Table 2.
GST inhibitors and pro-drugs with clinical perspective.
| GST Inhibitors and Pro-Drugs | Mechanism | Clinical Perspective | Structure |
|---|---|---|---|
| Ethacraplatin—containing micelles | enhances the accumulation of active cisplatin in GSTP1 and GSTT1 overexpressing cancer cells by inhibiting the activity of GSTs and circumventing deactivation of cisplatin | with FDA-approved adjuvant, 1,2-distearoylsn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000] (DSPE-PEG2000) |
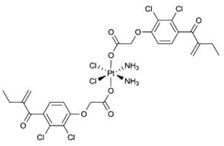 [122] |
| TLK199 | selective inhibitor of GSTP1-1 acting on MAPK signaling pathway and inhibitor of MDR-1 | completed or phase IIa clinical trial in non- small cell lung cancer and myelodysplastic syndrome |
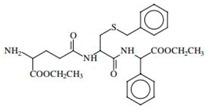 [72,73] |
| Auranofin | GSTP1 inhibitor which enables cells to overcome resistance to platinum-based drug | completed or recruiting phase II clinical trial in ovarian cancer, small and non-small cell lung carcinoma and lung adenocarcinoma |
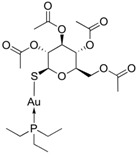 [123] |
| TLK286 | bio-activation by GSTP1-1 into alkylating metabolite capable of covalently binding DNA | completed phase IIa and terminated phase III clinical trial in ovarian, breast and non-small cell lung cancer |
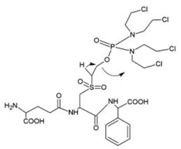 [124] |
| Brostallicin | activated in reactions catalyzed by GSTP and GSTM | completed phase II clinical trial in breast cancer |
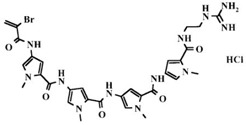 [125] |
