Abstract
Bovine herpes virus 1 (BoHV-1), an important bovine pathogen, causes conjunctivitis and disorders in the upper respiratory tract. Following acute infection, BoHV1 establishes life-long latency in sensory neurons. Recent studies demonstrated that viral gene products expressed in trigeminal ganglionic neurons during latency stabilize β-catenin levels, an important signaling molecule that interacts with a family of DNA binding proteins (T-cell factors) and subsequently stimulates transcription. In this study, we provide new evidence demonstrating that BoHV-1 transiently increased β-catenin protein levels in bovine kidney (CRIB) cells, but not in rabbit skin cells. β-catenin dependent transcription was also stimulated by infection of CRIB cells. The β-catenin small molecule inhibitor (iCRT14) significantly reduced the levels of BoHV-1 virus during productive infection of CRIB cells and rabbit skin cells. In summary, these studies suggested the ability of β-catenin to stimulate cell survival and cell cycle regulatory factors enhances productive infection in non-neuronal cells.
Keywords: bovine herpesvirus 1 (BoHV-1), productive infection, β-catenin, β-catenin inhibitor (iCRT14)
INTRODUCTION
Bovine herpesvirus 1 (BoHV-1), a widespread bovine pathogen, is an important cofactor for the most important disease in cattle, bovine respiratory disease complex (BRDC), reviewed in (1–3). BRDC is initiated by stress and/or viral infection(s) resulting in immune-suppression and life-threatening pneumonia. Mannheimia haemolytica, a commensal bacterium in the upper respiratory tract of cattle, is the cause of pneumonia in most BRDC cases. Bovine herpesvirus 1 (BoHV-1) infection can be a significant risk factor for BRDC because it causes clinical disease in the upper respiratory tract (4, 5), suppresses immune responses (2, 3), and cooperates with Mannheimia haemolytica to induce pneumonia in calves (6). Like many Alphaherpesvirinae subfamily members, BoHV-1 impairs immune responses during acute infection, which enhances replication (2). Following acute infection of mucosal linings in the oral, nasal or ocular cavity, BoHV-1 establishes latency in sensory neurons that reside within trigeminal ganglia, reviewed in (1–3). The ability to establish, maintain, and reactivate from latency is crucial for virus transmission.
We recently discovered that BoHV-1 regulates the Wnt/β-catenin signaling pathway in latently infected sensory neurons (7). These studies suggested that the ability of BoHV-1 to regulate the β-catenin signaling pathway was important during latency because this pathway stimulates axonal growth and navigation of axons to their synaptic targets, reviewed in (8–12). Wnt family members are secreted glycoproteins that interact with Frizzled receptors and a co-receptor LRP5/LRP6, reviewed in (13). Upon Wnt binding, the transcription factor β-catenin, is stabilized and enters the nucleus where it interacts with TCF (T-cell factor) family members specifically bound to a consensus DNA motif (5’-T/A-T/A-CAAAG-3’), reviewed in (13, 14). Binding of β-catenin to TCF displaces bound co-repressors and recruits co-activators to activate Wnt target genes (15). The Wnt/β-catenin signaling pathway is required for embryogenesis and adult homeostasis, reviewed in (13, 16).
In this study, we demonstrate that β-catenin steady state protein levels and β-catenin dependent transcription were increased following infection of bovine kidney (CRIB) cells. A specific inhibitor of the Wnt/β-catenin signaling pathway (iCRT14) reduced the plaque forming efficiency in CRIB and rabbit skin cells. These observations suggest that β-catenin has the potential to stimulate productive infection.
RESULTS
Analysis of β-catenin protein expression during productive infection of bovine kidney or rabbit skin cells
To understand whether the β-catenin signaling pathway influences productive infection, we initially examined β-catenin protein levels following infection of cultured cells. For these studies, bovine kidney (CRIB) and rabbit skin (RS) cells were infected with BoHV-1 because both cell lines support BoHV-1 replication. Relative to uninfected cells, β-catenin levels increased at four and eight hours after CRIB cells were infected but hen decreased to levels that were similar to mock-infected cells (Figure 1A). In contrast,β-catenin protein levels in RS cells were similar in mock-infected cells and four hours after infection (Figure 1B). At eight, 16, and 24 hours after infection, β-catenin levels were consistently reduced relative to mock-infected RS cells or cells infected for four hours. bICP0 protein expression was readily detected in CRIB, but not RS cells, at 4 hours after infection. In general, bICP0 expression was lower in RS cells compared to CRIB cells. Although β-catenin protein levels were transiently induced in CRIB cells, they were reduced at 16 and 24 hours after infection in CRIB and RS cells, which was likely a result of the virus host shutoff activity.
