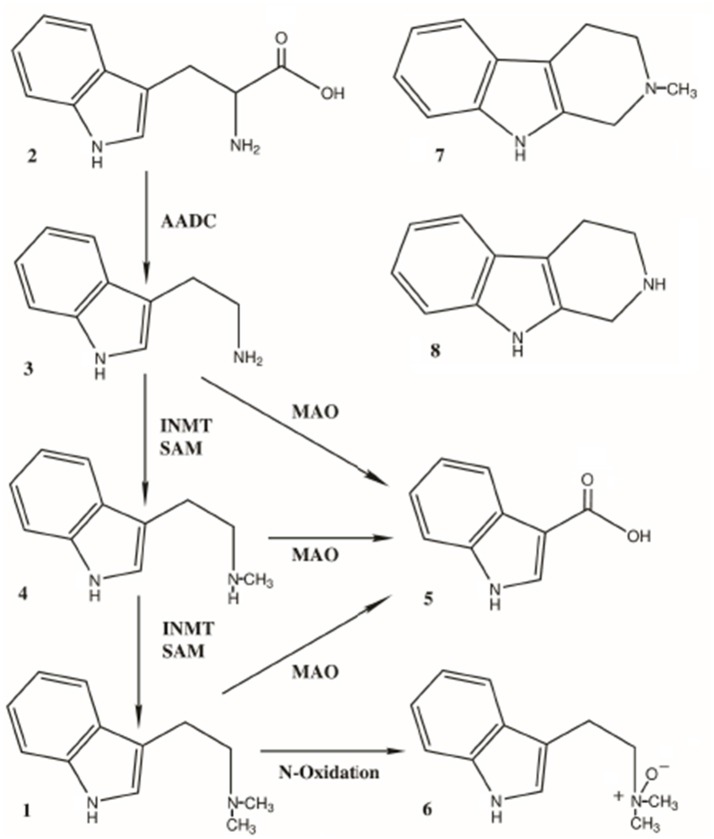
Figure 2.
Pathways for the biosynthesis and metabolism of DMT, 1. Biosynthesis: Tryptophan (2) is converted to tryptamine (TA, 3) by aromatic amino acid decarboxylase (AADC). TA is dimethylated to first yield N-methyltryptamine (NMT, 4) and then DMT (1) by indole-N-methyltransferase (INMT), using S-adenosyl-methionine (SAM) as the methyl source. Metabolism: TA, NMT and DMT are all substrates for monoamine oxidase, yielding indole-3-acetic acid (5, IAA) as both a common precursor metabolite and the most abundant metabolite of DMT itself. DMT is also converted to DMT-N-oxide (6) as the second-most abundant metabolite. Two 1,2,3,4-tetrahydro-beta-carbolines (THBCs) have also been identified as metabolites; 2-methyl-THBC (7, MTHBC) and THBC (8).