Abstract
Efflux pumps are membrane protein complexes conserved in all living organisms. Beyond being involved in antibiotic extrusion in several bacteria, efflux pumps are emerging as relevant players in pathogen-host interactions. We have investigated on the possible role of the efflux pump network in Shigella flexneri, the etiological agent of bacillary dysentery. We have found that S. flexneri has retained 14 of the 20 pumps characterized in Escherichia coli and that their expression is differentially modulated during the intracellular life of Shigella. In particular, the emrKY operon, encoding an efflux pump of the Major Facilitator Superfamily, is specifically and highly induced in Shigella-infected U937 macrophage-like cells and is activated in response to a combination of high K+ and acidic pH, which are sensed by the EvgS/EvgA two-component system. Notably, we show that following S. flexneri infection, macrophage cytosol undergoes a mild reduction of intracellular pH, permitting EvgA to trigger the emrKY activation. Finally, we present data suggesting that EmrKY is required for the survival of Shigella in the harsh macrophage environment, highlighting for the first time the key role of an efflux pump during the Shigella invasive process.
Subject terms: Bacteriology, Pathogens
Introduction
Efflux pumps (EPs) are present in all living organisms and represent an important and consistent group of transporter proteins, which contribute to the resistance to compounds used for treating different diseases1,2. In Gram negative bacteria, EPs either form tripartite complexes able to traverse both membranes, including the inner membrane, a membrane fusion protein and an outer membrane protein, or are present as single-component efflux transporter in the inner membrane2. According to their sequence similarity, composition, transport function, energy source and substrates, EPs are grouped into five families: the ATP binding cassette (ABC) superfamily, the major facilitator superfamily (MFS), the multidrug and toxic compound extrusion (MATE) family, the small multidrug resistance (SMR) family and the resistance nodulation division (RND) family1. The importance of EPs has been associated with their ability to extrude a wide range of antibiotics resulting in the emergence of multidrug resistance in many bacteria, including pathogens1,2.
In the last years, several studies have identified numerous functions for EPs that go beyond antibiotic extrusion: these functions range from bacterial interactions with plant or animal hosts, to detoxification of metabolic intermediates and maintenance of cellular homeostasis3,4. An interesting aspect concerns the involvement of EPs in the virulence of several bacterial pathogens3,5. In enteric pathogens such as Salmonella Typhimurium, Listeria monocytogenes, and Vibrio cholera it has been shown that EPs are relevant virulence factors contributing to persistence and replication in a bile rich environment6–8. Moreover, in Salmonella EPs are critical for the invasion and survival within macrophages and intestinal epithelial cells and contribute to the different steps of the pathogenicity process9–11, while in Vibrio cholera, besides favouring the colonization of the intestine, EPs are also required for the full expression of the major virulence determinants12. EPs contribute also to the pathogenicity of Pseudomonas aeruginosa and Stenotrophomonas maltophilia, where they play a significant role in biofilm formation13–15, and of Campylobacter jejuni and Neisseria gonorrhea, where they are involved in the colonization of the host epithelia16,17.
Shigella is an intracellular pathogen responsible of a life-threatening enteric syndrome in humans18. Invasion of the colonic epithelium by Shigella is the result of a complex multistep process. After ingestion, Shigella gains access to the intestinal mucosa by promoting its uptake into M cells in Peyer’s patches. The bacteria are then released into an intraepithelial pocket and invade resident macrophages, where they multiply and induce rapid cell death. Once released from dying macrophages, invasive bacteria can finally infect the neighboring enterocytes, where they actively replicate and, without any extracellular steps, disseminate from cell-to-cell, causing severe damage and inflammatory destruction of the colonic mucosa19.
Shigella shares strong homology with its commensal ancestor, Escherichia coli, and has derived repeatedly from different branches of the E. coli tree by convergent evolution involving both gain and loss of genes20–22. In particular, the acquisition by horizontal gene transfer of a large plasmid (pINV) carrying genes for a Type III secretion system and its effectors has been the crucial event towards pathogenic lifestyle19. This process has been paralleled by the loss of several chromosomal genes unnecessary or deleterious for the invasive process23–26.
