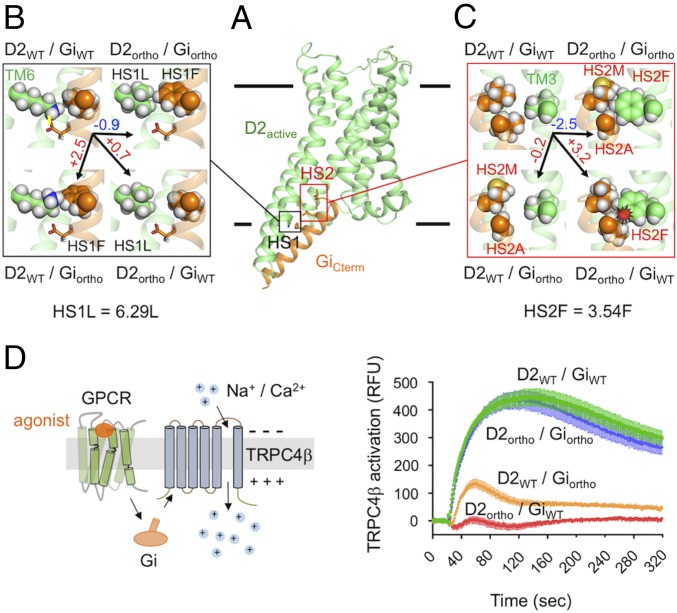
Fig. 2.
Designed D2–Gi orthogonal pair achieves high activity and coupling selectivity. (A) Representative active-state model of D2 (D2active) bound to the C-terminal helix of Gi (GiCterm). The designed binding interface includes two hot-spot motifs (HS1 and HS2). (B) Zoomed view of the D2–Gi interface at HS1. Predicted energetic contribution of designed residues at HS1 to the binding affinity of the designed D2ortho–Giortho and the noncognate ortho/WT pairs. D2 and Gi residues are colored in green and gold, respectively. The highlighted designed D2 residue HS1L (mutation K204L) is located on TM6 at the Ballesteros Weinstein (BW) position 6.29. Stabilization and destabilization effects correspond to negative and positive energy differences from WT, respectively. Energy differences are in Rosetta Energy Units and shown on top of the black arrows. (C) Similar to B but for HS2. The highlighted designed D2 residue HS2F (mutation V100F) is located on TM3 at the BW position 3.54. (D) TrpC4β channel activation upon D2 activation by dopamine through the Gi signaling pathway. Fluorescence changes are measured for cells stably expressing TrpC4β channels cotransfected with plasmids coding the D2 and Gi variants indicated on each curve (SI Appendix, Supplementary Methods). Under the experimental conditions, the D2 variants were expressed at the same level on the cell surface as measured by ELISA (SI Appendix, Fig. S3A).