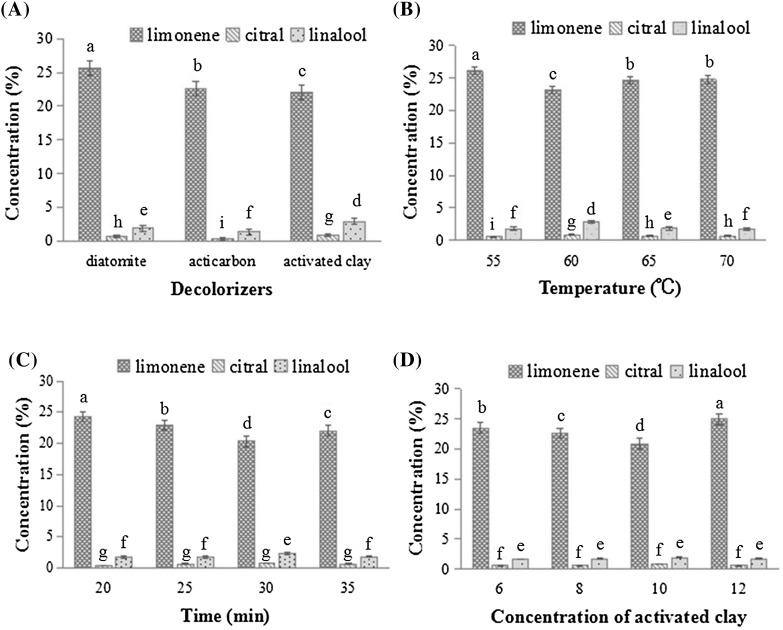
The aroma composition of orange essential oil was determined by GC–MS analysis. The contents of limonene, citral, and linalool were calculated according to the standard curve. Orange essential oil contained high amounts of limonene (94%) and low quantities of oxygenated compounds, such as citral (0.4%) and linalool (0.98%). This result is in accordance with the findings of previous studies that limonene was about 94%, citral 0.14%, and linalool 0.89% in orange essential oil [24, 26]. The effect of different decolorizers on the aroma compositions of orange essential oil is shown in Fig. 1(A). The highest limonene content was found in orange essential oil discolored by diatomite, followed by acticarbon and then activated clay. Moreover, the highest amounts of citral and linalool were detected in orange oil discolored by activated clay, followed by diatomite and then acticarbon. This indicated that the effect of the activated clay on the retention of the oxygenated compounds in orange essential oil during decolorization was the optimal among these three decolorizers.
Fig. 1.
Effect of different decolorizers (A), temperatures (B), heating duration (C) and decolorizer concentration (D) on the aromatic composition of orange essential oil. Columns marked by different letters are significantly different (p < 0.05)
Effect of different decolorizers on the chromaticity of orange essential oil
The initial L*, a*, and b* values of orange essential oil were 50.7, 11.7, and 56.8, respectively. The CIELAB coordinates exhibited dark brown color, which can be attributed to pigments such as carotenoids [11].
Different types of decolorizers significantly influenced the L* and b* values [Fig. 2(A)]. The highest L* value was obtained when the oil was decolorized by activated clay, followed by diatomite and then acticarbon. L* is a brightness index and ranges from 0 (black) to 100 (white). Therefore, the highest brightness was found in orange essential oil discolored by activated clay, followed by diatomite and then acticarbon. Furthermore, a* and b* are color quality indices. And a* represents the red–green dimension and ranges from − 80 (green) to + 80 (red), whereas b* represents the yellow–blue dimension and ranges from − 80 (blue) to + 80 (yellow). The highest b* value was detected in orange oil discolored by activated clay and was found to be within the range of 60–70, which showed a yellowish color. The highest a* value was observed in orange essential oil discolored by diatomite and was found to be within the range of 10–20, which showed a reddish color.
Fig. 2.
