Abstract
Cellulose synthase-like D (CSLD) family was characterized for their expression and functions in Populus trichocarpa. Ten members, PtrCslD1-10, were identified in the P. trichocarpa genome, and they belong to 4 clades by phylogenetic tree analysis. qRT-PCR and promoter:GUS assays in Arabidopsis and P. trichocarpa displayed divergent expression patterns of these 10 PtrCSLD genes in root hairs, root tips, leaves, vascular tissues, xylem and flowers. Among PtrCslD2, PtrCslD4, PtrCslD5, PtrCslD6, and PtrCslD8 that all exhibited expression in root hairs, only PtrCslD5 could restore the root hairless phenotype of the atcsld3 mutant, demonstrating that PtrCslD5 is the functional ortholog of AtCslD3 for root hair formation. Our results suggest more possible functions for other PtrCslD genes in poplar.
Introduction
Root hairs are integral for anchorage, enlarging surface area for absorption of water and nutrients, symbiosis interface between plants and soil biome, and expanding exploited soil area to avoid soil erosion1,2. Arabidopsis root hairs have been serving as a model to study cellular morphogenesis, such as plant cell growth and tip growth3. Trichoblast cell’s basal ends of root epidermis specialized to give rise to bulges, which serve as primordia and elongate into thin tubular structures called root hairs4,5. Trichoblasts progressively divide and expand specialized elongating cells at the tip-growing pole6. Differentiating morphological stages of root hairs include specification, initiation, elongation and cessation1,3. A number of genes such as AthA, AthB, CPC, ROP, RSW1, RHD, COW, TIP, CEN, SCN and BST play key roles in root hair tip growth7–10. For examples, CPC promotes trichoblast cell differentiation, AtROP2 and AtROP4 are key factors in bulge initiation11,12, COW1 (CAN OF WORMS), TIP1, CEN1 (CENTEPEDE), CEN2, CEN3 and BST1 (BRISTLED) control one root hair per trichoblast7,9,13. Rapid polarized exocytosis by cell division at the root hair tip supports cell wall maintenance by deposition of cellulose, and loss in any cellulose component would lead to rupture and ectopic root hair formation1,14. To elucidate gene functions in root hair formation, genetic analysis are required5.
The cellulose synthase like (CSL) gene superfamily is composed of 30 genes in Arabidopsis, which encode glycosyltransferases for biosynthesis of polysaccharides and have tissue-specific expression patterns15–18. Based on the sequences, the CSL gene family is divided into CSLA to CSLG groups19. CSLA, CSLC and CSLF are involved in mannan, xyloglucan, and (1 → 3; 1 → 4)-β-D-glucan biosynthesis, respectively15,20–25. The CSLD family member shares high amino acid similarity with the CESA family and is involved in root tip formation22,25. Abnormal flowers, pollen tubes and pollen grains were observed in atcsld1, atcsld4 and nacsld3 mutants26–28, while atcsld2, atcsld3, atcsld5, oscsld1 and oscsld4 mutant seedlings were root hairless16,29–31. PtrCslD2, an ortholog of AtCslD3, showed its expression level in xylem also32.
Yin et al. developed atcsld2/csld3, atcsld2/csld5, atcsld3/csld5 and atcsld2/csld3/csld5 double and triple Arabidopsis knockout mutants. All mutants were dwarf and displayed severe necrosis, indicating the collaborative effects among AtCslD2, AtCslDd3 and AtCslD533. The expression of AtCslD2 is AtCslD3-dependent, and the defects in the atcsld3 mutant were partially compensated for by AtCslD2 overexpression14. Two other CSLD genes, PdCslD5 and PdCslD6, were complementation remedies for defects and abnormalities of atcsld3 mutants, which proved that the aforementioned genes are functional orthologs of the AtCslD334.
AtCslD1 and AtCslD4 are responsible for cellulose deposition in cell walls to avoid ectopic pollen tubes and pollen grains35. AtCslD3 is crucial for the tensile strength of root hair tip cells by deposition of cellulose, and atcsld3 mutants were unable to maintain homeostasis, and terminated bulge elongation at early stage was observed14,36. Stunted root and shoot growth, a decreased concentration of homoglacturonan and xylans, and an elevated concentration of the cellulose synthase inhibitor isoxaben were observed in the atcsld5 mutants15. The rice oscsld1 mutant had normal root hair initiation, but displayed stunted root hair growth, swelling and kinking, showing that OsCslD1 is a functional ortholog of AtCslD3/KOJAK/RHD7 and functioning in root hair elongation30. Retarded growth and arrested cell division due to lack of cellulose deposition in culm and root tips of rice nd1 mutants (OsCslD4) was observed37.
Root and root hair growth have already been explored in maize, rice, cotton and Arabidopsis14,37–39. In this study, we identified 10 CSLD genes (PtrCslD1-10) in P. trichocarpa and investigated their possible functions. We studied their expression pattern by qRT-PCR and promoter::GUS staining, and their involvement in root hair formation was investigated by complementation in the Arabidopsis atcsld3 mutant. We demonstrate the functions of PtrCslD5 in root hair formation and provide preliminary evidence of the involvement of CSLD members in xylem formation.
