Abstract
Inflammatory response in Chagas disease is related to parasite and host factors. However, immune system regulation has not been fully elucidated. Thus, this study is aimed at evaluating IL-4 influence on acute phase of Trypanosoma cruzi experimental infection through dosage of cytokine levels in cardiac homogenate of infected Balb/c WT and Balb/c IL-4−/− as well as its histopathological repercussions. For such purpose, mice were divided into two groups: an infected group with 100 forms of the Colombian strain and an uninfected group. After 21 days of infection, animals were euthanized and the blood, spleen, and heart were collected. The spleen was used to culture splenic cells in 48 h. Subsequently, cytokines TNF-α, IL-12p70, IL-10, IFN-γ, and IL-17 were measured in the blood, culture supernatant, and heart apex by ELISA. The base of the heart was used for histopathological analysis. From these analysis, infected Balb/c IL-4−/− mice showed milder inflammatory infiltrate compared to Balb/c WT, but without changes in nest density and collagen deposition. IL-4 absence culminated in lower cardiac tissue IFN-γ production, although it did not affect TNF-α expression in situ. It also decreased TNF-α systemic production and increased IL-10, both systemically and in situ. In addition, IL-4 absence did not influence IL-17 expression. Splenocytes of IL-4-deficient mice produced higher amounts of IFN-γ, TNF-α, and IL-17 and lower amounts of IL-10. Thus, IL-4 absence in acute phase of experimental infection with T. cruzi Colombian strain reduces myocarditis due to lower IFN-γ production and greater IL-10 production in situ and this pattern is not influenced by splenocyte general repertoire.
1. Introduction
Chagas disease, also known as American trypanosomiasis, is a chronic systemic infectious disease caused by the protozoan Trypanosoma cruzi (T. cruzi), an important public health problem in American continent. According to the World Health Organization (WHO), the disease affects around 7.5 million people worldwide, the majority in Latin America [1, 2].
Murine experimental model reproduces human Chagas' disease infections, and genetic background has influences both, triggering a series of reactions that evolve from an acute symptomatic phase to chronic phase [3]. Some variables interfere in Chagas' disease pathogenicity such as the infecting strain and the mouse lineage. Balb/c strain mice are known to be susceptible to T. cruzi infection, present a high parasitemia and the vast majority succumb early to acute infection [4]. Colombian strain has been used in several studies with the purpose of evaluating cardiac function due to its evident myotropism [5]. Prominent myocardial lesions with intense inflammatory process, evolution in parasitemia with peak between 21 and 25 days of postinfection, distinct myotropism with involvement of the skeletal musculature resulting in extensive lesions in the skeletal muscle fibers, electrocardiographic alterations, and resistance to chemotherapeutics such as benznidazole and nifurtimox are characteristics of this strain. [6, 7].
Knockout model is widely used in studies that evaluate direct or indirect implication of certain cytokines in resistance to T. cruzi infection. IL-10 KO mice infected with T. cruzi Y strain had lower serum and tissue parasite load and higher IFN-gamma and nitric oxide production by spleen cells than wild-type mice [8]. Furthermore, IL-10 KO mice infected with the Tulahuen strain, in addition to lower serum parasite load, also showed higher levels of serum TNF-α, IL-12, and IFN-gamma compared with infected IL-10+/+ mice [9]. Knockout models for other molecules as B2-microglobulin [10], CD4 or CD8 [11], gamma delta T cells [12], and Fas ligand [13] were also used in studies on Chagas disease.
In acute phase, Chagas disease has variable clinical presentations, from asymptomatic to symptomatic and even fatal in some cases. Parasite proliferation and dissemination by blood or lymphatic vessels characterized this phase, with a parasitemia peak that regresses in some days and tends to be undetectable in chronic phase. Acute phase dissemination affects several cellular types, especially cardiac muscle fibers, with amastigotes nest formation [14], myocarditis, and subsequent collagen deposition between cardiac fibers, causing progressive functional damage. Excessive immune system activation with proinflammatory cytokine production of Th1-type IL-12, TNF-α, and IFN-γ characterizes this phase. On the other hand, cytokines such as IL-10 and TGF-β are activated to modulate immune response with consequent reduction of tissue damage [15, 16].
IL-4 is described as an anti-inflammatory cytokine, prototypic of Th2 profile, and related to susceptibility to protozoa infection, such as cutaneous leishmaniasis [17, 18], although even in models of polarized clinical manifestations, its relation with Th1 response still raises major controversies [19]. IL-4 reduces IFN-γ production [20] controlling Th1 response and preventing excessive tissue inflammation [21]. IL-4 knockout animals tend to have better response to infection by intracellular parasites, and when infected by T. cruzi, they increase Th1-type immune response, resulting in decreased parasitemia and mortality and increased cardiac inflammatory reaction, although these effects are observed in late stages of acute or in chronic phase [21, 22]. On the basis of in vitro testing, two contrasting roles for IL-4 have been described: enhancement of intracellular T. cruzi destruction by macrophages [23] and inhibition of IFN-γ-mediated trypanocidal activity [24].
Our group had already pointed out that infected and reinfected animals with Colombian strain present a modulation of immune response leading to higher production of proinflammatory cytokines, such as IFN-γ and TNF-α, resulting in marked myocarditis and lower survival rate [25]. As Balb/c mice are usually good IL-4 producers [26] and due to IL-4 relevance in modulating response against infectious and parasitic agents, including chagasic cardiomyopathy, IL-4−/− knockout animals were used to analyze if this cytokine has any influence on immune response modulation in mice infected with T. cruzi Colombian strain, which has marked myotropism in acute phase. We demonstrate that in early stages of acute phase, IL-4−/− knockout animals presented milder myocarditis with lower IFN-γ production and higher in situ IL-10 production, despite Th1 cells increase in splenocyte general repertoire. Our results open perspectives of IL-4 role in chagasic myocarditis initial events in a more complex frame than the dichotomy Th1/Th2.