Effect of different decolorizers (A), temperatures (B), heating duration (C) and decolorizer concentration (D) on the chromaticity of orange essential oil. Columns marked by different letters are significantly different (p < 0.05)
Sensory evaluation of essential oil treated by the three decolorizers
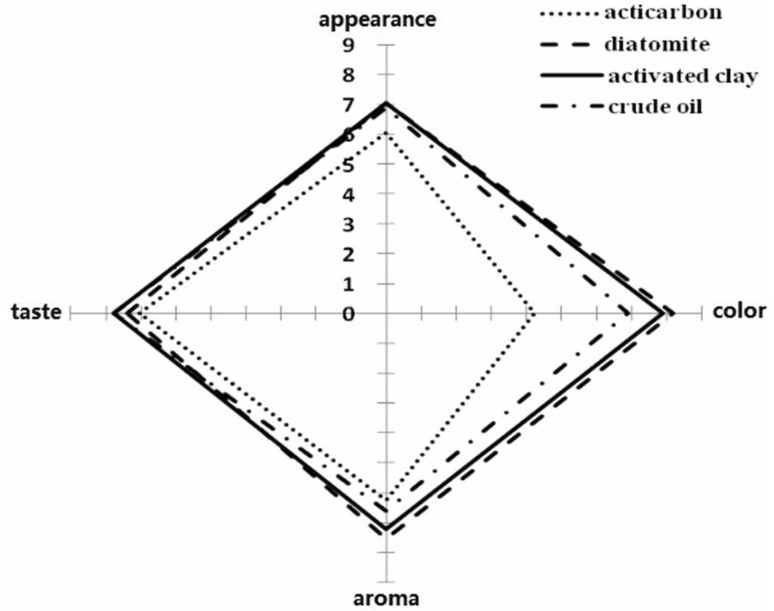
The results of the sensory evaluation of the essential oil treated by the three decolorizers are shown in Fig. 3. The assessors rated the quality of orange essential oil discolored by activated clay as the most satisfactory, followed by diatomite. All of the p values of each sample were less than 0.001.
Fig. 3.
Sensory evaluation of essential oil treated with the three decolorizers
The given marks by assessors were reflected in the axis of the corresponding index in the radar graph (Fig. 3). The highest scores for appearance, color, and aroma were found in orange essential oil discolored by activated clay. And the highest score for taste was found in crude orange essential oil. This finding indicated that the quality of orange essential oil discolored by activated clay was the best. Hence, activated clay was the most suitable decolorizer for orange essential oil.
Effect of activated clay concentration on the decolorization of orange essential oil
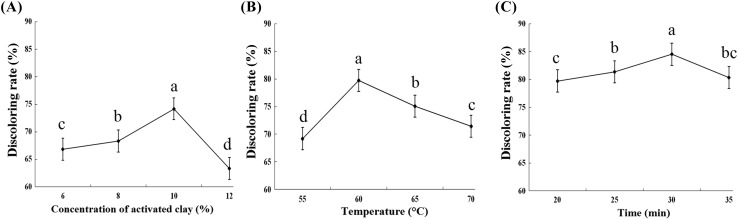
The results showed that the concentration of activated clay affected the decolorization. As shown in Fig. 4(A), the decolorization rate initially increased and then decreased as the amount of activated clay increased from 6 to 12%. The decolorization rate reached to the highest level of 74.2% when the amount of activated clay was 10%. The decolorization rate increased more significantly when the amount of activated clay was within 8–10% than that within 6–8%. This finding indicated that high amounts of activated clay could improve the decolorization effect. Rossi et al. [21] studied the role of activated clay in removing pigments, and the result showed a clear dependence of pigment reduction on clay concentration. Similarly, Dubravka et al. [4] found that high clay content enhanced the removal of impurities in oil. However, the decolorization rate rapidly decreased to 63.3% when the amount of activated clay increased to 12%. Hence, the amount of activated clay should be controlled in a certain amount. Additionally, the excessive use of activated clay can also cause environmental problems because of increased landfill disposal [10]. Therefore, the most suitable amount of activated clay should be 12%.
