Introduction
Congenital plasminogen deficiency (OMIM # 217090) is a genetic disease with a recessive inheritance pattern. The formation of ligneous pseudomembranes owing to the accumulation of nondegraded fibrin is the most frequent cutaneous manifestation.1 This disease usually appears in childhood, often in a severe manner, leading to complications such as blindness, hydrocephalus, respiratory failure caused by bronchial obstruction, or even death.2 Patients who reach adulthood usually have a history of pneumonia, cervicitis, conjunctivitis, hoarseness, or sterility.3 We report on a 60-year-old woman with conjunctival and vulvar pseudomembranous lesions finally diagnosed as congenital plasminogen deficiency.
Case report
A 60-year-old woman was evaluated in our dermatology department for oral and genital mucosal lesions. The patient reported several admissions for pneumonia and recurrent cervicitis. She has 2 siblings, and her sister, who died in childhood from pneumonia, presented a similar phenotype, with mucosal lesions, and had Cowden syndrome clinically diagnosed.
Physical examination found multiple asymptomatic palmar and plantar pits and a gingival nonbleeding verrucous hypertrophy with papillomatous formations that broke off when shaved. The absence of numerous teeth was observed. Ophthalmologic examination found a large, yellowish-white pedunculated pseudomembrane in the superior tarsal conjunctiva (Fig 1). Vulvar exploration found the presence of similar asymptomatic white-yellowish membranes in the vaginal introitus (Fig 2). Histologic study of the conjunctival and genital lesions showed hyaline-positive periodic acid–Schiff material in the stroma (Fig 3). Direct immunofluorescence showed multiple deposits of fibrinogen in the dermis (Fig 4). Plasma plasminogen activity showed a 47% value (reference values, 75%-150%). Genetic study by complete Sanger sequencing of the PLG gene, found the homozygotic mutation in c.2377T>A (p.Tyr793Asn), not previously described and classified as deleterious, which confirmed diagnosis of congenital deficiency of plasminogen type 1.
Fig 1.
Large pedunculated white-yellowish pseudomembrane in upper tarsal conjunctiva.
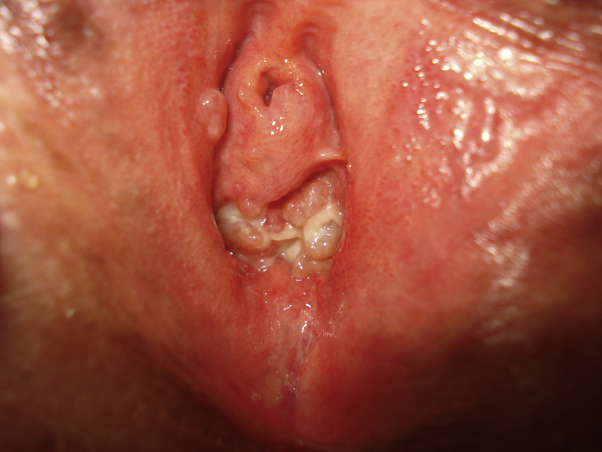
Fig 2.
White-yellowish pseudomembranes in vaginal introitus.
Fig 3.
Conjunctival pseudomembrane biopsy shows a periodic acid–Schiff–positive deposit in the dermis. (Original magnification: ×20.)
Fig 4.
Direct immunofluorescence of conjunctival pseudomembrane with antifibrinogen antibodies where deposits of fibrinogen are observed in the dermis.
The homozygosis mutation in the patient suggested a consanguinity in the family, which was confirmed after the pedigree elaboration with 5 generations. Genetic analysis was performed in her alive brother, who presented the mutation in heterozygosis. He also presented a decreased plasma plasminogen activity (46%) and was considered positive for congenital plasminogen deficiency, postulating the possibility of a codominance mutation. However, he was asymptomatic and lost to follow-up. His daughter was a heterozygous carrier without symptoms. Regarding the deceased sister with Cowden disease, we postulate she presented the same genotype than the index case, but it was incorrectly diagnosed.
The patient received treatment with stanozolol and danazol without significant improvement. She is in multidisciplinary follow-up by the dermatology, pneumology, hematology, surgery, and gynecology departments.