2. Material and Methods
2.1. Animals and Infection
We evaluated male animals of Balb/c WT and Balb/c IL-4−/− lineages infected with T. cruzi Colombian strain (n = 8) and an uninfected group (n = 6), both aged 10 weeks and weighing 20-25 g. Infections were performed using trypomastigote forms obtained at peak of parasitemia from previously infected Swiss mice (Waynforth and Flecknell, 1992). Briefly, Swiss mice were euthanized and peripheral blood was collected by cardiac puncture. Five microliters of blood was used to quantify parasite number in a hematocytometer, counted in fifty microscopic fields at a final magnification of ×400. Inoculum was adjusted to 1000 trypomastigotes/ml. One hundred (100) trypomastigote forms were subcutaneously inoculated. Animals were euthanized on the 21st day of infection for acute phase. Next, necropsy was performed and the heart was collected through a ventral incision in thoracic cavity. This research was approved by CEUA/UFG.
2.2. Histological Analysis
To evaluate inflammatory infiltrate, slides of cardiac tissue (ventricle) stained with hematoxylin and eosin (HE) were used. Qualitative analysis classified infiltrate as predominantly mononuclear (macrophages and lymphocytes) or polymorphonuclear (neutrophils and eosinophils), according to the cellular type observed in more than 50% of the infiltrate. Semiquantitative analysis classified the inflammatory infiltrate in mild (involvement < 25% of the tissue), moderate (25%–50% of the tissue), or severe (involvement > 50% of the tissue) [25].
Tissue parasitism was quantified under light microscopy by the number of myocardium T. cruzi nests using three slides with serial slices. All infected animals had nests.
For collagen quantification, we used the heart sections stained with sirius red. Slides were analyzed in polarized light microscope at a final magnification of ×400 with a semiautomatic interactive image analyzer system, ImageJ® (National Institutes of Health, Bethesda, EUA).
2.3. Immunological Analysis
2.3.1. Cardiac Tissue Homogenate Preparation
The heart tissue sections were immersed in PBS solution containing complete protease inhibitor (Sigma, St. Louis, MO, USA) and Nonidet-P40. After that, they were submitted to tissue homogenizer. The homogenate obtained was centrifuged at 14,000 ×g for 10 minutes, and the supernatant was maintained for quantification of cytokines and total proteins [25].
2.3.2. Culture of Splenocytes
Mouse splenocytes were collected, maintained in RPMI 1640 medium (GE Healthcare, Uppsala, Sweden) and macerated for cell individualization. These suspended cells were washed three times by centrifugation at 400 ×g for 15 min at 8°C in RPMI 1640. Then, they were counted in a Neubauer chamber and resuspended to 2 × 106 cells/ml in RPMI 1640 medium with addition of 50 mM Hepes (Gibco, Grand Island, NY, USA), 5% of inactivated fetal bovine serum (Gibco, USA), 2 mM L-glutamine (Gibco, USA), 0.05 mM 2β-mercaptoethanol (Gibco, USA), and 40 μg/ml gentamicin (Neoqumica, Anápolis, GO, BR). Then, 2 × 106 cells were incubated without stimulus or with 10 μg/ml of concanavalin A in 24-well culture plates (BD Pharmingen, San Diego, CA, USA). Cultures were kept in a moist incubator with 5% CO2 at 37°C for 48 hours. Supernatants were collected and maintained at −70°C until analysis [25].
2.3.3. TNF-α, IL-12p70, IL-10, IFN-γ, and IL-17 Quantification
Cytokine quantification was performed on cardiac homogenate, spleen culture, and serum cells by ELISA (enzyme-linked immunosorbent assay), using antibody pairs from BD commercial kit (Biosciences, USA) and following manufacturer's instructions. Reaction was developed using 3,3′,5,5′-tetramethylbenzidine (TMB) peroxidase substrate and read at 450 nm. For cardiac homogenate, results were normalized to total protein concentration, determined by Bradford assay (Bio-Rad, Hercules, CA, USA) of each heart and expressed as picogram of cytokine per gram of tissue (pg/g of tissue).
2.4. Statistical Analyses
The GraphPad Prism 6.0 software (GraphPad Software, USA) was used. Student's t-test was used for analysis between two groups with normal distribution.
Qualitative variables were expressed as percentage and associations were analyzed using the chi-square (χ2) test. Results were considered statistically significant when p < 0.05.
3. Results
3.1. Balb/c IL-4−/− Mice Infected with T. cruzi Strain Presented Less Intense Inflammatory Infiltrate in Acute Phase of Infection
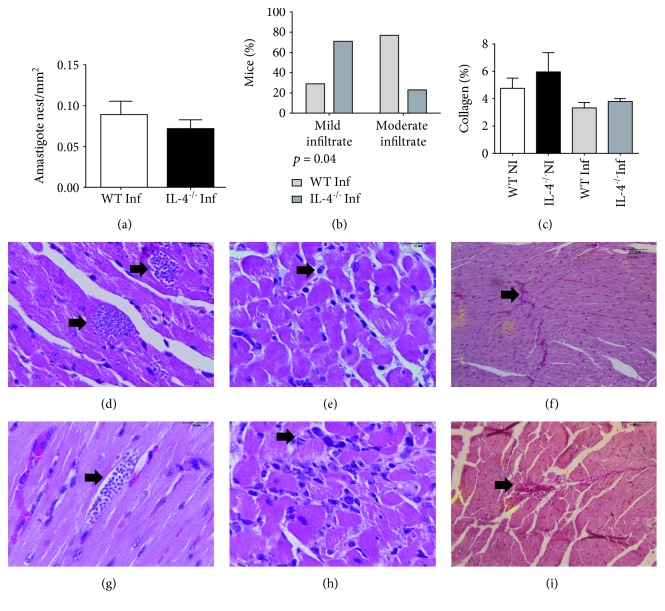
Inflammatory cardiac infiltrate in Balb/c WT and Balb/c IL-4−/− mice infected with Colombian strain in acute phase of experimental Chagas disease was analyzed. IL-4 absence had an impact on inflammatory infiltrate reduction, as Balb/c IL-4−/− had predominantly moderate inflammatory infiltrate (p = 0.04, chi-square test, Figure 1). Regarding quantification of heart fibrosis and amastigotes nests, no difference was observed in group comparison (Figure 1).
