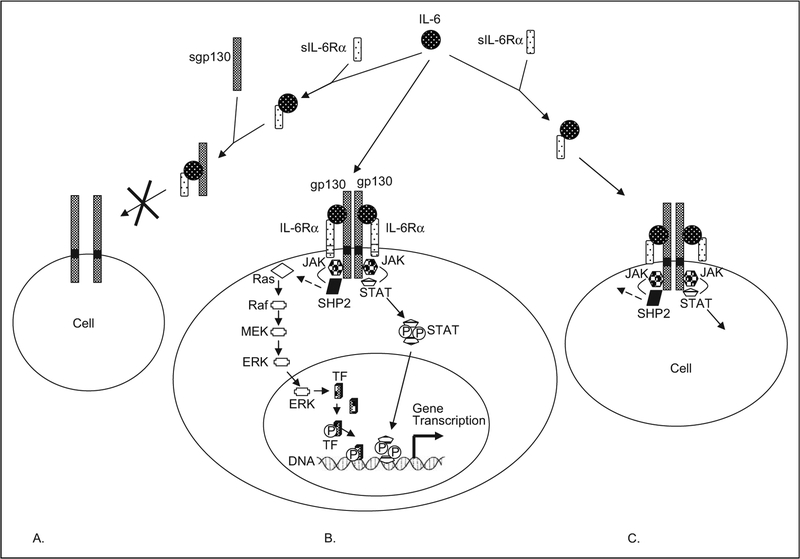
Figure 2.
A schematic of IL-6 and IL-6 receptor interactions. A. Soluble gp130, an antagonist of IL-6 activity. Serum IL-6 binds with sIL-6Rα. This complex in turn binds to sgp130. Soluble gp130 thereby competitively antagonizes the binding of the IL-6-sIL-6Rα complex to membrane-bound gp130, and thereby inhibits IL-6 effects on this cell. B. Membrane-bound gp130 and membrane-bound IL-6Rα. Membrane-bound IL-6Rα is restricted in location and is found in only select cell types, whereas gp130 is ubiquitous. In this case, serum IL-6 complexes with membrane-bound IL-6Rα and then associates with membrane-bound gp130, to generate a signaling-capable hexameric complex. As classically described, signaling is mediated proximally by the Jak kinases and STAT proteins. Phosphorylation of STAT proteins by the Jak kinases and their subsequent translocation to the nucleus induces gene transcription. The Jak kinases, with the help of tyrosine phosphatase, SHP2, also activate a series of additional kinases (Ras, Raf, Mek, and ERK), which in turn eventually phosphorylate a transcription factor (TF) and thereby induce gene transcription. A third pathway, not illustrated involves PI3K (see text for details). C. Soluble IL-6Rα, an agonist for IL-6 activities. Soluble IL-6Rα binds to serum IL-6. This complex then engages membrane-bound gp130, which is found on most cells, to transduce signal in those cells that lack membrane-bound IL-6Rα. This phenomenon, termed “trans-signaling,” allows IL-6 to target multiple cell types. Adapted from Arthritis Res. 2002; 4(suppl 3): S233-S242,9 Biochem J. 2003; 374(pt 1):1–20,70 Crit Care Med. 2005; 33(8):1839–1844,71 and Acta Biochim Pol. 2003; 50(3):603–611.72 IL indicates interleukin; gp, glycoprotein; sIL-6Rα, soluble IL-6Rα STAT, signal transducer and activator of transcription.