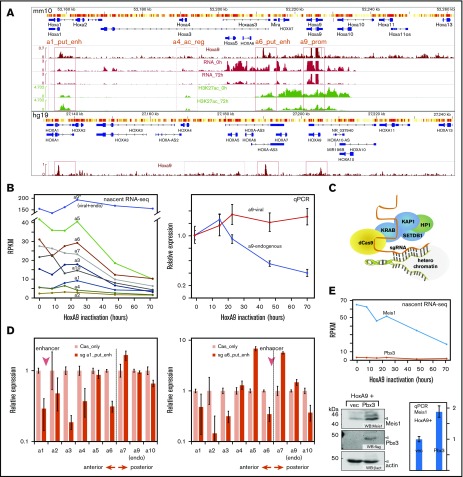
Figure 5.
HoxA9 controls itself and the Hox-A locus. (A) Overview of the Hox-A locus in mouse and human genomes with conserved Hox/HOXA9 binding sites and associated nascent RNA and H3K27ac profiles. Potential HoxA9-dependent regulatory regions are boxed and labeled as follows: a1_put_enh = HoxA1 putative enhancer, a4_ac_reg = HoxA4 acetylated region, a6_put_enh = HoxA6 putative enhancer, a9_prom = HoxA9 promoter. For a detailed explanation, see the text and supplemental Figure 5. (B) Hox-A transcription is under control of HoxA9. Kinetics of nascent RNA Hox production after cessation of HoxA9 activity (left panel). RPKM values are shown. Differential qPCR for endogenous and viral-derived HoxA9 RNA (right panel). Expression was normalized to β-actin as a housekeeping gene, and it is plotted as relative value with 0 hours set to 1 unit. (C) Principle of epigenome editing by dCas9-KRAB. Targeting of a catalytically inactive Cas9-KRAB fusion to a specific locus by an sgRNA creates local heterochromatin through recruitment of a repressor complex.20 (D) Epigenome editing of HoxA1/A6 putative enhancers affects expression of anterior Hox-A genes. Expression levels of all Hox-A genes were recorded by qPCR in cells expressing sgRNAs targeting the HoxA1/A6 putative enhancers and a dCas-KRAB fusion or dCas-KRAB alone, as a control, as indicated. Relative expression normalized to β-actin is shown, with control expression set to 1 unit. (E) Pbx3 limits Meis1 protein availability. Nascent RNA production of Meis1 and Pbx3 RNAs in response to HoxA9 (upper panel). Western blot analysis of HoxA9-transformed cells virally coexpressing Pbx3 or vector only as control (lower left panel). qPCR quantification of Meis1 RNA in the same cells (lower right panel). HP1, heterochromatin protein 1; KAP1, KRAB-associated protein 1; KRAB, Krüppel-associated box; SETDB1, H3K9 methyltransferase.