Fig. 4.
Effect of decolorizer amount (A), temperatures (B) and bleaching time (C) on the decolorizing effect of orange essential oil. Columns marked by different letters are significantly different (p < 0.05)
Effect of temperature on the decolorization effect of orange essential oil
Physical and chemical characteristics changes, as well as energy exchange, occurred during adsorption and decolorization. The effect of temperature on the decolorization rate was studied using 10% activated clay for 20 min [Fig. 4(B)].
As shown in Fig. 4(B), the decolorization effect decreased significantly at relative high or low temperature. It initially increased and then decreased with the increased of the temperature from 55 to 70 °C. The decolorization rate reached the maximum value at 60 °C and then decreased at temperatures exceeding 60 °C. This result could be attributed to the pigment adsorption by the decolorizer, and this reaction was determined as exothermic. This process hindered pigment adsorption and induced the original colorless chromogen to form a new pigment, which is difficult to be removed at high temperatures. It is reported that the effective decolorization temperatures for soy and palm oils were 110 °C [17, 25], respectively. And the optimal decolorization temperature was at 60 °C with the decolorization rate of 80%.
Effect of time on the decolorization of orange essential oil
The heating time also influenced the decolorization rate of orange essential oil. A single-factor experiment was performed to determine the optimal decolorization time by controlling other conditions. The experiment was performed at 60 °C by using 10% activated clay to determine the most effective decolorization time [Fig. 4(C)].
As shown in Fig. 4(C), the decolorization rate increased with the increase of decolorization time from 20 to 30 min. This finding indicated that the increase of the decolorization time was conducive for decolorization of orange essential oil using activated clay. However, decolorization rate decreased when the decolorization time exceeded 30 min. The decolorizer and oil could easily come in full contact, and this hastened the speed of mass exchange. Consequently, the decolorization effect became evident [5]. A part of the pigment in orange essential oil will be desorbed with the increase of the decolorization time. In addition, the adsorption of activated clay could accelerate the oxidation of several components in orange essential oil, and this led to the formation of a deep color [25]. Therefore, the most effective decolorization time for the orange essential oil using activated clay was 30 min.
The optimization of decolorization conditions using activated clay
A 43 factorial design was used to determine the optimum decolorization conditions through a single-factor experiment with three experimental factors, namely, temperature (A), time (B), and amount of activated clay (C). A factorial experimental design including 16 runs was performed, in which each variable was set at four levels: 55, 60, 65, and 70 °C for temperature; 6, 8, 10, and 12% for amount of activated clay; and 20, 25, 30, and 35 min for time. Decolorization rate of the orange essential oil using activated clay was set as the index. The results are shown in Table 2.
Table 2.
Results of orthogonal experiment
| No. | A (temperature) | B (time) | C (amount of activated clay) | Decolorization rate (%) |
|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 69.2 |
| 2 | 1 | 2 | 2 | 65.3 |
| 3 | 1 | 2 | 3 | 71.7 |
| 4 | 1 | 4 | 4 | 68.8 |
| 5 | 2 | 1 | 1 | 79.7 |
| 6 | 2 | 2 | 2 | 80.4 |
| 7 | 2 | 3 | 3 | 84.5 |
| 8 | 2 | 4 | 4 | 81.4 |
| 9 | 3 | 1 | 1 | 75.1 |
| 10 | 3 | 2 | 2 | 71.4 |
| 11 | 3 | 3 | 3 | 79.7 |
| 12 | 3 | 4 | 4 | 69.2 |
| 13 | 4 | 1 | 1 | 68.4 |
| 14 | 4 | 3 | 2 | 66.9 |
| 15 | 4 | 4 | 3 | 74.2 |
| 16 | 4 | 3 | 4 | 63.3 |
| K1 | 68.74 | 73.08 | 73.08 | |
| K2 | 81.49 | 72.19 | 70.97 | |
| K3 | 73.85 | 73.61 | 77.54 | |
| K4 | 68.18 | 73.39 | 70.67 | |