Results
Characterization of the CSLD family in Populus trichocarpa
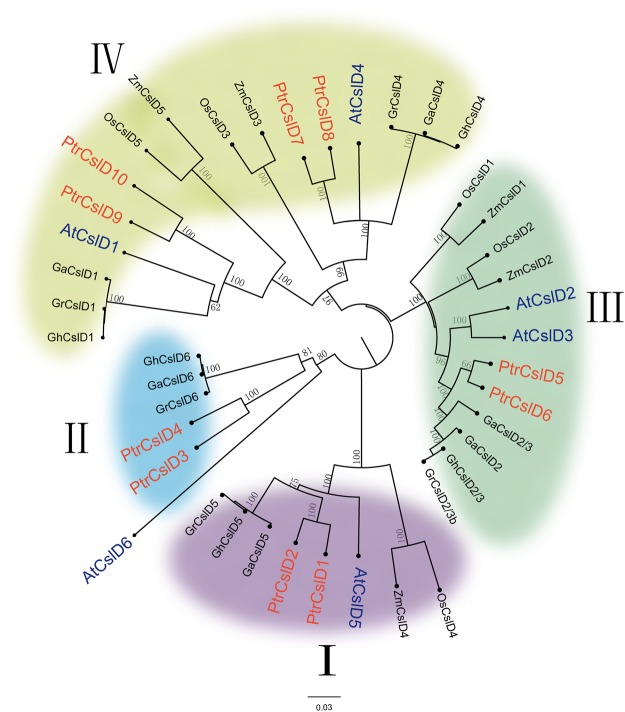
We used Arabidopsis CSLD gene sequences to BLASTN (E-value ≤ 1.0) P. trichocarpa genome and obtained 10 homologous gene models. We named these genes PtrCslD1 (Potri.002G200300), PtrCslD2 (Potri.014G125100), PtrCslD3 (Potri.003G097100), PtrCslD4 (Potri.001G136200), PtrCslD5 (Potri.019G046700), PtrCslD6 (Potri.013G082200), PtrCslD7 (Potri.004G208800), PtrCslD8 (Potri.009G170000), PtrCslD9 (Potri.003G177800), and PtrCslD10 (Potri.001G050200). To understand dynamic topological evolution, a neighbor joining phylogenetic tree was constructed by MEGA 7.040–42 using CSLD genes, including the above P. trichocarpa CSLD genes and the CSLD gene in Arabidopsis7,14,15,31,33,35,43,44, rice, cotton and maize30,34,37. Phylogenetic tree analysis and amino acid sequence comparison among these 10 genes belong to four clades (I–IV). Among these 10 PtrCslD genes, two genes in the same clade had ~89–91% sequence identity, indicating that they belong to gene pairs, probably formed by chromosome duplication45,46.
On phyletic lineage, gene pair PtrCslD1 and PtrCslD2 shared the same clade I with ZmCslD4, OsCslD4, AtCslD5, GaCslD5, GhCslD5, and GrCslD5 (Fig. 1). In this clade, functions of OsCslD4 and AtCslD5 were studied, and both mutants displayed defective root hairs15,37. In clade II, PtrCslD3 and PtrCslD4 shared the same lineage with AtCslD6, GrCslD6, GaCslD6, GhCslD6; none of those were identified for their functions. PtrCslD5 and PtrCslD6 belong to gene pair, sharing high similarity with AtCslD2, AtCslD3, OsCslD1, OsCslD2, GrCslD2, GaCslD2, GhCslD2, NaCslD3, ZmCslD1 and ZmCslD2 in clade III. Among these 9 genes, AtCslD2, AtCslD3, OsCslD1 are required for root hair morphogenesis, and ZmCslD1 is essential for cell division of rapidly growing tissues14,29,30,43,47. Gene pair PtrCslD7 and PtrCslD8 and gene pair PtrCslD9 and PtrCslD10 belong to clade IV. PtrCslD7 and PtrCslD8 are closely related to AtCslD4, and PtrCslD9 and PtrCslD10 are closely related to AtCslD1. Mutation of both AtCslD1 and AtCslD4 caused abnormal flowers, pollen tubes, and pollen grains35. Based on the phylogenetic tree analysis, PtrCslD1, PtrCslD2, PtrCslD5 and PtrCslD6 may function in root hair formation, and PtrCslD7, PtrCslD8, PtrCslD9 and PtrCslD10 may participate in flower and pollen tube development.
Figure 1.
Phylogenetic tree of CSLD genes. A neighbor-joining (NJ) tree was constructed by MEGA 7.0 using 42 CSLD genes, including 10 genes from Populus trichocarpa, 6 genes from Arabidopsis, 5 genes from rice, 5 genes from maize, and 16 genes from cotton. The tree shows 4 distinct clades.
Expression patterns of CSLD members in P. trichocarpa plants
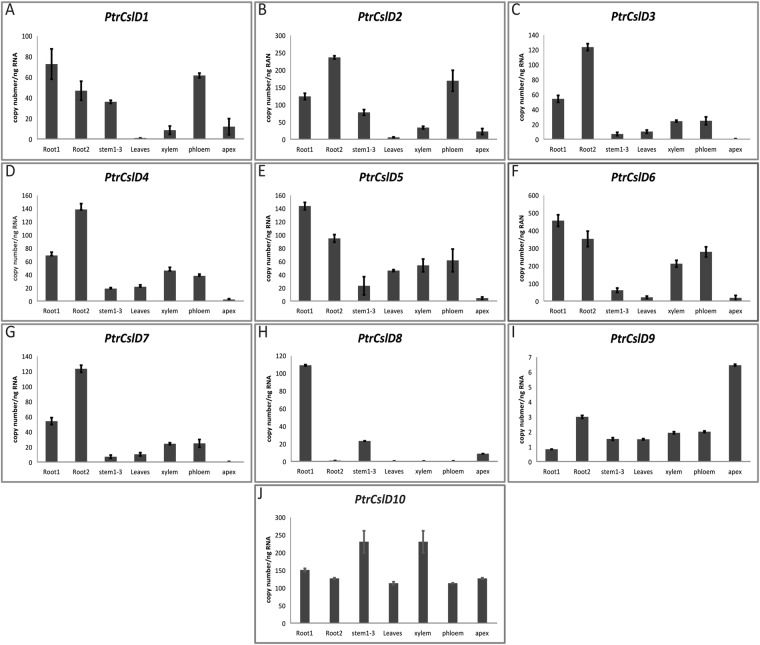
Understanding the gene expression pattern can give some clue as to their possible functions. We used quantitatively RT-PCR to examine the expression patterns of these CSLD genes in P. trichocapra. Their absolute transcript abundance in young roots, mature roots, young stem, leaves, xylem, phloem, and apex were determined. Considering the high nucleotide similarity between gene pairs, we designed specific primers to distinguish the gene pairs.
