Fig. 3.
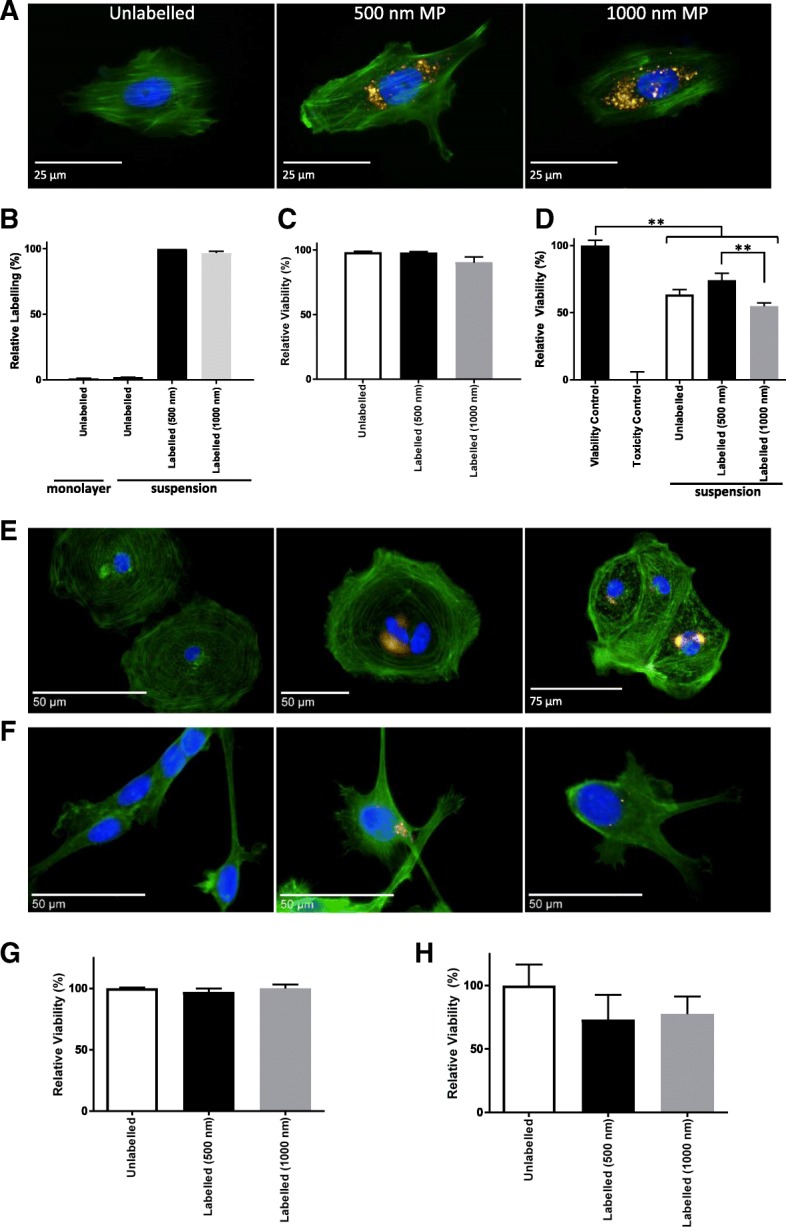
Development of suspension cell labelling approach for human cells. a, b MP-labelled MSC examined by fluorescence microscopy 24 h post labelling (a), after staining with Phalloidin (green) and Hoechst 33342 (blue), and by flow cytometry (b). c, d MSC viability examined by membrane integrity assay (c) and metabolic activity measurement (d) 24 h post labelling. Viable control consisted of unlabelled monolayer cells, Toxicity control consisted of cells exposed to 70% methanol for 10 min. e, f Fluorescence microscopy evaluation of CMC (e) and ReN (f) following suspension labelling with 500 nm (middle panel) or 1000 nm (right panel) MPs (gold) compared to unlabelled cells (left panel), with Phalloidin (green) and Hoechst 33342 (blue) counterstain. g, h Metabolic activity for CMC (g) and ReN (h) 24 h post suspension labelling. Error bars presented as SEM, n = 3. **P ≤ 0.01. MP magnetic particle