Discussion
Plasminogen deficiency is a rare entity that affects 1.6 of every 1 million individuals and is inherited in an autosomal recessive manner by mutations in homozygosis or compound heterozygosis in the PLG gene, which codes for plasminogen. Despite this inheritance pattern, it is estimated that 0.13% to 0.42% of the population are asymptomatic heterozygous individuals.4
Plasminogen is the proenzyme of plasmin and is synthesized primarily in the liver. Although its role in intravascular and extravascular fibrinolysis is well defined, it also plays an important role as a proteolytic enzyme, degrading the proteins of the extracellular matrix, activating metalloproteases, and repairing wounds. Its deficit leads to an inability to lysis of fibrin, producing an accumulation of this protein, which is the cause of the clinically observed manifestations.5
Two types of plasminogen deficiency have been described: type 1 (hypoplasminogenemia), in which plasminogen levels and activity are reduced, and type 2 (dysplasminogenemia), in which plasminogen levels are normal but their specific activity is reduced. The clinical symptoms of type 1 plasminogen deficiency are derived by the accumulation of fibrinoid material in different systems of the organism, giving rise to mucocutaneous, otorhinolaryngologic, respiratory, or gynecologic manifestations. The most frequent manifestation is the formation of pseudomembranes of woody consistency in the ocular conjunctiva and less frequently in the upper and lower respiratory tract, vagina, and gastrointestinal tract. Plasminogen deficiency has also been associated with aggressive periodontitis and gingival hyperplasia caused by the accumulation of this material in the oral cavity, leading to loss of teeth.4
The differential diagnosis of this pathology is essential, and this clinical entity should be considered with respect to other diseases that occur with deposit of foreign material such as amyloidosis or lipoid proteinosis as well as other dermatoses that cause gingival hyperplasia, especially Cowden disease. The use of tranexamic acid, an antifibrinolytic agent, as a causative agent of pseudomembranous disease6 should also be ruled out. The onset during childhood, conjunctival involvement, probable history of respiratory infections and/or recurrent cervicitis, and the presence of stromal fibrin deposits in direct immunofluorescence are keys to suspecting congenital deficiency of plasminogen. Finally, it must be confirmed by the diminished plasminogen activity in plasma and the genetic study of the PLG gene by complete sequencing (next-generation sequencing or Sanger), for the detection and description of the genetic alterations responsible for the appearance of this disease in each patient and their family in the face of subsequent genetic counseling.
Palmar and plantar pits observed in our patient are not described in this condition. Although they are frequently associated with Gorlin syndrome, they have been described in patients with rare conditions, such as Darier disease, generalized follicular hamartoma, or reticulate acropigmentation of Kitamura. Plasminogen deficiency could be another entity that we should include in the differential diagnosis in case of palmar and plantar pits.
Treatment of these patients is challenging. Currently, there is no effective treatment. Multiple topical and systemic treatments have been used. Surgical removal of lesions is indicated when they present local problems but may be a trigger for new lesions. Therapeutic trial with an anabolic steroid like stanozolol or danazol can normalize plasminogen levels.7 The use of fresh plasma transfusions has been described with good results, especially in combination with systemic corticosteroids.2 The use of substitution therapy with humanized plasminogen may represent a new therapeutic modality in these patients, having shown encouraging results in both pediatric and adult patients.8
Footnotes
Drs Magdaleno-Tapial and Pablo Hernández-Bel contributed equally to this article.
Funding sources: None.
Conflicts of interest: None disclosed.
References
- 1.Bhave S.S., Joshi S.S. Ligneous conjunctivitis in a patient of juvenile colloid milia: a rare association. Indian J Ophthalmol. 2015;63(4):350–352. doi: 10.4103/0301-4738.158091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shapiro A.D., Nakar C., Parker J.M. Plasminogen replacement therapy for the treatment of children and adults with congenital plasminogen deficiency. Blood. 2018;131(12):1301–1310. doi: 10.1182/blood-2017-09-806729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baithun M., Freeman-Wang T., Chowdary P., Kadir R.A. Ligneous cervicitis and endometritis: a gynaecological presentation of congenital plasminogen deficiency. Haemophilia. 2018;24(3):359–365. doi: 10.1111/hae.13418. [DOI] [PubMed] [Google Scholar]
- 4.Neering SH, Adyani-Fard S, Klocke A, Rüttermann S, Flemmig TF, Beikler T. Periodontitis associated with plasminogen deficiency: a case report. BMC Oral Health [Internet]. 2015 May 14 [cited 2018 May 1];15. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4438564/ [DOI] [PMC free article] [PubMed]
- 5.Stoopler E.T., Alawi F. Gingival swelling associated with hypoplasminogenemia. Rev Bras Hematol E Hemoter. 2016;38(3):274–275. doi: 10.1016/j.bjhh.2016.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Song Y., Izumi N., Potts L.B., Yoshida A. Tranexamic acid-induced ligneous conjunctivitis with renal failure showed reversible hypoplasminogenaemia. BMJ Case Rep. 2014:2014. doi: 10.1136/bcr-2014-204138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mannucci P.M., Kluft C., Traas D.W., Seveso P., D'Angelo A. Congenital plasminogen deficiency associated with venous thromboembolism: therapeutic trial with stanozolol. Br J Haematol. 1986;63(4):753–759. doi: 10.1111/j.1365-2141.1986.tb07559.x. [DOI] [PubMed] [Google Scholar]
- 8.Aslan D., Akata R.F. The search for new approaches to treating type 1 plasminogen deficiency. Pediatr Blood Cancer. 2018;65(4) doi: 10.1002/pbc.26922. [DOI] [PubMed] [Google Scholar]