PtrCslD1 and PtrCslD2 displayed similar expression patterns, with high expression levels in young roots, mature roots, young stems, and phloem, while with relatively low levels in xylem and apex, and the lowest level in leaves. However, PtrCslD1 and PtrCslD2 displayed inverse expression levels between young and mature roots (Fig. 2A,B). A comparatively higher transcript abundance of PtCslD3, 4, 5 and 6 was detected in roots than in young stems, xylem and phloem, and apex had low transcript abundance (Fig. 2C–F). A high expression level in roots was observed for PtrCslD7 and PtrCslD8. PtrCslD7 was highly expressed in both young and mature roots, while PtCslD8 was detected only for its expression in young roots (Fig. 2G,H). PtrCslD9 and PtrCslD10 were expressed in all tissues, but absolute transcript abundance of PtrCslD10 was very low in these tissues (Fig. 2I,J). Although similar expression patterns between each gene pair were generally observed, in some tissues the gene pair exhibited variable expression levels. For example, high transcript abundance was detected in mature roots for PtrCslD7 but not for PtrCslD8.
Figure 2.
Expression analyses of PtrCSLD genes in P. trichocarpa. Absolute transcript abundance of PtrCslD1 (A), PtrCslD2 (B), PtrCslD3 (C), PtrCslD4 (D), PtrCslD5 (E), PtrCslD6 (F), PtrCslD7 (G), PtrCslD8 (H), PtCslD9 (I), and PtrCslD10 (J), were examined in young roots (root 1), mature roots (root 2), stems of internodes 1–3 (stem 1–3), leaves, xylem, phloem, and shoot apex. The plasmid containing the gene was used as a standard for establishing a quantitative correlation between the copy number of the target gene transcript molecules and the CT values. Error bars represent standard errors of triplicate assay.
Expression analysis by promoter::GUS staining
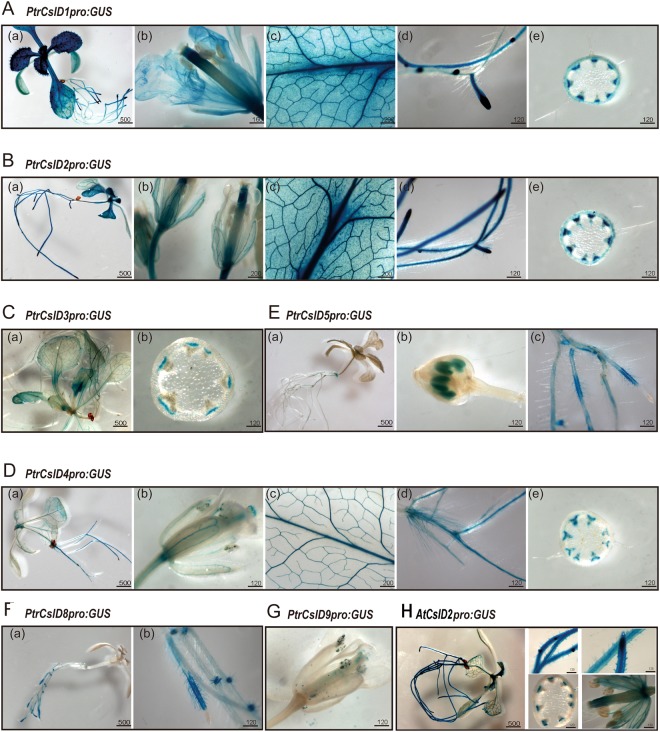
We used the β-glucuronidase (GUS) gene driven by these 10 CSLD gene promoters to provide more detailed information about their gene expression patterns. About 2.3–3.4 kb of the promoter regions upstream start codon were amplified and the promoter:GUS was transformed into Arabidopsis. GUS signals were stained in roots, leaves, xylem and flowers (petal and style) in both pPtrCslD1:GUS and pPtrCslD2:GUS transgenic Arabidopsis (Fig. 3A,B). Strong signals were observed in root tips and leaf vascular tissues in both transgenics, but GUS signals were observed only in root hairs of pPtrCslD2:GUS transgenics (Fig. 3A,B). PtrCslD4 promoter-driven GUS signals were detected in various tissues, including root hairs, vascular tissues of leaves and petals, vascular bundles of stems, and pollen grains (Fig. 3D). Compared to the PtrCslD4 promoter, the PtrCslD3 promoter only gave weak GUS signals in phloem and vascular tissue of leaves (Fig. 3C). GUS staining showed both PtrCslD5 and PtrCslD8 promoters were activated in root hairs (Fig. 3E,F), and PtrCslD5 promoter-driven GUS expression was also detected in pollen sac (Fig. 3E). The observed GUS signals in Arabidopsis root hairs were consistent with the high transcript abundance determined by qRT-PCR (Fig. 2H). In pPtrCslD9:GUS transgenic Arabidopsis, GUS signals were detected only in pollen grains (Fig. 3G). We did not observe any GUS signals in the transgenic Arabidopsis of pPtrCslD6:GUS and pPtrCslD7:GUS.
Figure 3.
GUS staining of promoter:GUS in Arabidopsis. PtrCslD1 promoter:GUS (A). PtrCslD2 promoter:GUS (B). PtrCslD3 promoter:GUS (C). PtrCslD4 promoter:GUS (D). PtrCslD5 promoter:GUS (E). PtrCslD8 promoter:GUS (F). PtrCslD9 promoter:GUS (G). AtCslD2 promoter:GUS (H). Scale bar unit is μm.
