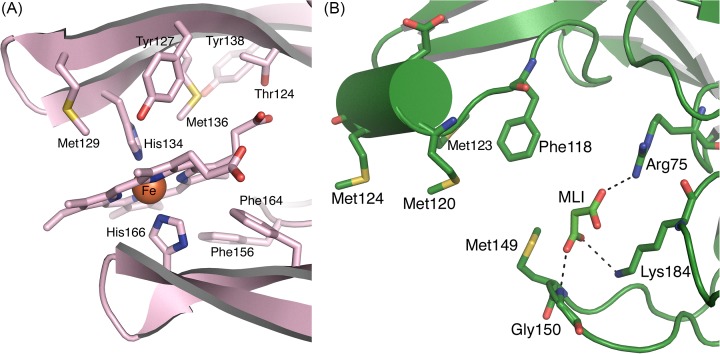
Figure 13. Close-up of the heme-binding pocket in P. gingivalis holo-HmuY (A) and the equivalent site in T. forsythia apo-Tfo (B).
The Tfo site lacks the His residues that are the axial ligands to the Fe-heme in HmuY. The binding pocket is narrower in the apo-Tfo crystal structure, with a malonate ligand (MLI) and the Met149 in positions occupied by the heme in HmuY. The malonate forms H-bonds (2.8-3.1 Å, indicated by dashed lines) to residues Gly150, Arg75, and Lys184 and to water molecules (not shown). Numbering of amino acid residues in the case of Tfo is shown according to the protein lacking the signal peptide sequence (20 amino acid residues), which was crystallized and examined by X-ray analysis.