Figure 1.
Morphological analysis of the heart of Balb/c WT and Balb/c IL-4−/− mice infected and not infected with the Colombian strain of T. cruzi in the acute phase of experimental Chagas' disease. (a) Density of amastigote nests in cardiac tissue of infected WT and IL-4−/− mice. Bars represent the mean, and vertical lines represent the standard error. (b) Intensity of cardiac inflammatory infiltrate in infected WT and IL-4−/− mice (p = 0.04, chi-square test). (c) Percentage of collagen fibers in cardiac tissue of infected and uninfected WT and IL-4−/− mice. Bars represent the mean, and vertical lines represent the standard error. Histological sections of the WT and IL-4−/−-infected mice heart. (d, g) HE-stained amastigote nests. (e) Mild inflammatory infiltrate. (h) Moderated inflammatory infiltrate. (f, i) Collagen fibers stained red by sirius red.
3.2. Absence of IL-4 Culminates in Lower IFN-γ Cardiac Tissue Production with Similar Expression of TNF-α
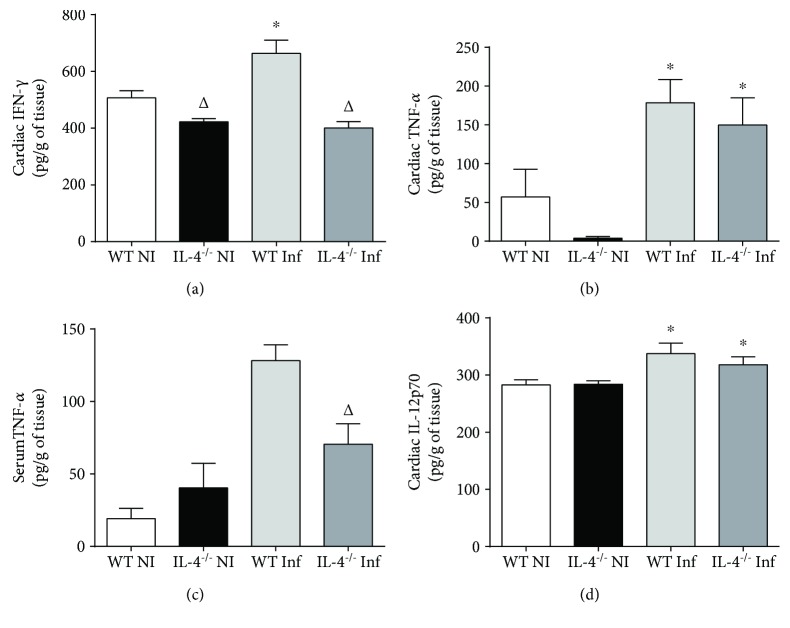
Once infection with T. cruzi Colombian strain leads to lower inflammatory infiltrate in IL-4 knockout animals, we evaluated immune response quality in situ through cardiac homogenate and systemically through serum dosages. As expected, wild-type animal infection triggered increased IFN-γ production in cardiac tissue (noninfected WT vs. infected WT, p = 0.02, t = 2.611). However, in IL-4 absence, IFN-γ expression was significantly reduced, either basal (NI WT vs. NI IL-4−/−, p = 0.009, t = 3.315) or after infection with Colombian strain (Inf WT vs. Inf IL-4−/−, p = 0.001, t = 4.43). No statistically significant difference was observed in IFN-γ serum levels (data not shown).
However, despite significant IFN-γ reduction in cardiac tissue, TNF-α expression was similar between WT and IL-4−/−, both in uninfected mice (NI WT vs. NI IL-4−/−, p > 0.17) and after infection (Inf WT vs. Inf IL-4−/−, p > 0.5). In both groups of animals, infection with Colombian strain induced a significant increase in TNF-α (NI WT vs. Inf WT, p = 0.02, t = 2.61 and NI IL-4−/− vs. Inf IL-4−/−, p = 0.002, t = 4.74). Interestingly, TNF-α systemic levels were significantly elevated in WT-infected animals compared to IL-4−/− animals (p = 0.01, t = 3.08).
To investigate if nonproduction of IFN-γ was due to nonexpression of Th1-inducing cytokines, we evaluated in situ expression of IL-12p70. In both groups, infection induced significantly increased IL-12p70 expression in cardiac tissue (NI WT vs. Inf WT, p = 0.04, t = 2.32 and NI IL-4−/− vs. Inf IL-4−/−, p = 0.04, t = 2.39), but with no difference between WT and IL-4−/−, in situ or systemically (p > 0.05, Figure 2).
Figure 2.
Expression of proinflammatory cytokines in cardiac tissue and serum of Balb/c WT and Balb/c IL-4−/− mice infected and not infected with the Colombian strain of T. cruzi in the acute phase of experimental Chagas' disease. (a) Tissue IFN-γ production (pg/g). (b) Tissue TNF-α production (pg/g). (c) Serum levels of TNF-α (pg/ml). (d) Tissue IL12p70 production (pg/g). Student's t-test. Bars represent the mean, and vertical lines represent the standard error. ∗Significant differences between infected versus uninfected animals. ΔSignificant differences between the WT versus IL-4−/− group.