| R | 13.31 | 1.42 | 6.87 | |
| Primary factor | A | C | B | |
| Optimal plan | A2 | B3 | C3 | |
| Sum of squares of deviations | 124.005298 | 8.008123 | 49.007398 | |
| Degrees of freedom | 3 | 3 | 3 | |
| Value of F | 13.00447 | 6.003407 | 3.003679 | |
| Critical value of F | 9.12 | 9.12 | 9.12 |
Table 2 showed that decolorization temperature significantly influenced the experimental results compared with decolorizer amount and decolorization time. K values showed the following trends: A2 > A3 > A1 > A4; B3 > B4 > B1 > B2; and C3 > C1 > C2 > C4. Thus, the most suitable decolorization temperature, amount of activated clay, and decolorization time were 60 °C, 10%, and 30 min, respectively. And the decolorization rate was 84.5% under the optimized conditions. Fan et al. [5] reported that the optimum decolorization conditions for sesame oil were the use of 3% activated clay at 80 °C for 30 min. In the present study, the optimum decolorization conditions were as follows: decolorization temperature 60 °C, activated clay amount 10%, and time 30 min.
Temperature (A) significantly influenced the decolorization effect of the orange essential oil, whereas amount (C) and time (B) showed insignificant effects. The significance of the effects of these parameters could be arranged in the following order: temperature > amount of activated clay > time.
Effect of temperature on the aroma compositions and chromaticity of orange essential oil treated with activated clay
As shown in Fig. 1(B), limonene content in orange essential oil discolored by activated clay decreased significantly as the temperature increased from 55 to 60 °C and then increased significantly as the temperature further increased from 60 to 65 °C. And then it increase slowly when the temperature increased from 65 to 70 °C. The lowest limonene content in orange essential oil discolored by activated clay was found in the sample heated at 60 °C. The changes in citral and linalool contents in orange essential oil were similar. The contents increased first and then decreased with increase of temperature.
As shown in Fig. 2(B), heating temperature minimally influenced the a* value. Meanwhile, the b* value increased when the temperature increased from 55 to 60 °C and decreased at temperatures exceeding 60 °C. Therefore, the b* value was the highest at 60 °C. And the color of the orange essential oil exhibited the strongest bias toward yellow at this temperature. Moreover, the L* value reached the maximum at 60 °C, at which the brightness of orange essential oil was the highest.
Effect of activated clay concentration on the aroma compositions and chromaticity of orange essential oil
As shown in Fig. 1(D), the decolorizer concentration minimally influenced the content of oxygenated compounds, which increased slowly as the activated clay amount increased from 6 to 10%. However, the content of oxygenated compounds decreased when the activated clay concentration further increased from 10 to 12% and reached the highest value when the concentration was 10%. By contrast, changes in the concentration of activated clay significantly affected limonene content in orange essential oil. Limonene content decreased continuously as the amount of activated clay increased from 6 to 10% and then increased rapidly when the amount increased from 10 to 12%. Furthermore, the lowest limonene content was found in the sample added with 10% activated clay.
As shown in Fig. 2(D), the concentration of activated clay minimally influenced the L* and b* values in the chromaticity of orange essential oil. Both the L* and b* values were very high when the clay amount was 10%. Moreover, the influence of different amounts of the decolorizer on the a* value was not significant. When 10% activated clay was added, the a* value was significantly lower than those in other cases. In summary, the brightness of orange essential oil was high with a strong bias toward yellow and a low bias toward red after the addition of 10% activated clay.
