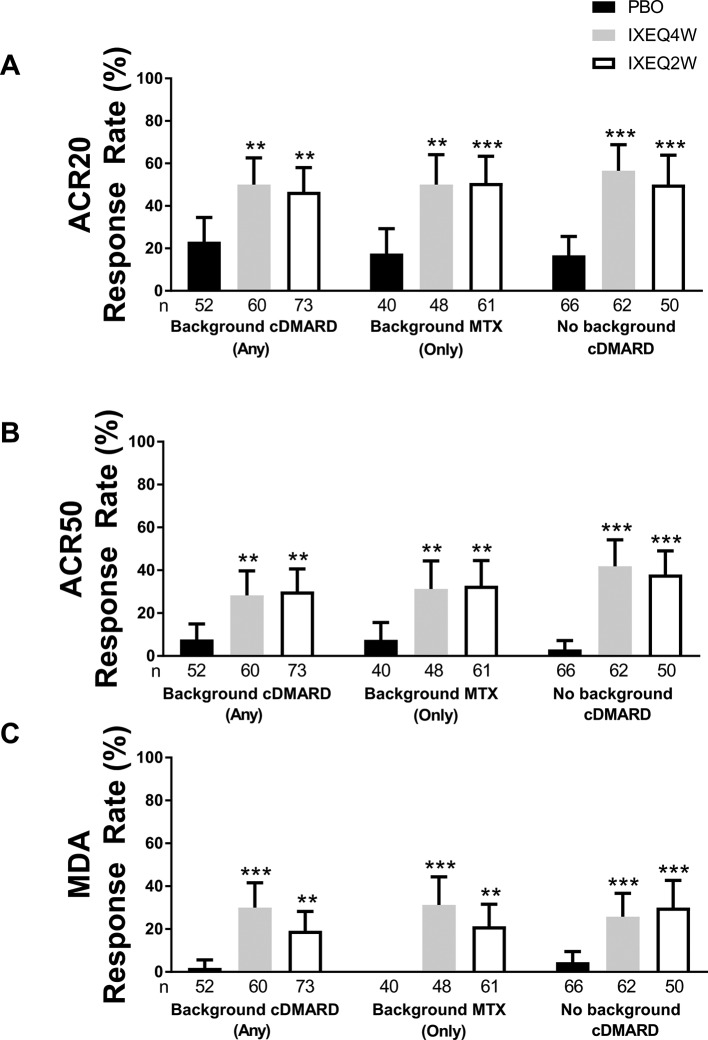
Figure 1.
ACR20 (A), ACR50 (B) and MDA (C) response rates at 24 weeks in patients treated with PBO, IXEQ4W or IXEQ2W alone or when added to background cDMARDs or MTX. ACR20 results in the ITT population for the background cDMARD and monotherapy subgroups were previously published by Nash and colleagues.13 Response rates reported with 95% confidence intervals. The treatment-by-cDMARD interaction p-values for treatment by cDMARD use were 0.5115 for ACR20 and 0.2616 for ACR50.**p<0.01 versus PBO; ***p<0.001 versus PBO. ACR20/50, American College of Rheumatology criteria 20%/50% improvement; cDMARD, conventional disease-modifying antirheumatic drugs; IXEQ4W, ixekizumab every 4 weeks; IXEQ2W, ixekizumab every 2 weeks; ITT, intent-to-treat; MDA, minimal disease activity; MTX, methotrexate; n, number of patients; PBO, placebo.