3.3. Lower Inflammation and Lower IFN-γ Production in Cardiac Tissue of IL-4−/− Animals Infected with T. cruzi Colombian Strain Are due to Increased IL-10 Production
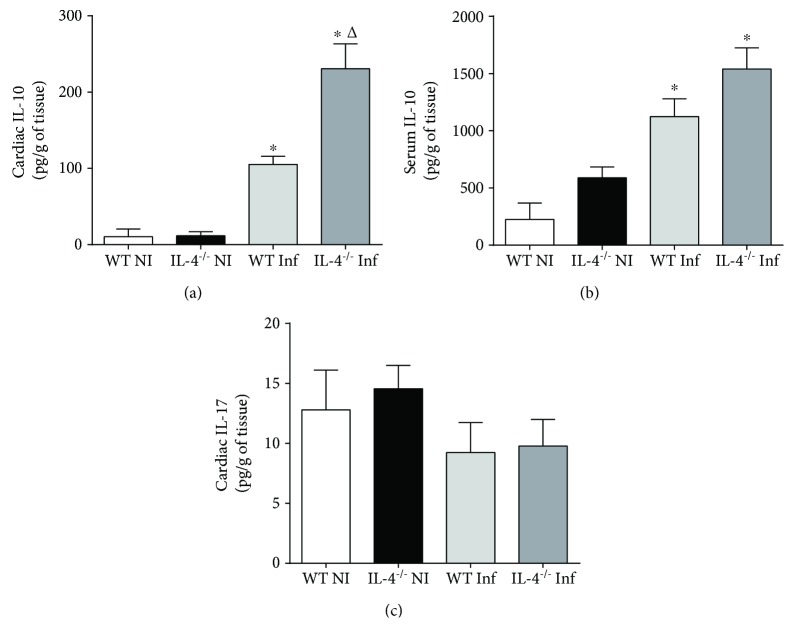
Once we observed Balb/c mice with IL-4 absence and infected with T. cruzi cardiotropic strain had a significant IFN-γ expression reduction in cardiac tissue, and that this reduction was not due to a decrease in innate immunity cytokine production related to Th1 cell differentiation or function, such as TNF-α and IL-12p70, we evaluated a possible differential production of IL-10, classically implicated in anti-inflammatory mechanisms.
Our results indicate that T. cruzi Colombian strain infection induced significant IL-10 production, both in WT mice (NI WT vs. Inf WT, p = 0.007, t = 5.29) and Balb/c IL-4−/− (NI IL-4−/− vs. Inf IL-4−/−, p = 0.001, t = 4.93). Furthermore, in IL-4 absence, significantly higher IL-10 levels were observed in cardiac tissue following infection (Inf WT vs. Inf IL-4−/−, p = 0.004, t = 3.42). A significant increase in serum IL-10 between WT and IL-4 animals−/− (p > 0.05) was not observed, although both had higher systemic levels after infection (NI WT vs. Inf WT, p = 0.002, t = 4.25 and NI IL-4−/− vs. Inf IL-4−/−, p = 0.001, t = 4.89) (Figure 3).
Figure 3.
Cytokine expression with regulatory profile in cardiac tissue and serum of Balb/c WT and Balb/c IL-4−/− mice infected and not infected with the T. cruzi Colombian strain in the acute phase of experimental Chagas' disease. (a) Production of IL-10 in cardiac tissue (pg/g) and (b) serum levels of IL-10 (pg/ml). (c) Production of IL-17 in cardiac tissue (pg/g). Student's t-test. Bars represent the mean, and vertical lines represent the standard error. ∗Significant differences between infected versus uninfected animals. ΔSignificant differences between the WT versus IL-4−/− group.
Finally, considering that IL-4 absence could imply alterations of other non-Th1 inflammatory profiles, we assessed Th17 profile through IL-17 in situ expression. No statistical differences were observed in IL-17 cytokine expression between groups, which shows that nonactivation of Th2 profile by IL-4 absence did not lead to Th17 profile activation in a compensatory fashion (Figure 3).
3.4. Cytokine Pattern in Cardiac Tissue Is Not due to Difference in Splenocyte General Repertoire
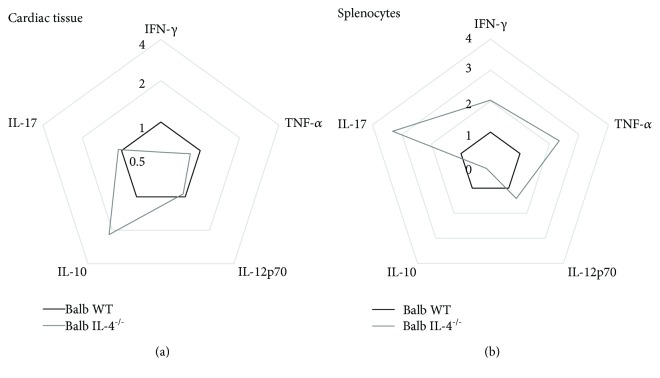
To assess if observed differences in cardiac tissue cytokine expression of IL-4−/− animals were due to a global difference in lymphocyte repertoire, we stimulated splenocytes with 10 μg/ml of concanavalin A for 48 h and calculated cytokine production (expressed in fold increment related to nonstimulated splenocytes). We demonstrate here that IL-4−/− mouse splenocytes produce significantly increased amounts of IFN-γ (Inf WT vs. Inf IL-4−/−, p = 0.02, t = 2.79), TNF-α (Inf WT vs. Inf IL-4−/−, p = 0.009, t = 3.39), IL-17 (Inf WT vs. Inf IL-4−/−, p = 0.006, t = 5.47), and lower IL-10 production (Inf WT vs. Inf IL-4−/−, p = 0.007, t = 5.76) (Figure 4). This result shows a clear difference between repertoire generated by T. cruzi Colombian strain infection and cytokines expressed at the infection site, more clearly demonstrated by radar plots (Figure 5). These results were calculated using the mean expression of each cytokine in cardiac tissue homogenate or CON-A-stimulated splenocytes from Balb/c IL-4−/−-infected mice and expressed as fold change relative to Balb/c WT-infected mice.
Figure 4.