Effect of heating time on the aroma compositions and chromaticity of orange essential oil treated with activated clay
As shown in Fig. 1(C), heating time significantly influenced limonene content in orange essential oil. Limonene content decreased with the increase of the heating time from 20 to 30 min and then rapidly increased from 30 to 35 min. Moreover, the lowest limonene content was found in the sample heated for 30 min. However, heating time did not significantly affect the content of oxygenated compounds in orange essential oil. Linalool content initially increased slowly and then significantly increased; meanwhile, citral content continued to increase slowly as the heating time increased from 20 to 30 min. When the heating time was further increased from 30 to 35 min, linalool content significantly decreased and citral content slightly decreased. Both citral and linalool contents reached the maximum values when the sample was heated for 30 min.
The lowest limonene content and the highest linalool and citral levels were observed in the sample added with 10% activated clay and heated at 60 °C for 30 min. Limonene is unstable under light and heat and could be easily oxidized, leading to reduced quality and aroma of orange essential oil. Whereas, linalool and citral belonged to oxygenated compounds and they are the main aroma sources of orange essential oil [6, 8, 14, 16]. This indicated that the highest linalool and citral levels and the lowest limonene content led to the highest oil quality and stability. Therefore, the highest oil quality and stability was found in orange essential oil samples discolored under the optimized conditions.
As shown in Fig. 2(C), the a* value was slightly higher, whereas the b* value fluctuated considerably and was significantly higher when heated for 30 min than those in other treatments. By contrast, changes in the L* value was relatively slow and reached the maximum value after heating for 30 min, during which the brightness of orange essential oil was the highest.
Color stability of crude and discolored orange essential oil treated with activated clay
The chromaticity values of orange essential oil discolored by activated clay and crude orange oil were measured during a 10 days’ storage. The effect of decolorization using activated clay on the storage duration of orange essential oil was also investigated.
As shown in Fig. 5(A), the chromaticity values of crude orange essential oil changed slowly. Changes in the chromaticity value were significant at prolonged storage. This indicated that long-term preservation of orange essential oil could induce significant color changes. However, Fig. 5(B) showed that changes in the chromaticity values of orange essential oil discolored by activated clay was minimal during prolonged storage. This finding indicated that decolorization with activated clay could maintain orange essential oil color and prolong its storage.
Fig. 5.
Changes in the chromaticity values of crude (A) and discolored essential oil (B) treated with activated clay. Columns marked by different letters are significantly different (p < 0.05)
Optimizing the decolorization condition of oil samples could lead to the production of yellowish oils, whereas the color intensity of the oil decreased after the chroma reduction [9]. As shown in Fig. 2(A), the highest L* and b* values were found in the orange essential oil discolored by activated clay. This finding indicated that color quality was the most satisfactory when activated clay was used. Therefore, activated clay was the most suitable decolorization agent. As shown in Fig. 2, orange oil showed the highest brightness and the most yellowish color when the activated clay amount, heating time, and temperature were 10%, 30 min, and 60 °C, respectively; hence, the most satisfactory color quality can be obtained under this condition.
In conclusion, the most effective decolorization rate (84.5%) was obtained when using 10% decolorizer at 60 °C for 30 min. Moreover, orange essential oil discolored by activated clay exhibited a mellow and characteristic orange aroma, with satisfactory appearance and taste. Under the optimized condition, the content of limonene, which belongs to terpene hydrocarbons, decreased from 94.4 to 22.1%, whereas the linalool and citral contents increased from 1.4 to 3.1%. Furthermore, discolored oil exhibited the highest color quality and excellent transparency, with a yellow color similar to the original sample. Therefore, decolorization using activated clay could maintain the quality of orange essential oil and prolong its storage duration.
Acknowledgements
This study was supported by the National key Research and Development Program of China (2017YFD0400101), National Natural Science Foundation of China (Program No. 31671824), the Major Scientific and Technological Innovation Project in Hubei Province (2015ABA035, 2016ABA112).
