-
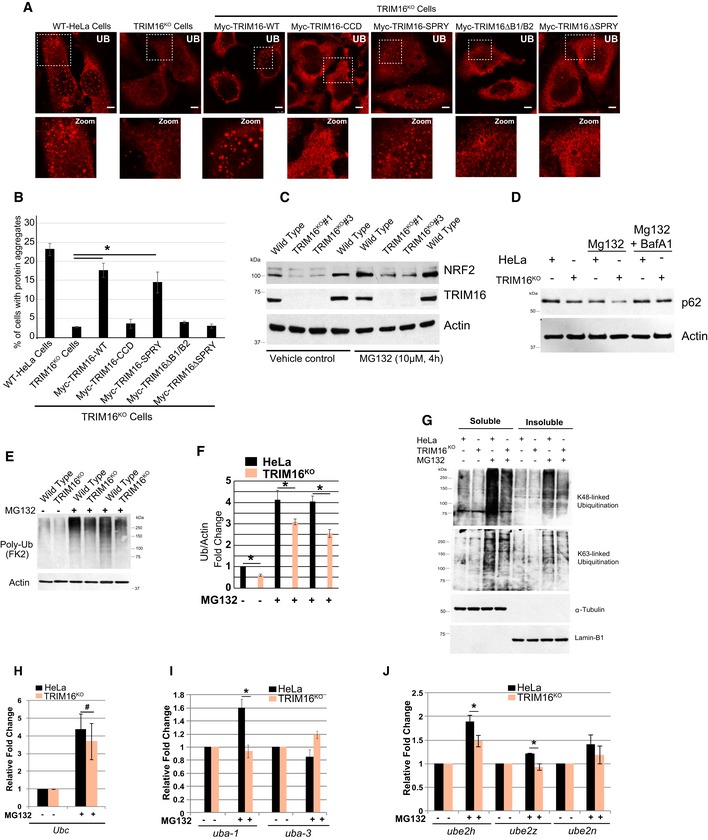
A
Representative confocal images of HeLa, TRIM16KO cells, and TRIM16KO cells complemented with TRIM16 deletion constructs where cells were treated with MG132 (20 μM, 2 h) and the samples were processed for IF analysis with Ub antibody.
-
B
The graph shows the percentage of cells with Ub‐positive protein aggregates. Data from ≥ 10 fields (40×), n = 3, mean ± SD, *P < 0.005 (Student's unpaired t‐test).
-
C
HeLa and TRIM16KO cells were treated with MG132 (10 μM, 4 h), and lysates were subjected to Western blotting with indicated antibodies.
-
D
WB analysis of HeLa and TRIM16KO lysates of cells treated with MG132 (10 μM, 4 h) or/and BafA1 (300 nM, 4 h) as indicated and probed with antibodies as shown.
-
E
HeLa and TRIM16KO cells were treated with MG132 (10 μM, 4 h), and lysates were subjected to Western blotting with indicated antibodies.
-
F
Quantification of UB lane intensity relative to actin. Mean ± SD, n = 3, *P < 0.05.
-
G
HeLa and TRIM16KO cells were treated with MG132 (10 μM, 4 h), and lysates were subjected to Western blotting with K48 and K63 and other indicated antibodies.
-
H–J
RNA isolated from HeLa and TRIM16KO cells, untreated or treated with MG132 (20 μM, 2 h), were subjected to qRT–PCR with primers of genes as indicated. The fold induction in MG132‐treated samples is calculated relative to untreated samples. Mean ± SD, n = 3, *P < 0.05, #Insignificant.
Data information: Unless otherwise stated, scale bar: 10 μm.