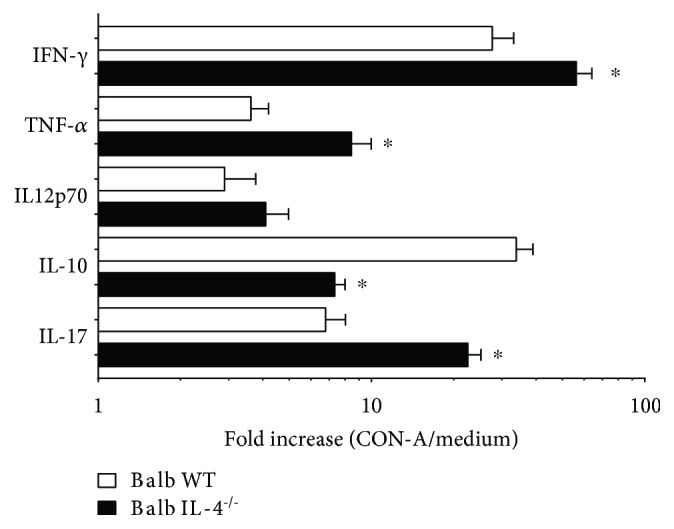
Cytokine production in splenocytes of WT Balb/c and IL-4−/− Balb/c mice infected with Colombian strain of T. cruzi stimulated with 10 μg/ml of concanavalin A in the acute phase of experimental Chagas' disease. Fold change between nonstimulated versus CON-A stimulated splenocytes. Student's t-test. Bars represent the mean, and vertical lines represent the standard error. ∗Significant differences between infected versus uninfected animals.
Figure 5.
Radar plot representation of cytokine profile in cardiac tissue and splenocytes. The lines highlight the fold change in cytokine production in IL-4−/− Balb/c (gray line) in relation to WT Balb/c mice (black line). Data were obtained by calculating the ratio between the mean concentrations of each cytokine in the IL-4−/− Balb/c-infected group and WT Balb/c-infected mice.
4. Discussion
This study is aimed at evaluating IL-4 role in immune response modulation of mice infected with T. cruzi Colombian strain in acute phase of infection. Our results point out the relationship between IL-4 and Th1 cells, classically described as antagonistic. In acute myocarditis triggered by cardiotropic strain of T. cruzi, IL-4 absence implies a general polarization for Th1 in the spleen, but in cardiac tissue, inflammatory balance is significantly regulated by an increase in IL-10, triggering a lower inflammatory infiltrate.
The profile of cytokines released during T. cruzi infection may be associated with a protective or disease susceptible profile. Th1-type response is characterized by IL-2 and IFN-γ secretion, leading to activation of macrophages and cell-mediated response, whereas in Th2 response, synthesis of IL-4, IL-5, and IL-10 is observed, culminating in relevant humoral response. Balance between these cytokines is fundamental to determine which response has to be developed [27, 28].
In our study, IL-4 absence at 21 days of infection did not determine a change in amastigotes nest density in cardiac tissue, using 100 forms of Colombian strain of T. cruzi, a reference strain for chagasic myocarditis studies due to its high heart tropism in Balb/c mice [29, 30]. These results conflict with other studies that demonstrated the potentiating IL-4 effect on infection, since IL-4−/− mice would better control tissue parasitism [21, 31]. However, here and in both studies, nest density was small, even though several serial cuts were evaluated. Another study using greater inoculum with Y strain found no difference in parasite load as did our study [32]. Taken together, all these studies show in the early events of T. cruzi infection that intense myocarditis has no direct relation with the amount of amastigote nests that will persist in chronic phase.
In the present study, Balb/c IL-4−/−-infected mice had milder inflammatory infiltration compared to WT Balb/c. No significant differences were observed in intensity of diffuse and focal inflammation in cardiac tissue compared IL-4 KO and WT animals [31]; however, this work evaluated late acute phase in 30 days of infection, where parasitemia is reduced and transition to chronic infection begins, while we evaluate the moment when Colombian strain presents its parasitemia peak, that is, greater parasitic circulation [25].
Fibrosis is a fundamental substrate of Chagas' heart disease and progression to heart failure [33]. However, there is no consensus on the exact moment process takes place. In face of a continuous inflammatory process, it is believed that extracellular matrix increases collagen production evolving to fibrosis, especially secondary to tissue damage. In the present study, difference in collagen deposition was not found, possibly due to the time infection was evaluated, when myocarditis is prominent and fibrosis is not yet fully installed. In chronic phase, it is well established that induction of inflammatory response and death of cardiac fibers contribute to continuous deposition of collagen and consequently fibrosis [1, 34].
IL-4 absence culminated in lower cardiac tissue IFN-γ production; this fact justifies decrease of in situ infiltrate. However, IFN-γ expression is extremely relevant for immunity against intracellular pathogens, including T. cruzi [35]. We believe systemic production of IFN-γ suppressed local decrease of this cytokine, since its levels were well expressed in splenocytes culture in the IL-4−/− group. However, increased IFN-γ and nitric oxide production was demonstrated in mice infected with T. cruzi Tulahuen strain with IL-4 suppression, showing that IL-4 absence is related to a greater proinflammatory state activation in T. cruzi infection [36].
IL-4 absence did not alter TNF-α in situ production, despite IFN-γ local reduction. TNF-α is especially synthesized by macrophages, T lymphocytes, and NK cells, with a great diversity of functions including recruitment and activation of macrophages, which stimulate nitric oxide production and intracellular destruction of protozoan, actively participating in the initial proinflammatory response of the disease [37, 38]. Other studies have shown that increased TNF-α is also associated with increased cardiac damage [39, 40], although treatment with TNF-α inhibitors, such as etanercept, in chronic phase, aggravates chagasic myocarditis [41], demonstrating that its presence in cardiac tissue has more complex repercussions than conceptual simplification increase is equal to damage. It is important to emphasize that splenocyte production capacity was positively impacted by IL-4 absence, suggesting cardiac tissue-specific control mechanisms.
Among infected animals, there was no difference in IL-12p70 cytokine expression in IL-4 absence. It is known that this cytokine is a fundamental mediator of innate immune response, secreted by mononuclear phagocytes and dendritic cells, and important in stimulating IFN-γ production by NK cells and T lymphocytes [42, 43]. It can be inferred that, in this study, at the beginning of infectious process, the ability to produce IL-12p70 contributes to a Th1 response profile influenced by pathogen itself.
In fact, animals in the present study had no histological evidence of cardiac involvement. We believe this is due to immune response control represented in our study by in situ increase in IL-10 and decrease in IFN-γ. Our data also point out that immune response local regulation associated with a good repertoire of systemic Th1 immune response may have been sufficient to maintain the effectiveness of the response to T. cruzi infection. These data become more consistent, since we observed increased serum and in situ IL-10 with reduced systemic repertoire production, as demonstrated in stimulated culture.