Fig 1. Expression of β-catenin in cultured bovine kidney and rabbit skin cells during productive infection.
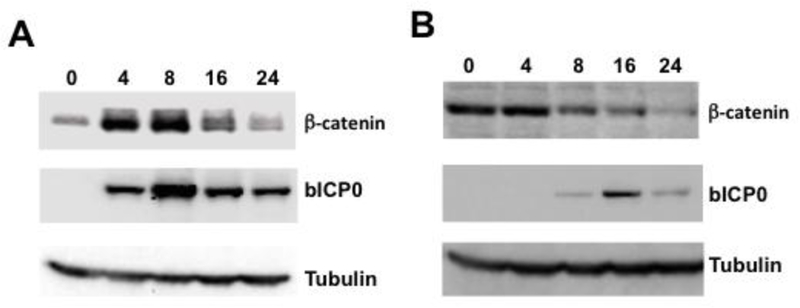
Bovine kidney (CRIB; Panel A) or rabbit skin (RS; Panel B) cells were infected with BoHV-1 (1 pfu/cell) for the designated times (hours after infection). Mock-infected cells were denoted as 0. At the indicated times after infection, total cell lysate was prepared and Western blots performed using specific antibodies that recognize the denoted proteins. For each lane, 60 ug protein was loaded. These results are representative of five independent experiments.
Productive infection stimulates β-catenin dependent transcription
To test whether infection regulated β-catenin dependent transcription, CRIB cells were transfected with Super 8x TOPFlash and then promoter activity measured after infection. When β-catenin is localized to the nucleus, it can interact with a TCF (T-cell factor) family member bound to a consensus site (5’-T/A-T/A-CAAAG-3’), reviewed in (13, 17). β-catenin binding to TCF displaces bound corepressors and recruits transcriptional coactivators, thus stimulating transcription of promoters containing TCF binding sites (14). The plasmid Super 8x TOPFlash contains 8 TCF binding sites upstream of a minimal promoter that drives firefly luciferase reporter expression and thus accurately measures β-catenin dependent transcription. β-catenin dependent transcription increased as a function of the moi used for infection at 8 hours after infection (Figure 2A).
Fig 2. BoHV-1 infection of CRIB cells stimulated β-catenin dependent transcription.

CRIB cells in 12-wells plates (60% confluent) were transfected with 0.4 µg of the Super 8x TOPFlash luciferase reporter construct and 0.05 μg of the Renilla reporter construct that was used as an internal control to allow normalization of promoter activity. Panel A: At 48 hours after transfection, cells were infected with BoHV-1 at the designated MOI. At 12 hours after infection, dual luciferase activity was measured. Panel B: CRIB cells that were transfected as described in Panel A were infected with BoHV-1 at an MOI of 10. Certain cultures were treated iCRT14 at the designated concentrations. Dual luciferase assays were performed at 12 hours after infection. Mock-infected cells were normalized to a value of 1 and the fold activation for other samples calculated. The results are the average of three independent experiments and error bars denote standard error. An asterisk denotes significant differences (P < 0.05) in promoter activity compared to the indicated control by the one-way ANOVA and Fisher’s LSD multiple means comparison tests.
To test whether the effect on Super 8x TOPFlash was due to β-catenin dependent transcription or merely the effect of viral infection stimulating transcription, we analyzed promoter activity in the presence of a β-catenin specific inhibitor (iCRT14). This small molecule was chosen because it specifically interferes with interactions between β-catenin and TCF family members (18). Super 8x TOPFlash promoter activity was similar to uninfected cells when infected cultures were treated with 10 uM iCRT14 (Figure 2B). 1uM iCRT14 also reduced β-catenin dependent transcription but not significantly different compared to infected cells. In summary, these studies revealed that BoHV-1 infection stimulated β-catenin dependent transcription in CRIB cells at 8 hours after infection.