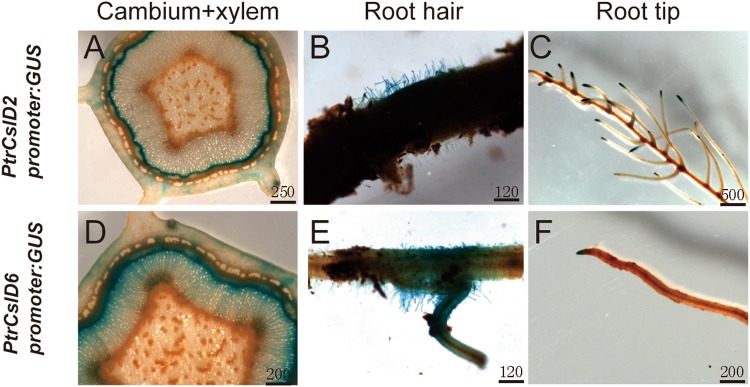
pPtrCslD2:GUS in Arabidopsis had GUS signals in root hairs, but pPtrCslD2:GUS did not, indicating PtrCslD2 might express in root hairs of P. trichocarpa. To confirm the expression of PtrCslD2 in root hairs of P. trichocarpa, we transformed pPtrCslD2:GUS into P. trichocarpa. GUS staining in P. trichocarpa showed strong signals in developing xylem, root hairs, and root tips (Fig. 4A–C), consistent with the GUS staining in Arabidopsis. Both PtrCslD6 and PtrCslD7 promoters did not give GUS signals in Arabidopsis. We selected the PtrCslD6 promoter to test its ability in P. trichocarpa. GUS was stained in xylem, root hairs and root tips in pPtrCslD6:GUS transgenic poplar (Fig. 4D–F).
Figure 4.
GUS staining of promoter:GUS in P. trichocarpa. GUS signals were detected in cambium and xylem (A), root hair (B) and root tips (C) for PtrCslD2 promoter:GUS. GUS signals were also detected in cambium and xylem (D), root hair (E) and root tips (F) for PtrCslD6 promoter:GUS. Scale bar unit is μm.
Complementation to the atcsld3 mutant
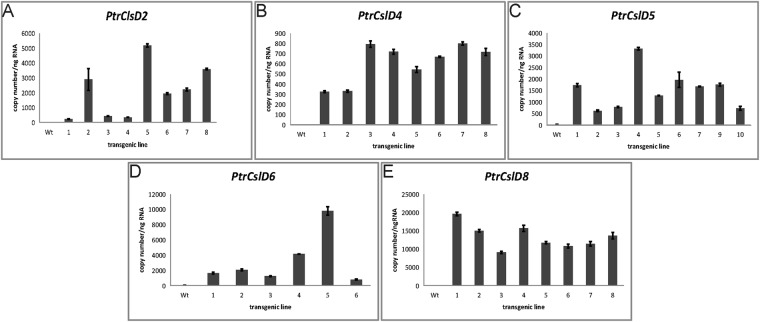
In the above promoter:GUS experiments, we observed GUS signals in root hairs in several transgenics. The promoters of PtrCslD2, PtrCslD4, PtrCslD5, and PtrCslD8 could drive GUS expression in Arabidopsis (Fig. 3). Transformation of PtrCslD2 and PtrCslD6 promoter-driven GUS in P. trichocarpa gave GUS signals in root hairs. To identify the functions of these five CSLD genes in root hairs, we overexpressed these genes in the root hair mutant atcsld3. Absolute transcript abundance of transgenes in the complementation Arabidopsis was determined by qRT-PCR to confirm their expression (Fig. 5). Numerous root hairs were observed in wildtype Arabidopsis (Fig. 6A). In the atcsld3 mutant, root hairs were hardly seen (Fig. 6B). Complementation of atcsld3 with PtrCslD2, PtrCslD4 and PtrCslD8 gave only a few short root hairs (Fig. 6C–E). Compared with PtrCslD2, PtrCslD4 and PtrCslD8, overexpression of PtrCslD6 in atcsld3 produced more and longer root hairs, but the root hair number was much fewer and root hairs were shorter than in wildtype (Fig. 6F). PtrCslD5 complemented mutants appeared with bunches of root hairs (Fig. 6G), and the root hair length was the same as in the wildtype, indicating that PtrCslD5 is the functional ortholog of AtCslD3.
Figure 5.
Transgene expression level in atcsld3 complemented roots. The atcsld3 mutant was complemented with PtrCslD2 (A), PtrCslD4 (B), PtrCslD5 (C), PtrCslD6 (D), and PtrCslD8 (E). 6–10 transgenic lines were examined for absolute transcript abundance of the transgene in roots.
Figure 6.
Complementation to the atcsld3 mutant. Comparison of root hair phenotype among wildtype (A), atcsld3 (B), atcsld3 complemented by PtrCslD2 (C), atcsld3 complemented by PtrCslD4 (D), atcsld3 complemented by PtrCslD8 (E), atcsld3 complemented by PtrCslD6 (F), and atcsld3 complemented by PtrCslD5 (G). Scale bar unit is μm.
Discussion
Root hairs, tubular appendages of trichoblast cells of rhizodermis are integral for plant growth, soil anchorage, water and mineral adsorption, symbiotic interface for mychorhizae and nitrogen-fixing bacteria. Arabidopsis thaliana root epidermis cells are being used as a model to study cell growth and function1,2,14,48–50. Rapid axillary mitotic division of root hairs requires proper cellulose deposition1,14. Cellulose synthase A (CesA) family members are responsible for cellulose biosynthesis, while cellulose synthesis like D (CSLD) family members are involved in cellulose deposition in both primary and secondary cell walls5. Disruption of cellulose deposition would affect root hair formation. In Arabidopsis, several CSLD members, including AtCslD2, AtCslD3 and AtCslD5, have been characterized for their functions in root hair formation33. The root hairless phenotype was observed in the atcsld3 mutant31, and mutant characterization showed that AtCslD3 is functioning in the initiation of root hair formation33. AtCslD2 is functioning at a later stage of root hair development, and the atcsld2 mutant had abnormal root hairs, with many rupturing late in development43. AtCslD5 has functions redundant with AtCslD2 and AtCslD333. In our studies on CSLD members in P. trichocarpa, only PtrCslD5 could restore the root hairless phenotype of atcsld3, indicating that PtrCslD5 is the functional ortholog of AtCslD3. However, PtrCslD6 shared 96% amino acid sequence identity with PtrCslD5 but had very little complementation with atcsld3. In another study, both PdCslD5 and PdCslD6 from P. deltoids could rescue the root hairless phenotype in the atcsld3 mutation34. The difference on the complementation to atcsld3 between PtrCslD6 and the other three CSLD genes (PtrCslD5, PdCslD5 and PdCslD6) indicates that some key amino acids may be changed in PtrCslD6, reducing its function in root hair formation. PtrCslD6 and PdCslD6 share a 99% amino acid identity, with two amino acid difference (valine vesus isoleucine, and glycine versus serine). It will be interesting to further investigate the roles of these two amino acids in root hair formation. Based on the GUS staining in promoter:GUS transgenic Arabidopsis and poplar, PtrCslD2, PtrCslD4, PtrCslD6, and PtrCslD8 may also function in root hair formation. It is interesting that the GUS expression driven by PtrCslD5 and PtrCslD8 promoters were at the same places, root hair and epidermis of root hair zone, but PtrCslD8 did not restore the phenotype of atcsld3. GUS staining shows that PtrCslD2 promoter activity is induced at an early stage of root hair development, while PtrCslD4 promoter activity is induced at a later stage of root hair development, suggesting PtrCslD2 and PtrCslD4 may function at different stages of root hair development. The functions of PtrCslD2, PtrCslD4, PtrCslD6, and PtrCslD8 genes in root hair formation and whether they are functioning cooperatively with PtrCslD5 need further studies, such as complementation to atcsld2 and atcsld5 mutants or knockout poplar mutant generation.