IL-4 absence did not influence IL-17 expression in situ; however, the repertoire in the ex vivo culture increased. IL-17 plays an important role in resolution of T. cruzi protozoan infection, and this cytokine is associated with protective and nonpathogenic responses [44–46]. In chagasic patients with heart disease, greater serum IL-17 levels were related to improvement of organ function; therefore, it has protective effect [47]. A study in IL-17−/− animals demonstrated that this cytokine actively participates in inflammatory response in initial phase of disease and its absence during T. cruzi infection results in a reduction in recruitment of defense cells, which favors parasitemia [48]. However, researches are still recent and limited on true role of this cytokine in Chagas' disease.
The relationship between IL-4 functions and Th1 cell differentiation is classically described as antagonistic and widely reported in several models of T lymphocyte differentiation [49–52]. However, we demonstrated in acute myocarditis triggered by T. cruzi cardiotropic strain that IL-4 absence implies a repertorial polarization for Th1, but in cardiac tissue, inflammatory balance is strongly regulated by an increase in IL-10, triggering a lower inflammatory infiltrate. Our results open perspectives of IL-4 role in initial events of chagasic myocarditis in a more complex frame than the dichotomy Th1/Th2.
Abbreviations
- CON-A:
Concanavalin A
- ELISA:
Enzyme-linked immunosorbent assay
- IFN-γ:
Interferon gamma
- IL-4:
Interleukin 4
- IL-10:
Interleukin 10
- IL-17:
Interleukin 17
- IL-12p70:
Interleukin 12p70
- KO:
Knockout for the gene
- T. cruzi:
Trypanosoma cruzi
- TNF-α:
Tumor necrosis factor-alpha
- TMB:
3,3′,5,5′-Tetramethylbenzidine
- WT:
Wild type.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Authors' Contributions
Marcos Vinicius da Silva and Vera Lúcia de Almeida equally contributed to this paper.
References
- 1.Marin-Neto J. A., Cunha-Neto É., Maciel B. C., Simões M. V. Pathogenesis of chronic Chagas heart disease. Circulation. 2007;115(9):1109–1123. doi: 10.1161/CIRCULATIONAHA.106.624296. [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization (WHO) Preventing Mother-to-Child Transmission of Chagas disease: from Control to Elimination. WHO: Neglected tropical disease; 2018. [Google Scholar]
- 3.Burleigh B. A., Andrews N. W. The mechanisms of Trypanosoma cruzi invasion of mammalian cells. Annual Review of Microbiology. 1995;49(1):175–200. doi: 10.1146/annurev.mi.49.100195.001135. [DOI] [PubMed] [Google Scholar]
- 4.de Araújo-Jorge T. C., de Castro S. L. Doença de Chagas: Manual Para Experimentação Animal. Rio de Janeiro: Editora FIOCRUZ; 2000. [DOI] [Google Scholar]
- 5.Federici E. E., Abelmann W. H., Neva F. A. Chronic and progressive myocarditis and myositis in C3H mice infected with Trypanosoma cruzi. The American Journal of Tropical Medicine and Hygiene. 1964;13(2):272–280. doi: 10.4269/ajtmh.1964.13.272. [DOI] [PubMed] [Google Scholar]
- 6.Andrade S. G., Magalhães J. B. Biodemes and zymodemes of Trypanosoma cruzi strains: correlations with clinical data and experimental pathology. Revista da Sociedade Brasileira de Medicina Tropical. 1997;30(1):27–35. doi: 10.1590/S0037-86821997000100006. [DOI] [PubMed] [Google Scholar]
- 7.Andrade S. G., Pimentel A. R., de Souza M. M., Andrade Z. A. Interstitial dendritic cells of the heart harbor Trypanosoma cruzi antigens in experimentally infected dogs: importance for the pathogenesis of chagasic myocarditis. The American Journal of Tropical Medicine and Hygiene. 2000;63(1):64–70. doi: 10.4269/ajtmh.2000.63.64. [DOI] [PubMed] [Google Scholar]
- 8.Abrahamsohn I. A., Coffman R. L. Trypanosoma cruzi: IL-10, TNF, IFN-γ, and IL-12 regulate innate and acquired immunity to infection. Experimental Parasitology. 1996;84(2):231–244. doi: 10.1006/expr.1996.0109. [DOI] [PubMed] [Google Scholar]
- 9.Hunter C. A., Ellis-Neyes L. A., Slifer T., et al. IL-10 is required to prevent immune hyperactivity during infection with Trypanosoma cruzi. The Journal of Immunology. 1997;158:3311–3316. [PubMed] [Google Scholar]
- 10.Tarleton R. L., Koller B. H., Latour A., Postan M. Susceptibility of β2-microglobulin-deficient mice to Trypanosoma cruzi infection. Nature. 1992;356(6367):338–340. doi: 10.1038/356338a0. [DOI] [PubMed] [Google Scholar]
- 11.Rottenberg M. E., Bakhiet M., Olsson T., et al. Differential susceptibilities of mice genomically deleted of CD4 and CD8 to infections with Trypanosoma cruzi or Trypanosoma brucei. Infection and Immunity. 1993;61(12):5129–5133. doi: 10.1128/iai.61.12.5129-5133.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Santos Lima E. C., Minoprio P. Chagas’ disease is attenuated in mice lacking γδ T cells. Infection and Immunity. 1996;64(1):215–221. doi: 10.1128/iai.64.1.215-221.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lopes M. F., Nunes M. P., Henriques-Pons A., et al. Increased susceptibility of Fas ligand-deficient gld mice to Trypanosoma cruzi infection due to a Th2-biased host immune response. European Journal of Immunology. 1999;29(1):81–89. doi: 10.1002/(SICI)1521-4141(199901)29:01<81::AID-IMMU81>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 14.Higuchi M. d. L. Chronic chagasic cardiopathy: the product of a turbulent host-parasite relationship. Revista do Instituto de Medicina Tropical de São Paulo. 1997;39(1):53–60. doi: 10.1590/S0036-46651997000100012. [DOI] [PubMed] [Google Scholar]
