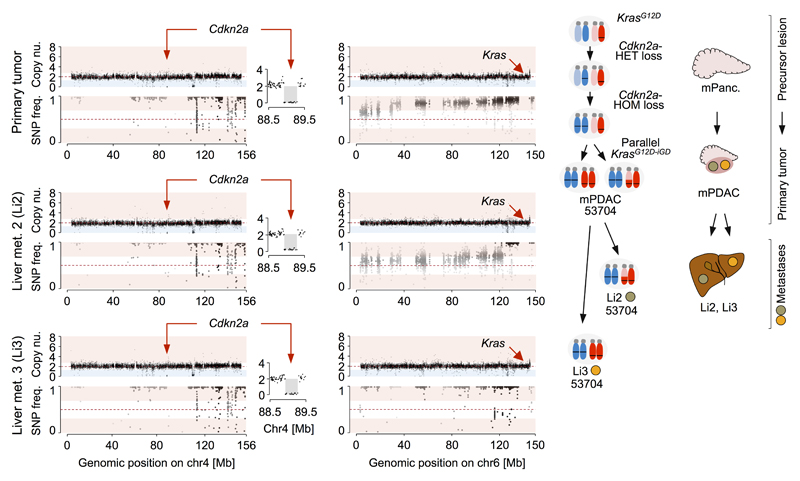
Extended Data Figure 6. Complete Cdkn2a barrier loss precedes KrasG12D-iGD in primary mPDAC of PK mouse 53704.
Copy number alterations at chr4 (Cdkn2a) and chr6 (Kras) in mPDAC 53704 and corresponding metastases, as detected by aCGH (top) and whole-exome sequencing based SNP pattern analysis (bottom). The primary cancer and both liver metastases display identical focal deletions of Cdkn2a and similar SNP patterns on chr4 revealing that all lesions share the same ancestor cell with complete Cdkn2a loss. By contrast, SNP analysis on chr6 revealed discordant patterns in the primary mPDAC and both metastases. Li2 shows partial LOH of a distal region on chr6 involving the Kras locus, while LOH in Li3 involves the whole chr6. This explains the step-wise LOH pattern observed on chr6 in the primary mPDAC. The graphic on the right shows the combined interpretation of CNV/LOH profiles, which suggests the following sequence of genetic events during tumor evolution: The initial KrasG12D mutation was followed by focal deletion of one copy of Cdkn2a. In a subsequent genetic event, the second copy of Cdkn2a was lost by chr4 missegregation and copy-number neutral LOH. Complete barrier loss allowed for convergent evolution of increased KrasG12D gene dosage through copy-number neutral LOH and gave rise to independent metastases in the liver. Note: A major obstacle for equivalent human studies is the limited availability of human matched primary/metastases samples, particularly of treatment naive ones. We performed cross-species analyses using data from a recent study, which analyzed human treatment-naive metastatic PDACs by whole-genome sequencing8 and provided CDKN2A and KRAS copy number data for matched primaries/metastases from 3 patients. In one patient the sequential order of CDKN2A deletion and KRAS amplification could be reconstructed: homozygous CDKN2A deletions were identical in all primaries and metastases, whereas there were 5 different KRAS gains in the 6 metastases. This suggests convergent evolution of mutant KRAS gene dosage gain upon homozygous CDKN2A loss in this patient, in line with similar data in large series of mouse cancers and their metastases (see Figure 3e).