In Arabidopsis and other species, CSLD members function not only in root hair formation but also in other tissues, such as vascular tissues and pollen35. Strong GUS staining of PtrCslD1 and PtrCslD2 promoters was observed in vascular tissue of leaves and vascular bundles of stems (Fig. 3A,B), and comparatively light GUS signals in vascular tissue were detected for PtrCslD3 and PtrCslD4 promoters (Fig. 3C,D). Phylogenetic tree analysis shows PtrCslD1 and PtrCslD2 are in the clade with AtCslD5 and OsCslD4, and PtrCslD3 and PtrCslD4 are in the same clade with AtCslD6. Triple mutant csld2/csld3/csld5 had asymmetric loops and discontinuous vascular elements, showing that AtCslD5, a gene important for root hair formation, is also functioning in vascular tissues33. OsCslD4 is expressed in the apex of many organs with rapid growth, and its mutation had many effects, such as inhibited plant growth, thin culms, small grains etc37. In clade IV, AtCslD1 and AtCslD4 are closely related to gene pair PtrCslD9/10 and gene pair PtrCslD7/8. Both AtCslD1 and AtCslD4 are important for pollen tube growth35. Combining the gene expression patterns and functional characterizations of these 10 PtrCSLD genes and other CSLD genes in other species, we found that some CSLD genes are functioning in multiple tissues, such as root hairs, pollen tubes and vascular tissues. For example, the qRT-PCR and promoter:GUS staining experiment (Figs 2 and 4) showed that PtrCslD2 was expressed in various tissues, including root hairs, root tips, and xylem. The expression is in accordance with AtCslD5 expression in Arabidopsis15. The expression of PtrCslD6 was observed in root hairs, root tip, and xylem in P. trichocarpa, indicating its roles in the root hairs, root tip and xylem. The expression pattern and predicted function of PtrCslD6 are in accordance with the functions of AtCslD2, AtCslD3 and AtCslD5 in root hairs, xylem and tip growth33. Although PtrCslD2 and PtrCslD6 promoters also drove GUS signals in the P. trichocarpa cambium that is lacking in Arabidopsis stems, we assume the expression in cambium is in accordance with the expression of AtCslD2, AtCslD3 and AtCslD5 in tip tissue51 (Fig. 3H). These results indicate that the CSLD genes share a certain level of conservation between Arabidopsis and poplar, and the CSLD genes in P. trichocarpa may play roles in the same tissues of root hair, vascular tissue and pollen tube, as in Arabidopsis.
We also observed difference between Arabidopsis and poplar related to CSLD gene functions and regulation. The activities of the promoters of PtrCslD1 to PtrCslD9 were studied in Arabidopsis through promoter:GUS experiments. Surprisingly, no GUS staining was observed for PtrCslD6 and PtrCslD7 promoters. However, the PtrCslD6 promoter was active in P. trichocarpa, with staining in root hairs, root tips, cambium, and xylem (Fig. 4). This difference indicates that the upstream regulator(s) of PtrCslD6 between Arabidopsis and poplar may be different. PtrCslD1 and PtrCslD2 transcripts were detected at a very low level in leaves in P. trichocarpa (Fig. 2A,B), but both promoters gave strong signals in Arabidopsis leaves (Fig. 3A,B), indicating that the promoters are activated differently between Arabidopsis and poplar. Besides, the expression level of PtrCslD1 in young roots was higher than that in mature roots, but PtrCslD2 displayed an opposite expression pattern in young roots and mature roots. The occurrence of the different expression patterns between PtrCslD1 and PtrCslD2 in roots may be formed after chromosome duplication.
Methods
Plant Materials and Growth
Populus trichocarpa (Nisqually-1) were obtained from tissue culture and grown on Murashige and Skoog (MS) medium on 16 h/8 h light and dark under aseptic conditions at 25–28 °C as described previously52. Seeds of the Arabidopsis thaliana atcsld3 mutant line (AT3G03050) were obtained from Nottingham Arabidopsis Stock Centre (NASC, Nottingham, UK). The seeds were surface-sterilized with sterilizing solution (0.1% Trition and 20% NaClO) for 12 minutes, washed with sterilized distilled water and sown on the solid medium containing MS salts for three days before the seeds were put into an illumination incubator at 22 °C with fluorescent white light at 16/8 h light and dark cycles.