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
References
- 1.Capparucci C, Frattari S, Gironi F. Supercritical equilibrium data of the systems carbon dioxide-linalool and carbon dioxide-orange essential oil. Am. J. Ana. Chem. 2013;4:763–770. doi: 10.4236/ajac.2013.412093. [DOI] [Google Scholar]
- 2.Chanthaphon S, Chanthachum S, Hongpattarakere T. Antimicrobial activities of essential oils and crude extracts from tropical Citrus spp. against food-related microorganisms. Songklanakarin. J. Sci. Technol. 2008;30:125–131. [Google Scholar]
- 3.Chung YY, Kim SR, Kang HG, Noh YS, Park MC, Finkel D, An G. Characterization of two rice MADS box genes homologous to GLOBOSA. Plant Sci. 1995;109:45–56. doi: 10.1016/0168-9452(95)04153-L. [DOI] [Google Scholar]
- 4.Dubravka S, Tomislav D, Klara K, Jasenka GK, Sandra N, Marko O. Optimization of bleaching parameters for soybean oil. Food Technol. Biotech. 2012;50:199–207. [Google Scholar]
- 5.Fan ZJ, Chen YP, Liu GH. Study on the decolouring property of activated bentonite in sesame oil. Food Eng. 2007;4:26–27. [Google Scholar]
- 6.Fanciullino AL, Tomi F, Luro F, Desjobert JM, Casanova J. Chemical variability of peel and leaf oils of mandarins. Flavour Fragr. J. 2006;21(2):359–367. doi: 10.1002/ffj.1658. [DOI] [Google Scholar]
- 7.García-Moreno PJ, Guadix A, Gómez-Robledo L, Guadix EM. Optimization of bleaching conditions for sardine oil. J. Food Eng. 2013;116:606–612. doi: 10.1016/j.jfoodeng.2012.12.040. [DOI] [Google Scholar]
- 8.Hosni K, Zahed N, Chrif R, Abid I, Medfei W, Kallel M, Brahim NB, Sebei H. Composition of peel essential oils from four selected Tunisian Citrus species: Evidence for the genotypic influence. Food Chem. 2010;123:1098–1104. doi: 10.1016/j.foodchem.2010.05.068. [DOI] [Google Scholar]
- 9.Huang J, Sathivel S. Purifying salmon oil using adsorption, neutralization, and a combined neutralization and adsorption process. J. Food Eng. 2010;96:51–58. doi: 10.1016/j.jfoodeng.2009.06.042. [DOI] [Google Scholar]
- 10.Hussin F, Aroua MK, Daud WMAW. Textural characteristics, surface chemistry and activation of bleaching earth: A review. Chem. Eng. J. 2011;170:90–106. doi: 10.1016/j.cej.2011.03.065. [DOI] [Google Scholar]
- 11.Indrasena WM, Barrow CJ. Oxidation stability of food-grade fish oils: Role of Antioxidants. In: Alasalvar C, Shahidi F, Miyashita K, Wanasundara U, editors. Handbook of Seafood Quality. Oxford: Safety and Health Applications. Wiley; 2010. [Google Scholar]
- 12.Jafari S, Esfahani S, Fazeli MR, Jamalifar H, Samadi M, Samadi N, Najarian Toosi A, Shams Ardekani MR, Khanavi M. Antimicrobial activity of lime essential oil against food-borne pathogens isolated from cream-filled cakes and pastries. Int. J. Biol. Chem. 2011;5:258–265. doi: 10.3923/ijbc.2011.258.265. [DOI] [Google Scholar]
- 13.Junior MRM, Silva TAA, Franchi GC, Nowill A, Pastore GM, Hyslop S. Antioxidant potential of aroma compounds obtained by limonene biotransformation of orange essential oil. Food Chem. 2009;116:8–12. doi: 10.1016/j.foodchem.2009.01.084. [DOI] [Google Scholar]
- 14.Lota ML, Serra DR, Tomi F, Casanova J. Chemical variability of peel and leaf essential oils of mandarins from Citrus reticulata Blanco. Biochem. Syst. Ecol. 2000;28:61–78. doi: 10.1016/S0305-1978(99)00036-8. [DOI] [PubMed] [Google Scholar]