- 15.Silva J. S., Twardzik D. R., Reed S. G. Regulation of Trypanosoma cruzi infections in vitro and in vivo by transforming growth factor β (TGF-β) The Journal of Experimental Medicine. 1991;174(3):539–545. doi: 10.1084/jem.174.3.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Savino W., Villa-Verde D. M. S., Mendes-da-Cruz D. A., et al. Cytokines and cell adhesion receptors in the regulation of immunity to Trypanosoma cruzi. Cytokine & Growth Factor Reviews. 2007;18(1-2):107–124. doi: 10.1016/j.cytogfr.2007.01.010. [DOI] [PubMed] [Google Scholar]
- 17.Hurdayal R., Brombacher F. The role of IL-4 and IL-13 in cutaneous leishmaniasis. Immunology Letters. 2014;161(2):179–183. doi: 10.1016/j.imlet.2013.12.022. [DOI] [PubMed] [Google Scholar]
- 18.Alexander J., Bryson K. T helper (h)1/Th2 and Leishmania: paradox rather than paradigm. Immunology Letters. 2005;99(1):17–23. doi: 10.1016/j.imlet.2005.01.009. [DOI] [PubMed] [Google Scholar]
- 19.Alexander J., Brombacher F. T helper1/T helper2 cells and resistance/susceptibility to Leishmania infection: is this paradigm still relevant? Frontiers in Immunology. 2012;3:p. 80. doi: 10.3389/fimmu.2012.00080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Martinez O. M., Gibbons R. S., Garovoy M. R., Aronson F. R. IL-4 inhibits IL-2 receptor expression and IL-2-dependent proliferation of human T cells. The Journal of Immunology. 1990;144:2211–2215. [PubMed] [Google Scholar]
- 21.Soares M. B. P., Silva-Mota K. N., Lima R. S., Bellintani M. C., Pontes-de-Carvalho L., Ribeiro-dos-Santos R. Modulation of chagasic cardiomyopathy by interleukin-4: dissociation between inflammation and tissue parasitism. The American Journal of Pathology. 2001;159(2):703–709. doi: 10.1016/S0002-9440(10)61741-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hölscher C., Köhler G., Müller U., Mossmann H., Schaub G. A., Brombacher F. Defective nitric oxide effector functions lead to extreme susceptibility of Trypanosoma cruzi-infected mice deficient in gamma interferon receptor or inducible nitric oxide synthase. Infection and Immunity. 1998;66(3):1208–1215. doi: 10.1128/iai.66.3.1208-1215.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wirth J. J., Kierszenbaum F., Zlotnik A. Effects of IL-4 on macrophage functions: increased uptake and killing of a protozoan parasite (Trypanosoma cruzi) Immunology. 1989;66(2):296–301. [PMC free article] [PubMed] [Google Scholar]
- 24.Golden J. M., Tarleton R. L. Trypanosoma cruzi: cytokine effects on macrophage trypanocidal activity. Experimental Parasitology. 1991;72(4):391–402. doi: 10.1016/0014-4894(91)90085-B. [DOI] [PubMed] [Google Scholar]
- 25.Reis Machado J., Silva M. V., Borges D. C., et al. Immunopathological aspects of experimental Trypanosoma cruzi reinfections. BioMed Research International. 2014;2014:9. doi: 10.1155/2014/648715.648715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Paludan S. R. Interleukin-4 and interferon-γ: the quintessence of a mutual antagonistic relationship. Scandinavian Journal of Immunology. 1998;48(5):459–468. doi: 10.1046/j.1365-3083.1998.00435.x. [DOI] [PubMed] [Google Scholar]
- 27.Pinazo M.-J., Thomas M. C., Bustamante J., Almeida I. C., Lopez M. C., Gascon J. Biomarkers of therapeutic responses in chronic Chagas disease: state of the art and future perspectives. Memórias do Instituto Oswaldo Cruz. 2015;110(3):422–432. doi: 10.1590/0074-02760140435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dutra W. O., Menezes C. A. S., Magalhães L. M. D., Gollob K. J. Immunoregulatory networks in human Chagas disease. Parasite Immunology. 2014;36(8):377–387. doi: 10.1111/pim.12107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Camandaroba E., Thé T. S., Pessina D. H., Andrade S. G. Trypanosoma cruzi: clones isolated from the Colombian strain, reproduce the parental strain characteristics, with ubiquitous histotropism. International Journal of Experimental Pathology. 2006;87(3):209–217. doi: 10.1111/j.1365-2613.2006.00476.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Andrade L. O., Machado C. R. S., Chiari E., Pena S. D. J., Macedo A. M. Trypanosoma cruzi: role of host genetic background in the differential tissue distribution of parasite clonal populations. Experimental Parasitology. 2002;100(4):269–275. doi: 10.1016/S0014-4894(02)00024-3. [DOI] [PubMed] [Google Scholar]
- 31.Michailowsky V., Silva N. M., Rocha C. D., Vieira L. Q., Lannes-Vieira J., Gazzinelli R. T. Pivotal role of interleukin-12 and interferon-γ axis in controlling tissue parasitism and inflammation in the heart and central nervous system during Trypanosoma cruzi infection. The American Journal of Pathology. 2001;159(5):1723–1733. doi: 10.1016/S0002-9440(10)63019-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Abrahamsohn I. A., da Silva A. P. G., Coffman R. L. Effects of interleukin-4 deprivation and treatment on resistance to Trypanosoma cruzi. Infection and Immunity. 2000;68(4):1975–1979. doi: 10.1128/IAI.68.4.1975-1979.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lopes E. R., Chapadeiro E., Tafuri W. L., Almeida A. O., Abraão D. Peso do coração e tipo de morte no chagásico. Revista do Instituto de Medicina Tropical de São Paulo. 1970;12:293–297. [PubMed] [Google Scholar]
