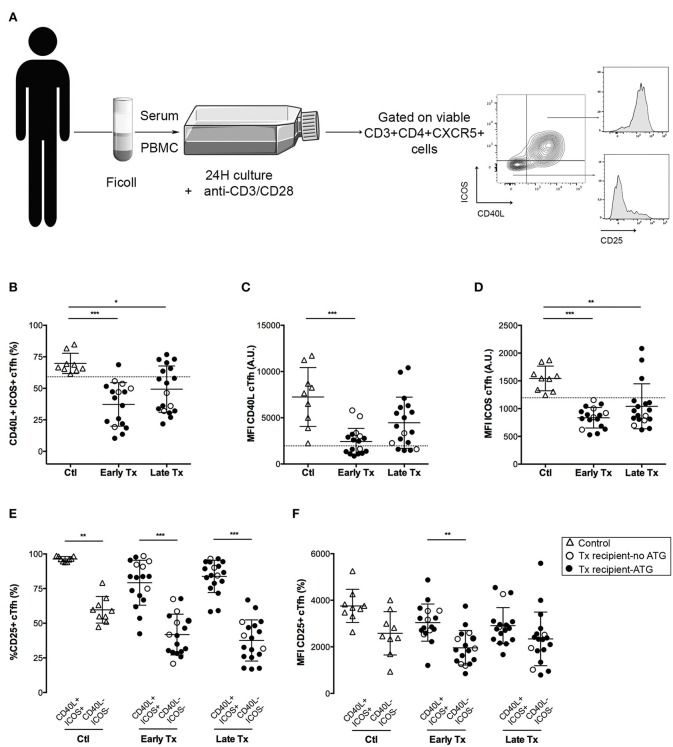
Figure 2.
Comparison of the functional profile of cTfh of controls and transplants patients. (A) Graphical summary of the procedure used to evaluate the functionality of cTfh. PBMC of patients were collected just before vaccination. The level of expression of CD40L ICOS and CD25 was measured by flow cytometry on the surface of cTfh, after 24 h stimulation with anti-CD3/CD28 microbeads in patient's own serum. (B) The percentage of CD40L+ICOS+ cTfh was enumerated by flow cytometry after 24 h of in vitro stimulation in healthy volunteers (n = 9; controls, Ctl; open triangles), and transplant patients (n = 36, circles). The nature of induction immunosuppressive therapy received by transplant patient is indicated. Transplant patients were distributed into two groups according to the time elapsed since transplantation: (i) 6–12 months: early post-transplantation (Early Tx; n = 18), and (ii) >12–72 months: late post-transplantation (Late Tx; n = 18). Dotted line indicates the lower value observed in control patients. (C,D) The level of expression of CD40L (C) and ICOS (D), as reflected by mean fluorescence intensity (MFI), was measured by flow cytometry after 24 h of in vitro stimulation of cTfh and compared between the 3 groups. (E) The percentage of CD25-positive cells was compared between CD40L+ICOS+ and CD40L−ICOS− cTfh subsets in the 3 groups. (F) The level of expression of CD25 was compared between CD40L+ICOS+ and CD40L−ICOS− cTfh subsets in the three groups. Each symbol represents a patient, mean ± SD is indicated. *p < 0.05; **p < 0.01; ***p < 0.001; ANOVA, Dunn's multiple comparisons test.