Bioinformatics analysis
The CSLD family genes of A. thaliana were downloaded from the Arabidopsis Tair database (https://www.arabidopsis.org/) and blasted in P. trichocarpa genome via BLASTn search tool with E-value ≤ 1.0. The homologous gene sequences were downloaded from the Phytozome 10.1 plant genomics portal (https://phytozome.jgi.doe.gov/pz/portal.html). We also downloaded CSLD family genes already characterized in different species from the NCBI (https://www.ncbi.nlm.nih.gov/gene) database. The unrooted phylogenetic tree for multiple alignment analysis of protein sequences predicted from cDNA sequences of A. thaliana, G. hirsutum, G. arboreum, G. raimondii, O. sativa, Z. mays, and P. trichocarpa CSLD genes was constructed with the MEGA 7.0 tool using the Neighbor-Joining (NJ) method through 2000 bootstrap replicates40. Each protein encoded by the P. trichocarpa CLSD gene family was assigned a specific name according to Van Erp and Walton39.
Quantitative reverse transcription and PCR (qRT-PCR)
For the PtrCslD gene expression pattern analysis, leaves, shoot apices, young stems of 1–3 internodes, xylem, phloem, young roots and mature roots were collected from six-month-old trees and put in liquid nitrogen immediately. Total RNA was extracted using the CTAB method53. For the qRT-PCR analysis of PtrCslD genes in the atcsld3 mutant, the total RNA was extracted from the roots using an RNeasy Plant Mini Kit (Qiagen). The reverse transcription of RNA to cDNA and quantitative polymerase chain reaction (PCR) were carried out as described previously52. The primers used in the qRT-PCR are listed in Supplemental Table S1.
Promoter-driven GUS expression in Arabidopsis and P. trichocarpa
The promoter regions of 2.4–3.4 kb upstream start codon were amplified for PtrCSLD genes using specific primers (Supplemental Table S1). The sizes of amplified fragments were 3.44 kb (PtrCslD1), 2.34 kb (PtrCslD2), 2.73 kb (PtrCslD3), 2.73 kb (PtrCslD4), 2.5 kb (PtrCslD5), 2.7 kb (PtrCslD6), 2.75 kb (PtrCslD7), 2.78 kb (PtrCslD8), 2.8 kb (PtrCslD9), and 2.67 kb (PtrCslD10). The promoters of PtrCslD1 to PtrCslD9 were successfully amplified. The amplified fragments were cloned into pCR2.1 for sequencing. Further, the promoter fragments were excised from pCR2.1 vectors and inserted into pBI121 by replacing the 35 S promoter, generating pPtrCslD1:GUS, pPtrCslD2:GUS, pPtrCslD3:GUS, pPtrCslD4:GUS, pPtrCslD5:GUS, pPtrCslD6:GUS, pPtrCslD7:GUS, pPtrCslD8:GUS, and pPtrCslD9:GUS. All constructs were introduced into the Agrobacterium tumefaciens strain GV3101. Transformation in Arabidopsis followed the floral dip method54. T1 transgenic plants were screened on MS plates with 30 mg/L kanamycin and transferred to MS plates without kanamycin. Agrobacterium-mediated transformation in P. trichocarpa was conducted using 5–8 internode stems as explants following the previous publication55. After being verified by PCR using DNA as templates, the transgenic plants were moved into pots and maintained in a greenhouse. GUS staining and observation were conducted as described previously52.
Mutant complementation
The total RNA isolated from the xylem for qRT-PCR analysis was reverse-transcribed to cDNA using an Omniscript RT kit (Qiagen). Using the cDNA as templates, the full-length cDNAs of PtrCslD2, PtrCslD4, PtrCslD5, PtrCslD6 and PtrCslD8 were amplified with designed primers (Supplemental Table S1). The PCR fragments were inserted into pBI121 to replace the GUS gene, generating 35 S:PtrCslD2, 35 S:PtrCslD4, 35 S:PtrCslD5, 35 S:PtrCslD6, and 35 S:PtrCslD8. After transformation in the atcsld3 mutant by floral dip method, 6–10 lines were confirmed for transgene expression in roots by qRT-PCR as described above. The root hairs in the wildtype, atcsld3 mutant, and complementation plants were photographed under a Zeiss (Stemi DV4) microscope.
Electronic supplementary material
Acknowledgements
This work was supported by a grant from Fundamental Research Funds of Chinese Academy of Forestry (CAFYBB2017MA030).
Author Contributions
Q.L. designed the experiments; X.P., H.P., M.A., H.L, X.Y. and X.D. performed the experiments; Y.L. and Q.L. analyzed the data; and M.A., H.P. and Q.L. wrote the manuscript. All authors reviewed the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiaopeng Peng, Hongying Pang and Manzar Abbas contributed equally.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-36529-3.