The β-catenin inhibitor, iCRT14, inhibits BoHV-1 productive infection
The effect that the β-catenin signaling pathway has on productive infection was examined using the small molecule inhibitor iCRT14. Initial studies tested whether iCRT14 was toxic for CRIB cells. Ten uM iCRT14 had no obvious toxic effects on CRIB cells, as judged by measuring intracellular levels of ATP (Figure 3A). At higher iCRT14 concentrations (25, 50, and 100 uM), toxicity increased in a dose dependent manner. Furthermore, 10 uM iCRT4 had little effect on steady state levels of β-catenin protein levels in CRIB cells (data not shown). We then examined the effect of increasing concentrations of iCRT14 on BoHV-1 replication in CRIB cells. Relative to the DMSO control, 1 uM iCRT14 had no obvious effect on virus replication, conversely 10 or 25 uM iCRT14 reduced the levels of infectious virus by approximately 3 logs (Figure 3B). When cell morphology was examined, 10 uM iCRT14 reduced the effect of virus induced cytopathology (Figure 3C). Additional studies were performed to examine the effects that ten uM iCRT14 had on virus infection at different times after infection. Ten uM iCRT14 consistently inhibited virus production in CRIB cells by 2–3 logs at 16 or 24 hours after infection (Figure 3D).
Fig 3. The β-catenin inhibitor iCRT14 reduced BoHV-1 replication in Bovine kidney (CRIB) cells.

Panel A: The designated concentrations of iCRT14 were incubated with CRIB or RS cells for 24 hours. ATP levels in the respective cultures were measured to assess cell viability using CellTiter-Glo Luminescent Cell Viability Assay (Promega; G7572), according to the manufacturer’s instruction. Panel B: Confluent CRIB cells in 60 mm dishes were pretreated with the designated concentration of iCRT14 at two hours prior to infection. Cultures were infected with BoHV-1 at MOI of 1 for 1 h in the presence of iCRT14 or just DMSO. After extensive washing with PBS, fresh medium containing inhibitors or DMSO control were replaced. At 24 h post infection, the amount of total virus in the culture was measured by plaque assay. These studies were repeated 3 times and asterisks denote significant differences between the DMSO control and cultures treated with iCRT14 (**: P < 0.005) as determined by the Student t test. Panel C: Before collecting the virus in the study described in Panel B, cells were photographed to compare the effect of viral induced cytotoxicity to uninfected cells (magnification is 40x). Panel D: As in panel B, CRIB cells were pretreated with 10 uM iCRT14 two hours prior to infection. Cultures were then infected with BoHV-1 at an MOI of 1 for 1 h in the presence of iCRT14 or just DMSO. After extensive washing with PBS, fresh medium containing inhibitors or DMSO control were replaced. At 8, 16, or 24 h post infection, the amount of total virus in the culture was measured by plaque assay. These studies were repeated 3 times and an asterisk denotes significant differences between the DMSO control and cultures treated with iCRT14 (*: P < 0.05 and **: P < 0.005) as determined by the Student t test.
The effect of iCRT14 on BoHV-1 infection was then examined in RS cells. In contrast to CRIB cells, 10 uM iCRT14 exhibited low levels of toxicity in RS cells 24 hours after treatment, however 2.5 uM iCRT14 was not toxic (Figure 3A). Consequently, studies were performed using 2.5 uM iCRT14. BoHV-1 replication was inhibited approximately 2 logs at 24 hours after infection when treated with 2.5 uM iCRT14 (Figure 4). In summary, the β-catenin small molecule inhibitor, iCRT14, significantly reduced BoHV-1 replication in two permissive cell lines, CRIB and RS cells.
Fig 4. The β-catenin inhibitor iCRT14 reduced BoHV-1 replication in rabbit skin cells.

RS cells were incubated in 10% fetal calf serum and then sub-cultured 24 hours prior to infection. Two hours prior to infection, RS cells were pretreated with the designated concentration of iCRT14 and then infected with BoHV-1 at an MOI of 1. Cells were treated with iCRT14 throughout infection. Infectious virus was collected at 24 hours after infection by three rounds of freeze-thawing and then plaque assays performed. These studies were repeated 3 times and an asterisk denotes significant differences between the DMSO control and cultures treated with iCRT14 (P < 0.05) as determined by the Student t test.
DISCUSSION
In this study, we provide evidence that a β-catenin specific inhibitor reduced virus replication at least 100 fold. CRIB cells, but not RS cells, contained higher β-catenin protein levels during early stages of productive infection suggesting viral infection induced cell-type or species-specific effects. Regardless, inhibiting interactions between β-catenin and TCF family members with iCRT14 significantly reduced BoHV-1 replication in CRIB and RS cells. An active Wnt/β-catenin signaling pathway also stimulates human cytomegalovirus and herpes simplex virus 1 (HSV-1) replication (19, 20) suggesting β-catenin provides important functions that can directly or indirectly stimulate replication of more than one herpesvirus.