- 15.Makhoukhi B, Didi MA, Villemin D, Azzouz A. Acid activation of Bentonite for using as vegetable oil bleaching agent. Grasas Y Aceites. 2009;60:343–349. doi: 10.3989/gya.108408. [DOI] [Google Scholar]
- 16.Merle H, Moron M, Blazquez MA, Boira H. Taxonomical contribution of essential oils in mandarins cultivars. Biochem. System. Ecol. 2004;32:491–497. doi: 10.1016/j.bse.2003.09.010. [DOI] [Google Scholar]
- 17.Paquat C, Hautfenne A. Standard methods of analysis of oils, fats and derivatives. International Union of Pure and Applied Chemistry (IUPAC). 7th revised and enlarged edn. London: Wiley; 1987. [Google Scholar]
- 18.Park BJJC, Chung SH, Kim CB, Cha BS, Byun MW. Antibacterial and antifungal activity of citrus (Citrus unshiu) essential oil extracted from peel by-products. Food Sci. Biotechnol. 2004;13:384–386. [Google Scholar]
- 19.Proctor A, Toro-Vazquez JF. The Freundlich isotherm in studying adsorption in oil processing. Bleaching and Purifying Fats and Oils. 1996;73(12):209–219. [Google Scholar]
- 20.Puah CW, Choo YM, Ma AN, Chuah C. Degumming and bleaching: Effect on selected constituents of palm oil. J. Oil Palm Res. 2004;16:57–63. [Google Scholar]
- 21.Rossi M, Gianazza M, Alamprese C, Stanga F. The role of bleaching clays and synthetic silica in palm oil physical refining. Food Chem. 2003;82:291–296. doi: 10.1016/S0308-8146(02)00551-4. [DOI] [Google Scholar]
- 22.Salawudeen TO, Arinkoola AO, Jimoh MO, Akinwande BA. Clay characterization and optimisation of bleaching parameters for palm kernel oil using alkaline activated clays. J. Minerals Materials Characterization Eng. 2014;2:586–597. doi: 10.4236/jmmce.2014.26060. [DOI] [Google Scholar]
- 23.Sathivel S, Prinyawiwatkul W. Adsorption of FFA in crude catfish oil onto chitosan, activated carbon, and activated earth: A kinetics study. J. Am. Oil Chem. Soc. 2004;81(4):493–496. doi: 10.1007/s11746-004-0929-0. [DOI] [Google Scholar]
- 24.Stashenko EE, Martinez R, Pinzon MH, Ramírez J. Changes in chemical composition of catalytically hydrogenated orange oil (Citrus sinensis) J. Chromatogr. A. 1996;752:217–222. doi: 10.1016/S0021-9673(96)00481-5. [DOI] [Google Scholar]
- 25.Tai YH, Lin C. Variation of peroxide value in water-degummed and alkali-refined soy oil during bleaching under vacuum. Sep. Purif. Technol. 2007;56:257–264. doi: 10.1016/j.seppur.2007.01.038. [DOI] [Google Scholar]
- 26.Viuda-Martos M, Ruiz-Navajas Y, Fernández-López J, Pérez-Alvarez JA. Chemical composition of mandarin (C. Reticulata L.), grapefruit (C. Paradisi L.), lemon (C. Limon L.) and orange (C. Sinensis L.) essential oils. J. Essent. Oil Bear. Pl. 2009;1:236–243. doi: 10.1080/0972060X.2009.10643716. [DOI] [Google Scholar]
- 27.Wu ZS, Li C. Kinetics and thermodynamics of β-carotene and chlorophyll adsorption onto acid- activated bentonite from Xinjiang in xylene solution. J. Hazard. Mater. 2009;171:582–587. doi: 10.1016/j.jhazmat.2009.06.047. [DOI] [PubMed] [Google Scholar]
- 28.Wyszecki G, Stiles WS. Color Science: Concepts and methods, quantitative data and formulae. New York: Wiley; 2000. [Google Scholar]