References
- 1.Bibikova T, Gilroy S. Root Hair Development. Plant Growth Regul. 2002;21:383–415. doi: 10.1007/s00344-003-0007-x. [DOI] [Google Scholar]
- 2.Oldroyd GED. Dissecting Symbiosis: Developments in Nod Factor Signal Transduction. Annals of Botany. 2001;87:709–718. doi: 10.1006/anbo.2001.1410. [DOI] [Google Scholar]
- 3.Carol RJ, Dolan L. Building a hair: tip growth in Arabidopsis thaliana root hairs. Philosophical Transactions Biological Sciences. 2002;357:815–821. doi: 10.1098/rstb.2002.1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dolan L, et al. Clonal relationships and cell patterning in the root epidermis of Arabidopsis. Development. 1994;120:2465–2474. [Google Scholar]
- 5.Foreman J, Dolan L. Root Hairs as a Model System for Studying Plant Cell Growth. Annals of Botany. 2001;88:1–7. doi: 10.1006/anbo.2001.1430. [DOI] [Google Scholar]
- 6.Baluska F, et al. Root hair formation: F-actin-dependent tip growth is initiated by local assembly of profilin-supported F-actin meshworks accumulated within expansin-enriched bulges. Developmental Biology. 2000;227:618–632. doi: 10.1006/dbio.2000.9908. [DOI] [PubMed] [Google Scholar]
- 7.Grierson CS, Roberts K, Feldmann KA, Dolan L. The COW1 Locus of Arabidopsis Acts after RHD2, and in Parallel with RHD3 and TIP1, to Determine the Shape, Rate of Elongation, and Number of Root Hairs Produced from Each Site of Hair Formation. Plant Physiology. 1997;115:981–990. doi: 10.1104/pp.115.3.981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Parker JS, Cavell AC, Dolan L, Roberts K, Grierson CS. Genetic Interactions during Root Hair Morphogenesis in Arabidopsis. J Plant Cell. 2000;12:1961–1974. doi: 10.1105/tpc.12.10.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ryan E, Grierson CS, Cavell A, Steer M, Dolan L. TIP1 is Required for Both Tip Growth and Non-Tip Growth in Arabidopsis. New Phytologist. 2010;138:49–58. doi: 10.1046/j.1469-8137.1998.00896.x. [DOI] [Google Scholar]
- 10.Schiefelbein JW, Somerville C. Genetic Control of Root Hair Development in Arabidopsis thaliana. Plant Cell. 1990;2:235–243. doi: 10.1105/tpc.2.3.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Baumberger N, Ringli C, Keller B. The chimeric leucine-rich repeat/extensin cell wall protein LRX1 is required for root hair morphogenesis in Arabidopsis thaliana. Genes Dev. 2001;15:1128–1139. doi: 10.1101/gad.200201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wada T, Okada K. Epidermal cell differentiation in Arabidopsis determined by a Myb homolog. CPC. Science. 1997;277:1113. doi: 10.1126/science.277.5329.1113. [DOI] [PubMed] [Google Scholar]
- 13.Parker JS, Cavell AC, Dolan L, Roberts K, Grierson CS. Genetic Interactions during Root Hair Morphogenesis in Arabidopsis. Plant Cell. 2000;12:1961–1974. doi: 10.1105/tpc.12.10.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Galway ME, Eng RC, Schiefelbein JW, Wasteneys GO. Root hair-specific disruption of cellulose and xyloglucan in AtCslD3 mutants, and factors affecting the post-rupture resumption of mutant root hair growth. Planta. 2011;233:985–999. doi: 10.1007/s00425-011-1355-6. [DOI] [PubMed] [Google Scholar]
- 15.Bernal AJ, et al. Disruption of ATCslD5 results in reduced growth, reduced xylan and homogalacturonan synthase activity and altered xylan occurrence in Arabidopsis. The Plant Journal. 2007;52:791–802. doi: 10.1111/j.1365-313X.2007.03281.x. [DOI] [PubMed] [Google Scholar]
- 16.Hamann T, et al. Global expression analysis of CESA and CSL genes in Arabidopsis. Cellulose. 2004;11:279–286. doi: 10.1023/B:CELL.0000046340.99925.57. [DOI] [Google Scholar]
- 17.Keegstra K, Walton J. Plant science. Beta-glucans–brewer’s bane, dietician’s delight. Science. 2006;311:1872–1873. doi: 10.1126/science.1125938. [DOI] [PubMed] [Google Scholar]
- 18.Vogel J. Unique aspects of the grass cell wall. Current Opinion in Plant Biology. 2008;11:301–307. doi: 10.1016/j.pbi.2008.03.002. [DOI] [PubMed] [Google Scholar]
- 19.Holland N, et al. A comparative analysis of the plant cellulose synthase (CesA) gene family. Plant Physiology. 2000;123:1313–1323. doi: 10.1104/pp.123.4.1313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Arioli T, Burn JE, Betzner AS, Williamson RE. response: How many cellulose synthase-like gene products actually make cellulose? Trends in Plant Science. 1998;3:165–166. doi: 10.1016/S1360-1385(98)01245-X. [DOI] [Google Scholar]
- 21.Cocuron JC, et al. A gene from the cellulose synthase-like C family encodes a beta-1,4 glucan synthase. Proc. Natl. Acad. Sci. USA. 2007;104:8550–8555. doi: 10.1073/pnas.0703133104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Doblin MS, Pettolino F, Bacic A. Evans Review: Plant cell walls: the skeleton of the plant world. Functional Plant Biology. 2010;37:357–381. doi: 10.1071/FP09279. [DOI] [Google Scholar]
- 23.Dwivany FM, et al. TheCellulose-Synthase Like C (CSLC) Family of Barley Includes Members that Are Integral Membrane Proteins Targeted to the Plasma Membrane. Molecular Plant. 2009;2:1025–1039. doi: 10.1093/mp/ssp064. [DOI] [PubMed] [Google Scholar]
- 24.Lerouxel O, Cavalier DM, Liepman AH, Keegstra K. Biosynthesis of plant cell wall polysaccharides — a complex process Olivier Lerouxel 1, David M Cavalier 1, Aaron H Liepman 1,2 and Kenneth Keegstra 1,3. Current Opinion in Plant Biology. 2007;9:621–630. doi: 10.1016/j.pbi.2006.09.009. [DOI] [PubMed] [Google Scholar]
- 25.Richmond TA, Somerville CR. Integrative approaches to determining CSL function. Plant Molecular Biology. 2001;47:131–143. doi: 10.1023/A:1010627314782. [DOI] [PubMed] [Google Scholar]