Our previous studies demonstrated that BoHV-1 latent gene products stabilize β-catenin expression during latency suggesting the β-catenin pathway plays a role during the establishment and/or maintenance of latency (7), which seems at odds with the results from this study. However, the Wnt/β-catenin signaling pathway has tissue and cell-type specific functions that may enhance latency in one cell type but stimulate productive infection in other cell types. For example the Wnt/β-catenin signaling pathway in neurons maintains differentiation specific functions of neurons by stimulating axon growth and targeting axons to specific targets (8–12), as well as inhibiting neuro-degeneration (21–24). In non-neuronal cells, the β-catenin signaling pathway stimulates cell growth and inhibits cell death, as exemplified by the fact that β-catenin enhances oncogenesis (13, 16, 25).
The core Wnt/β-catenin response element (5’-T/A-T/A-CAAAG-3’) (26), which includes a TCF binding site, is only present in the BoHV-1 genome twice (data not shown). Thus, β-catenin does not appear to stimulate productive infection by directly trans-activating many, if any viral promoters. Consequently, we suggest β-catenin promotes productive infection by enhancing expression of factors that mediate certain aspects of cell proliferation and cell survival. For example, β-catenin trans-activates the cyclin D1 promoter (27) and it is generally accepted that cyclin D1, Rb, and E2F cooperate to promote G1 cell cycle progression, reviewed by (28, 29). These observations correlate with the finding that BoHV-1 replicates more efficiently in actively dividing cells, as demonstrated by the finding that silencing E2F1 or E2F2 inhibits BoHV-1 replication and E2F1 trans-activates the bICP0 early promoter (30, 31).
In conclusion, BoHV-1 productive infection is stimulated by the ability of β-catenin to maintain the growth potential of non-neuronal cells. In contrast, neuronal-specific functions of the Wnt/β-catenin signaling pathway are proposed to cooperate with BoHV-1 latent gene products to establish and maintain latency in sensory neurons.
MATERIALS AND METHODS
Cells and cell viability assay
Bovine kidney cells (CRIB) and rabbit skin (RS) cells were grown in Eagle’s minimal essential medium (EMEM) supplemented with 10% FCS, penicillin (10 U/ml), and streptomycin (100 ug/ml).
CRIB or RS cells were seeded at 3.5 ×106 cells/ml in 24-well plates and then incubated overnight at 37o C. Cells were treated with various concentrations of iCRT14 (Sigma-Aldrich) for 24 hours. To assess cell viability, ATP levels in cultures were then measured using CellTiter-Glo Luminescent Cell Viability Assay (Promega; G7572) according to the manufacturer’s instruction.
Viruses & plasmids
The Cooper strain of BoHV-1 (wt virus) was obtained from the National Veterinary Services Laboratory, Animal and Plant Health Inspection Services, Ames, IA. BoHV-1 stocks were prepared in CRIB cells.
Super 8x TOPFlash contains a simple promoter that is stimulated by β-catenin and was a gift from Randall Moon (Addgene plasmid # 12456) (32).
Western blot analysis
Cells were lysed in RIPA buffer (50 mM Tris-HCl, pH 8, 150 mM NaCl, 1% Triton X-100, 0.5% sodium deoxycholate, 0.1% SDS) with protease and phosphatase inhibitors (Thermo-Scientific). The respective samples were boiled in Laemmli sample buffer for 5 minutes and all samples were separated on an 10% SDS–polyacrylamide gel. Western blots were performed as previously described (7). The anti-β-catenin antibody (Abcam, ab32572) or a peptide specific bICP0 antibody was used for Western blot studies.
Dual-luciferase reporter assay
CRIB cells were cotransfected with the Super 8x TOPFlash plasmid and a plasmid encoding Renilla luciferase under the control of a minimal herpesvirus promoter (Promega) using Lipofectamine® 2000 Transfection Reagent (11668019, Invitrogen). At the designated times after infection, cells were harvested and protein extracts were subjected to a dual-luciferase assay using a commercially available kit (E1910; Promega) according to the manufacturer’s instructions. Luminescence was measured with a GloMax 20/20 luminometer (E5331; Promega).
Highlights.
