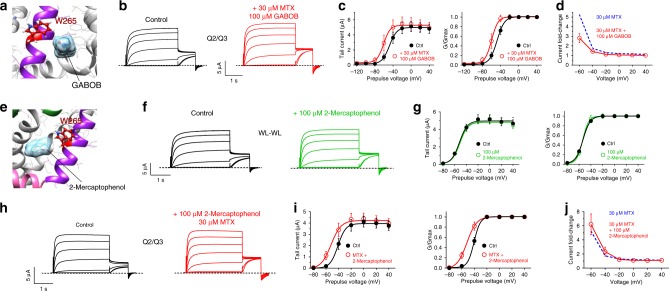
Fig. 4.
MTX outcompetes 2-mercaptophenol to activate KCNQ2/3. a Binding position of GABOB predicted by SwissDock using a chimeric KCNQ1–KCNQ3 structure model. b Exemplar traces showing effects of MTX (30 µM) with GABOB (100 µM) on KCNQ2/3 channels. Voltage protocol as in Fig. 1d. c Effects of MTX (30 µM) with GABOB (100 µM) on mean tail current (left) and G/Gmax (right) relationships for KCNQ2/3 (n = 6) calculated from traces as in panel b. d Current fold-change at −60 mV exerted by MTX (30 µM) alone (from Fig. 2a) or with 100 µM GABOB, from data as in panel c (n = 6). e Binding position of 2-mercaptophenol predicted by SwissDock using a chimeric KCNQ1–KCNQ3 structure model. f Exemplar traces showing effects of 2-mercaptophenol (100 µM) on KCNQ2-W236L/KCNQ3-W265 (WL-WL) channels. Voltage protocol as in Fig. 1d. g Effects of 2-mercaptophenol (100 µM) on mean tail current (left) and G/Gmax (right) relationships for KCNQ2-W236L/KCNQ3-W265 (WL-WL) channels (n = 9) calculated from traces as in panel f. h Exemplar traces showing effects of MTX (30 µM) with 2-mercaptophenol (100 µM) on KCNQ2/3 channels. Voltage protocol as in Fig. 1d. i Effects of MTX (30 µM) with 2-mercaptophenol (100 µM) on mean tail current (left) and G/Gmax (right) relationships for KCNQ2/3 (n = 9) calculated from traces as in panel h. j Current fold-change at −60 mV exerted by MTX (30 µM) alone (from Fig. 2a) or with 100 µM 2-mercaptophenol, from data as in panel i (n = 9). All error bars indicate SEM