- 26.Becker JD, Boavida LC, Carneiro J, Haury M, Feijó JA. Transcriptional profiling of Arabidopsis tissues reveals the unique characteristics of the pollen transcriptome. Plant Physiology. 2003;133:713–725. doi: 10.1104/pp.103.028241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Doblin MS, Melis LD, Newbigin E, Bacic A, Read SM. Pollen Tubes of Nicotiana alata Express Two Genes from Different β-Glucan Synthase Families. Plant Physiology. 2001;125:2040–2052. doi: 10.1104/pp.125.4.2040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Honys D, Twell D. Comparative analysis of the Arabidopsis pollen transcriptome. Plant Physiology. 2003;132:640–652. doi: 10.1104/pp.103.020925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Favery B, et al. KOJAK encodes a cellulose synthase-like protein required for root hair cell morphogenesis in Arabidopsis. Genes Dev. 2001;15:79–89. doi: 10.1101/gad.188801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kim CM, et al. OsCslD1, a cellulose synthase-like D1 gene, is required for root hair morphogenesis in rice. Plant Physiology. 2007;143:1220–1230. doi: 10.1104/pp.106.091546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang X, et al. AtCslD3, A Cellulose Synthase-Like Gene Important for Root Hair Growth in Arabidopsis. Plant Physiology. 2001;126:575–586. doi: 10.1104/pp.126.2.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Samuga A, Joshi CP. Cloning and characterization of cellulose synthase‐like gene, PtrCslD2 from developing xylem of aspen trees. Physiologia Plantarum. 2010;120:631–641. doi: 10.1111/j.0031-9317.2004.0271.x. [DOI] [PubMed] [Google Scholar]
- 33.Yin L, et al. The cooperative activities of CSLD2, CSLD3, and CSLD5 are required for normal Arabidopsis development. Plnat Biotech. 2011;4:1024–1037. doi: 10.1093/mp/ssr026. [DOI] [PubMed] [Google Scholar]
- 34.Qi G, et al. Two poplar cellulose synthase-like D genes, PdCslD5 and PdCslD6, are functionally conserved with Arabidopsis. CSLD3. Plant Physiology. 2013;170:1267–1276. doi: 10.1016/j.jplph.2013.04.014. [DOI] [PubMed] [Google Scholar]
- 35.Wang W, et al. Arabidopsis CSLD1 and CSLD4 are required for cellulose deposition and normal growth of pollen tubes. Journal of Experimental Botany. 2011;62:5161–5177. doi: 10.1093/jxb/err221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang X, et al. A Cellulose Synthase-Like Gene Important for Root Hair Growth in Arabidopsis. Plant Physiology. 2001;126:575–586. doi: 10.1104/pp.126.2.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li M, et al. Rice cellulose synthase-like D4 is essential for normal cell-wall biosynthesis and plant growth. Plant Journal. 2009;60:1055–1069. doi: 10.1111/j.1365-313X.2009.04022.x. [DOI] [PubMed] [Google Scholar]
- 38.Li Y, et al. Evolution, gene expression profiling and 3D modeling of CSLD proteins in cotton. BMC Plant Biology. 2017;17:119. doi: 10.1186/s12870-017-1063-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Van Erp H, Walton J. Regulation of the cellulose synthase-like gene family by light in the maize mesocotyl. Planta. 2009;229:885–897. doi: 10.1007/s00425-008-0881-3. [DOI] [PubMed] [Google Scholar]
- 40.Kumar S, Stecher G, Tamura K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Molecular Biology Evolution. 2016;33:1870. doi: 10.1093/molbev/msw054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Saitou NNM, Nei MC, Saitou N, Nei M. The Neighbor-Joining Method-a New Method for Reconstructing Phylogenetic Trees. Mol Biol Evol. 1987;4:406–425. doi: 10.1093/oxfordjournals.molbev.a040454. [DOI] [PubMed] [Google Scholar]
- 42.Tamura K, Nei M, Kumar S. Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc. Natl. Acad. Sci. USA. Proceedings of the National Academy of Sciences. 2004;101:11030–11035. doi: 10.1073/pnas.0404206101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bernal AJ, et al. Functional analysis of the cellulose synthase-like genes CSLD1, CSLD2, and CSLD4 in tip-growing Arabidopsis cells. Plant Physiology. 2008;148:1238–1253. doi: 10.1104/pp.108.121939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zeng W, Keegstra K. AtCslD2 is an integral Golgi membrane protein with its N-terminus facing the cytosol. Planta. 2008;228:823–838. doi: 10.1007/s00425-008-0785-2. [DOI] [PubMed] [Google Scholar]
- 45.Tuskan GA, et al. The Genome of Black Cottonwood, Populus trichocarpa (Torr. & Gray) Science. 2006;313:1596–1604. doi: 10.1126/science.1128691. [DOI] [PubMed] [Google Scholar]
- 46.Wu YC, Rasmussen MD, Bansal MS, Kellis M. Most parsimonious reconciliation in the presence of gene duplication, loss, and deep coalescence using labeled coalescent trees. Genome Research. 2014;24:475. doi: 10.1101/gr.161968.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hunter CT, et al. Cellulose Synthase-Like D1 is integral to normal cell division, expansion, and leaf development in maize. Plant Physiology. 2012;158:708–724. doi: 10.1104/pp.111.188466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cormack RGH. Investigations on the Development of Root Hairs. New Phytologist. 2010;34:30–54. doi: 10.1111/j.1469-8137.1935.tb06826.x. [DOI] [Google Scholar]
- 49.Galway ME, Heckman JW, Jr., Schiefelbein JW. Growth and ultrastructure of Arabidopsis root hairs: therhd3 mutation alters vacuole enlargement and tip growth. Planta. 1997;201:209–218. doi: 10.1007/BF01007706. [DOI] [PubMed] [Google Scholar]
- 50.Volkmann, D. & Peters, P. Structural basis of root hair formation: Early development of trichoblasts and atrichoblasts. (Springer Netherlands, 1995).
- 51.Yang W, et al. Regulation of Meristem Morphogenesis by Cell Wall Synthases in Arabidopsis. Curr. Biol. 2016;26:1404–1415. doi: 10.1016/j.cub.2016.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Li Q, et al. Down-regulation of glycosyltransferase 8D genes in Populus trichocarpa caused reduced mechanical strength and xylan content in wood. Tree Physiology. 2011;31:226–236. doi: 10.1093/treephys/tpr008. [DOI] [PubMed] [Google Scholar]
- 53.Lorenz, W. W., Yu, Y.-S. & Dean, J. F. D. An Improved Method of RNA Isolation from Loblolly Pine (P. taeda L.) and Other Conifer Species. Journal of Visualized Experiments: JoVE, 1751 (2010). [DOI] [PMC free article] [PubMed]
- 54.Clough SJ, Ben AF. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant Journal. 1998;16:735–743. doi: 10.1046/j.1365-313x.1998.00343.x. [DOI] [PubMed] [Google Scholar]
- 55.Song J, Lu S, Chen ZZ, Lourenco R, Chiang VL. Genetic transformation of Populus trichocarpa genotype Nisqually-1: a functional genomic tool for woody plants. Plant Cell Physiology. 2006;47:1582–1589. doi: 10.1093/pcp/pcl018. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.