BoHV-1 transiently increased β-catenin protein levels in bovine kidney cells
β -catenin dependent transcription was stimulated by BoHV-1 infection
A β -catenin inhibitor (iCRT14) reduced BoHV-1 productive infection
ACKNOWLEDGEMENTS
This research was supported by a grant from the USDA-NIFA Competitive Grants Program (13–01041), funds derived from the Sitlington Endowment, and support from the Oklahoma Center for Respiratory and Infectious Diseases (National Institutes of Health Centers for Biomedical Research Excellence Grant # P20GM103648). L.Z. was partially supported by the China Scholarship Council, Chinese National Science Foundation Grant (No. 31472172) and National Key Research Program (No. 2016YFD0500704).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Turin L, Russo S, and Poli G. 1999. BHV-1: new molecular approaches to control a common and widespread infection. Molec Med 5:261–284. [PMC free article] [PubMed] [Google Scholar]
- 2.Jones C 2009. Regulation of innate immune responses by bovine herpesvirus 1 and infected cell protein 0. Viruses 1:255–275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jones C and Chowdhury S. 2007. A review of the biology of bovine herpesvirus type 1 (BHV-1), its role as a cofactor in the bovine respiratory disease complex, and development of improved vaccines.. Adv in Anim Health 8:187–205. [DOI] [PubMed] [Google Scholar]
- 4.Hodgson PD, Aich P, Manuja A, Hokamp K, Roche FM, Brinkman FSL, Potter A, Babiuk LA, and Griebel PJ. 2005. Effect of stress on viral-bacterial synergy in bovine respiratoryt disease: novel mechanisms to regulate inflammation. Comparative and Functional Genomics 6:244–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jones C and Chowdhury S. 2010. Bovine herpesvirus type 1 (BHV-1) is an important cofactor in the bovine respiratory disease complex., p 303–321. In Broderson VLCaB (ed), Veterinary Clinics of North America, Food Animal Practice, Bovine Respiratory Disease, vol 26 Elsevier, New York, NY. [DOI] [PubMed] [Google Scholar]
- 6.Yates WD, Babiuk LA, and Jericho KW. 1983. Viral-bacterial pneumonia in calves: duration of the interaction between bovine herpesvirus 1 and Pasteurella haemolytica. Can J Comp Med 47:257–264. [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Y, Hancock M, Workman A, Doster A, and Jones C 2016. Beta-catenin, a transcription factor activated by canonical Wnt signaling, is expressed in sensory neurons of calves latently infected with bovine herpesvirus 1. J of Virol 90:3148–3159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Salinas PC. 2012. Wnt signaling in the vertebrate central nervous system: from axon guidance to synaptic function. Cold Spring Harb Perpect Biol 4:a008003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Purro SA, Galli S, and Salinas PC. Dysfunction of Wnt signaling and synaptic disassembly in neurodegenerative diseases. J of Molec Cell Biol 6:75–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bhardwaji D, Nager M, Camats J, David M, Benguira A, dopazo A, Canti C, and Herreros J. 2013. Chemokines induce axon outgrowth downstream of hepatocyte growth factor and TCF/beta-catenin signaling. Frontiers in Cellular Neuroscience 7:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Murase S, Mosser E, and Schuman EM. 2002. Depolarization drives beta-catenin into neuronal spines promoting changes in synaptic structure and function. Neuron 35:91–105. [DOI] [PubMed] [Google Scholar]
- 12.Bamji SX, Rico B, Kimes N, and Reichardt LF. 2006. BDNF mobilizes synaptic vesicles and enhances synaptic vesicles and enhances synapse formation by disrupting cadherin-beta-catenin interactions. J Cell Biol 174:289–299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Clevers H and Nusse R. 2012. Wnt/B-catenin signaling and disease. Cell 149:1192–1205. [DOI] [PubMed] [Google Scholar]
- 14.Bush BM, Brock AT, Deng JA, Nelson RA Jr, and Sumter TF. 2013. The Wnt/b-catenin/TCF pathway upregulates HMGA1 expression in colon cancer. Cell Biochem Funct 3:228–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Alves-Guerra MC, Ronchini C, and Capobianco AJ. 2007. Mastermind like 1 is a specific coactivator of beta-catenin transcription activation and is essential for colon carcinoma cell survival. Cancer Res 67:8690–8698. [DOI] [PubMed] [Google Scholar]