- 34.Torreão J. A., Ianni B. M., Mady C., et al. Myocardial tissue characterization in Chagas’ heart disease by cardiovascular magnetic resonance. Journal of Cardiovascular Magnetic Resonance. 2015;17(1):p. 97. doi: 10.1186/s12968-015-0200-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Higuchi M. d. L., de Brito T., Martins Reis M., et al. Correlation between Trypanosoma cruzi parasitism and myocardial inflammatory infiltrate in human chronic chagasic myocarditis: light microscopy and immunohistochemical findings. Cardiovascular Pathology. 1993;2(2):101–106. doi: 10.1016/1054-8807(93)90021-S. [DOI] [PubMed] [Google Scholar]
- 36.Hiyama K., Hamano S., Nakamura T., Nomoto K., Tada I. IL-4 reduces resistance of mice to Trypanosoma cruzi infection. Parasitology Research. 2001;87(4):269–274. doi: 10.1007/PL00008577. [DOI] [PubMed] [Google Scholar]
- 37.Langermans J. A., van der Hulst M. E., Nibbering P. H., van Furth R. Endogenous tumor necrosis factor alpha is required for enhanced antimicrobial activity against Toxoplasma gondii and Listeria monocytogenes in recombinant gamma interferon-treated mice. Infection and Immunity. 1992;60(12):5107–5112. doi: 10.1128/iai.60.12.5107-5112.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rodrigues A. A., Notário A. F. O., Teixeira T. L., et al. A high throughput analysis of cytokines and chemokines expression during the course of Trypanosoma cruzi experimental oral infection. Acta Tropica. 2016;157:42–53. doi: 10.1016/j.actatropica.2016.01.025. [DOI] [PubMed] [Google Scholar]
- 39.Haensel A., Mills P. J., Nelesen R. A., Ziegler M. G., Dimsdale J. E. The relationship between heart rate variability and inflammatory markers in cardiovascular diseases. Psychoneuroendocrinology. 2008;33(10):1305–1312. doi: 10.1016/j.psyneuen.2008.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cunha-Neto E., Nogueira L. G., Teixeira P. C., et al. Immunological and non-immunological effects of cytokines and chemokines in the pathogenesis of chronic Chagas disease cardiomyopathy. Memórias do Instituto Oswaldo Cruz. 2009;104(Suppl 1):252–258. doi: 10.1590/S0074-02762009000900032. [DOI] [PubMed] [Google Scholar]
- 41.Bilate A. M. B., Salemi V. M., Ramires F. J., et al. TNF blockade aggravates experimental chronic Chagas disease cardiomyopathy. Microbes and Infection. 2007;9(9):1104–1113. doi: 10.1016/j.micinf.2007.05.014. [DOI] [PubMed] [Google Scholar]
- 42.Rogge L., Barberis-Maino L., Biffi M., et al. Selective expression of an interleukin-12 receptor component by human T helper 1 cells. The Journal of Experimental Medicine. 1997;185(5):825–832. doi: 10.1084/jem.185.5.825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cardoso M. S., Reis-Cunha J. L., Bartholomeu D. C. Evasion of the immune response by Trypanosoma cruzi during acute infection. Frontiers in Immunology. 2015;6:p. 659. doi: 10.3389/fimmu.2015.00659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Miyazaki Y., Hamano S., Wang S., Shimanoe Y., Iwakura Y., Yoshida H. IL-17 is necessary for host protection against acute-phase Trypanosoma cruzi infection. The Journal of Immunology. 2010;185(2):1150–1157. doi: 10.4049/jimmunol.0900047. [DOI] [PubMed] [Google Scholar]
- 45.da Matta Guedes P. M., Gutierrez F. R. S., Maia F. L., et al. IL-17 produced during Trypanosoma cruzi infection plays a central role in regulating parasite-induced myocarditis. PLoS Neglected Tropical Diseases. 2010;4(2, article e604) doi: 10.1371/journal.pntd.0000604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bermejo D. A., Jackson S. W., Gorosito-Serran M., et al. Trypanosoma cruzi trans-sialidase initiates a program independent of the transcription factors RORγt and Ahr that leads to IL-17 production by activated B cells. Nature Immunology. 2013;14(5):514–522. doi: 10.1038/ni.2569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sousa G. R., Gomes J. A. S., Damasio M. P. S., et al. The role of interleukin 17-mediated immune response in Chagas disease: high level is correlated with better left ventricular function. PLoS One. 2017;12(3, article e0172833) doi: 10.1371/journal.pone.0172833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tosello Boari J., Amezcua Vesely M. C., Bermejo D. A., et al. IL-17RA signaling reduces inflammation and mortality during Trypanosoma cruzi infection by recruiting suppressive IL-10-producing neutrophils. PLoS Pathogens. 2012;8(4, article e1002658) doi: 10.1371/journal.ppat.1002658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lazarski C. A., Ford J., Katzman S. D., Rosenberg A. F., Fowell D. J. IL-4 attenuates Th1-associated chemokine expression and Th1 trafficking to inflamed tissues and limits pathogen clearance. PLoS One. 2013;8(8, article e71949) doi: 10.1371/journal.pone.0071949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.López-Bravo M., Minguito de la Escalera M., Domínguez P. M., et al. IL-4 blocks TH1-polarizing/inflammatory cytokine gene expression during monocyte-derived dendritic cell differentiation through histone hypoacetylation. The Journal of Allergy and Clinical Immunology. 2013;132(6):1409–1419.e13. doi: 10.1016/j.jaci.2013.08.039. [DOI] [PubMed] [Google Scholar]
- 51.Mosmann T. R., Cherwinski H., Bond M. W., Giedlin M. A., Coffman R. L. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. The Journal of Immunology. 1986;136:2348–2357. [PubMed] [Google Scholar]
- 52.Kurt-Jones E. A., Hamberg S., Ohara J., Paul W. E., Abbas A. K. Heterogeneity of helper/inducer T lymphocytes. I. Lymphokine production and lymphokine responsiveness. The Journal of Experimental Medicine. 1987;166(6):1774–1787. doi: 10.1084/jem.166.6.1774. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.