Figure 2.
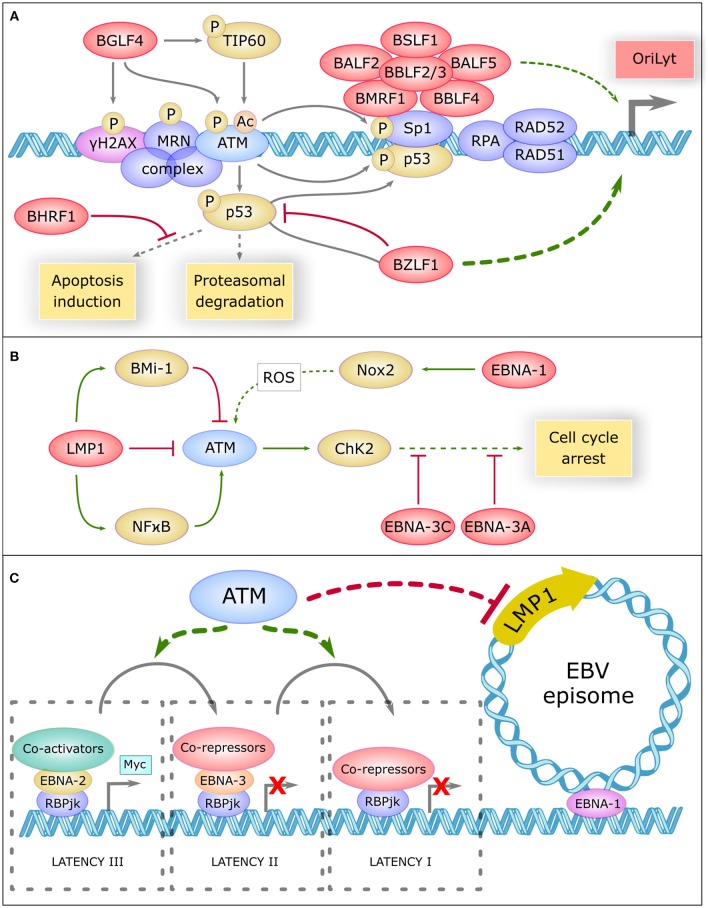
Role Of Atm In Ebv Life Cycle Regulation (A) The central role of ATM in the replication compartment of EBV. In the lytic cycle, DNA damage response proteins such as γH2AX, the MRN complex, ATM, SP1, RPA, RAD51, and RAD52 bind the viral genome and promote replication of the virus. Viral proteins are shown in red. BGLF4 phosphorylates H2AX, ATM, and TIP60 which acetylate ATM to promote this replication. ATM phosphorylates and activates Sp1 which is necessary to the formation of the replication compartment comprising a large complex of six core viral replication proteins (BSLF1, BALF2, BBLF2/3, BALF5, BMRF1, and BBLF4). ATM phosphorylates and activates P53, which is inhibited and driven by BZLF1 to the replication compartment. BZLF1 is a major transactivator of the lytic genes promoter OryLyt. P53 binds to Sp1 and promote the activation of OryLyt. P53 is regulated by proteasomal degradation and can induce apoptosis, but BHRF1 inhibits a panel of pro-apoptotic proteins. (B) ATM is regulated by EBV during latency. In the latent cycle, LMP1 downregulates ATM, and upregulates Bmi-1 which also downregulates ATM. On the other hand, LMP1 activates the NFκB pathway which activates ATM. EBNA-1 upregulates NOX2, which generates reactive oxygen species (ROS) that could activate ATM. Once activated, ATM activates CHK2 which promotes cell cycle arrest. However, EBNA-3C and EBNA-3A inhibit many proteins involved in cell cycle control. (C) Potential involvement of ATM in the regulation of EBV latency. ATM could be involv ed in inhibiting the expression of certain viral oncogenes, such as the main viral oncogene LMP1. ATM could also favor the progressive restriction of EBV latency, from type III latency to type I. In type III latency, EBNA-2 interacts with the target of the Notch pathway RBP-JK, recruits coactivators and induces the transcription of pro-proliferative genes like Myc. In type II latency, EBNA-3 replaces EBNA-2, and recruits co-repressors, thus preventing prolonged expression of MYC. In type I latency, RBP-JK is associated with corepressors and only EBNA-1 remains expressed, which allows the attachment of EBV episome on cellular chromosomes.