- 16.Polakis P 2012. Wnt signaling in cancer. In Nusse R, He X, and van Amerongen R (ed), Cold Spring Harb Perspect Biol Cold Spring Harbor Laboratory Press, [Google Scholar]
- 17.Biechele TL Kulikauskas RM, Toroni RA, Lucero OM, Swift RD, James RG, Robin NC, Dawson DW, Moon RT, and Chien AJ. 2012. Wnt/beta-catenin signaling and AXIN1 regulate apoptosis mediated by inhibition of BRAFV600E kinase in human melanoma. Science Signaling 5:206: ra203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gonsalves FC Klein K, Carson BC, Katz S, Ekas LA, Evans S, R. Nagourney, Cardozo T, Brown AMC, and DasGupta R 2011. An RNAi-based chemical genetic screen identifies threee small-molecular inhibitors of the Wnt/wingless signalling pathway. Proc Natl Acad Sci U S A 108:5954–5963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Choi E-J, Kim S, Jho E, Song K-J, and Kee S-H 2013. Axin expression enhances herpes simplex virus type 1 replication by inhibiting virus-mediated cell death in L929 cells. J Gen Virol 94:1636–1646. [DOI] [PubMed] [Google Scholar]
- 20.Kapoor A, He R, Venkatadri R, Forman M, and Arav-Boger R. 2013. Wnt modulating agents inhibit human cytomegalovirus replication. Antimicrobial Agents and Chemotherpay 57:2761–2767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chen S, Guttridge DC, You Z, Zhang Z, Fribley A, Mayo MW, Kitajewski J, and Wang C-Y. 2001. Wnt-1 signaling inhibits apoptosis by activating beta-catenin/T cell factor-mediated transcription. J of Cell Biology 152:97–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nestor T, Masckauchan H, Shawber CJ, Funahashi Y, Li C-M, and Kitajewski J 2005. Wnt/beta-catenin signaling induces proliferation, survival, and Interleukin-8 in human endothelial cells. Angiogenesis 8:43–51. [DOI] [PubMed] [Google Scholar]
- 23.Biechele TL, Kulikauskas RM, Toroni RA, Lucero OM, Swift RD, James RG, Robin NC, Dawson DW, Moon RT, and Chien AJ. 2012. Wnt/beta-catenin signaling and AXIN1 regulate apoptosis mediated by inhibition of BRAFV600E kinase in human melanoma. Sci Sig 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhao S, Fu J, Liu X, Wang T, Zhang J, and Zhao Y. 2012. Activation of Akt/GSK-3beta/beta-catenin signaling pathway is invovlved in survival of neurons after traumatic brain injury in rats. Neurological Res 34:400–407. [DOI] [PubMed] [Google Scholar]
- 25.Hayward P, Kalmar T, and Arlas AM. 2008. Wnt/Notch signalling and information processing during development. Development 135:411–424. [DOI] [PubMed] [Google Scholar]
- 26.Bottomly D, Kyler SL, McWeeney SK, and Yochum GS. 2010. Identification of b-catenin binding regions in colon cancer cells using ChIP-Seq. Nucleic Acids Res 38:5735–5745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shtutman M, Zhurinsky J, Simcha I, Albanese C, D’Amico M, Pestell R, and Ben-Zee’Ev A. 1999. The cyclin D1 gene is a target of the b-catenin/LEF-1 pathway. Proc Natl Acad Sci U S A 96:5522–5527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Goodrich DW, Wang NP, Qian YW, Lee EY, and Lee WH. 1991. The retinoblastoma gene product regulates progression through the G1 phase of the cell cycle. Cell 67:293–302. [DOI] [PubMed] [Google Scholar]
- 29.Harbour JW and Dean DC. 2000. The Rb/E2F pathway: expanding roles and emerging paradigms. Genes Dev 14:2393–2409. [DOI] [PubMed] [Google Scholar]
- 30.Workman A and Jones C. 2010. Bovine herpesvirus 1 productive infection and bICP0 early promoter activity are stimulated by E2F1. J Virol 84:6308–6317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Workman A and Jones C. 2011. Analysis of the cell cycle regulatory protein (E2F1) after infection of cultured cells with bovine herpesvirus 1 (BHV-1) or herpes simplex virus type 1 (HSV-1). Virus Res 160:66–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Veeman MT, Slusarski DC, Kaykas A, Louie SH, and Moon RT. 2003. Zebrafish prickle, a modulator of noncanonical Wnt/Fz signaling, regulates gastrulation movements. Curr Biol 13:680–685. [DOI] [PubMed] [Google Scholar]


