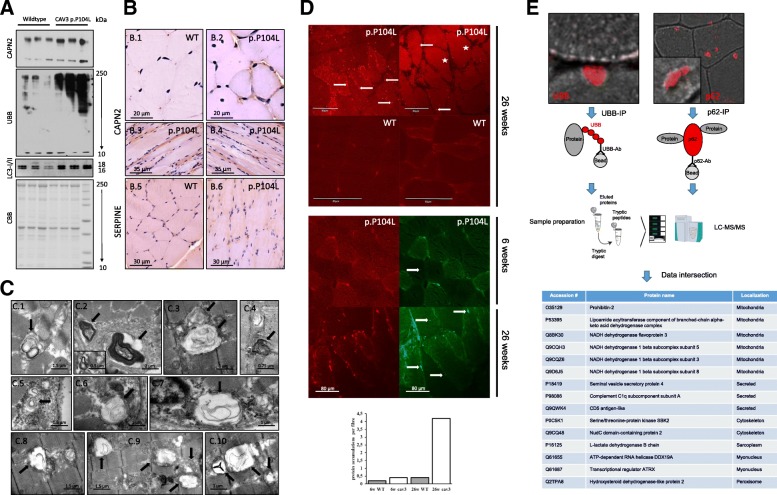
Fig. 6.
Activation of proteolysis and protein aggregation in p.P104L caveolinopathy. A Immunoblot studies confirming the increased abundance of CAPN2 identified in the proteome profile and showing increased abundance of ubiquitinated proteins as well as of LC3. Coomassie brilliant blue was used as loading control. B Increased abundance of CAPN2 was confirmed via immunohistochemical studies which also revealed elevated level of SERPINE (a protein functioning as a protease) at the sub-sarcolemmal region. C EM showing autophagic vacuoles containing electron-dense myelin-like structures (6C.1–6C.7) in quadriceps muscles of animals with advanced myopathy as well as damaged, probably degenerating mitochondria (6C.8–6C.10). D CARS (two photon) microscopy confirms the presence of sarcoplasmic and sub-sarcolemmal protein-dense structures in quadriceps muscle fibres of p.P104 CAV3 transgenic animals (red fluorescent dots) but not in control muscles from wild-type littermates. Concomitant second harmonic generation signals (green fluorescent dots) corresponding to organized molecular structures such as composed myosin and myelin also indicate the presence of small abnormal build-up of organized structures within the sarcoplasm of quadriceps muscle fibres of diseased animals frequently overlapping with the protein aggregates identified by two photon microscopy. Comparative analyses and quantification of aggregates in younger and older animals revealed that aggregate-build-up coincides with disease progression. E Immunoprecipitation of ubiquitinated and p62-binding proteins with subsequent mass spectrometry-based identification towards the molecular characterization of the protein aggregates detected via electron and CARS microscopy. Data intersection revealed similar alteration of 15 proteins