Very few data are available on the role that EPs might have in the lifestyle of Shigella and these are limited to the MdtJI and AcrAB EPs27,28. Concerning MdtJI, it has been shown that its expression is higher in Shigella compared to E. coli and is positively controlled by spermidine and by the VirF protein, the major regulator of the Shigella invasive genes29. On the basis of its ability to secrete putrescine, it has been proposed that, maintaining the spermidine at an optimal level, MdtJI might contribute to the Shigella survival within the host cells27,30. In the case of AcrAB it has been shown that also in Shigella it contributes to bile salts resistance28.
Taking into account the emerging role of EPs in bacterial virulence and the importance of Shigella as human pathogen we asked whether EPs might be involved in the Shigella mediated invasive process. To this end, we searched for the EPs conserved in the Shigella flexneri genome and then investigated whether the expression of their encoding genes was influenced by the host cell environment. Remarkably, we found the emrKY genes, enconding the MSF efflux pump EmrKY, to be strongly activated in macrophages. The up-regulation of the emrKY expression in the macrophage environment led us to investigate further the regulatory mechanism underlying its specific activation and determine its potential role during Shigella intracellular life.
Results
In silico identification of the Shigella flexneri EP encoding genes
E. coli synthesizes 20 functional EPs31,32. By using the NCBI genome BLAST, homologs for each of the 20 E. coli EP encoding operons were searched in the genome of S. flexneri M90T, a strain widely used in laboratory to analyze Shigella-host interactions33. As shown in Table 1, only 14 out of the 20 genetic systems described in E. coli are conserved in S. flexneri. A more detailed inspection of the rearrangement that have caused the silencing of six EP encoding loci in M90T, revealed that three of them (cusCFBA, mdtABCD, mdtEF) were disrupted by the insertion of IS elements within the coding or regulatory regions, while the remaining three were completely lost (acrEF, yceE and yjiO). The 14 EP encoding genes identified in M90T strain are conserved in all the other S. flexneri genomes deposited in NCBI database, suggesting a potential involvement in Shigella virulence.
Table 1.
Analysis of the efflux pump encoding genes present in the S. flexneri M90T genome.
| Family | E. coli | S. flexneri |
|---|---|---|
| ABC | macAB | + |
| MFS | bcr | + |
| emrAB | + | |
| emrD | + | |
| emrKY | + | |
| Fsr | + | |
| mdfA | + | |
| mdtG (yceE)* | − | |
| mdtH (yceL)* | + | |
| mdtL(yidY)* | + | |
| mdtM (yjiO)* | − | |
| RND | acrAB | + |
| acrD | + | |
| acrEF | − | |
| cusB | NF | |
| mdtABC | NF | |
| mdtEF | NF | |
| SMR | emrE | + |
| mdtJI | + | |
| MATE | mdtK (ydhE)* | + |
Functional efflux pumps conserved or silenced in the S. flexneri genome. The 20 genetic systems encoding functional efflux pumps in E. coli have been selected according to Kobayashi et al.32. The comparative analysis between the genome of E. coli K-12 and S. flexneri M90T strain was carried out using NCBI BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi). The nucleotide sequences of each efflux pump encoding gene were obtained from EcoCyc (https://ecocyc.org).
+Efflux pump encoding genes present in the S. flexneri genome (sequence homology ≥92%).
−Not present in the S. flexneri genome.
NF: pseudogene, i.e. not Functional due to gene disruption.
*The denomination in brackets corresponds to the former one adopted in E. coli and is still in use in S. flexneri.
Expression profile of the Shigella EP encoding genes during infection of host cells
Transcription profile of the S. flexneri EP genes was assessed during infection of human cells. Shigella pathogenicity process is characterized by the ability of bacteria to overcome the macrophage attack and subsequently to invade the epithelial cells. Thus, we first analyzed the expression profile of the EP encoding genes in Shigella-infecting human U937 monoblasts differentiated into macrophage-like cells. As shown in Fig. 1A, expression of some EP genes increased when Shigella invades macrophages as compared to growth in RPMI medium. In particular, transcription of the emrK gene was induced more than 30-fold with its expression steadily increasing during infection. Similarly, transcript levels of emrA, emrD and emrE genes increased significantly during Shigella infection (up to 2, 8 and 10- fold, respectively) following similar kinetics. Expression of mdtJ gene was also induced, while in this case, maximum level was reached almost immediately upon entry into the macrophage (up to 7-fold). Conversely, expression of acrA gene was found to be down-regulated in the macrophage environment, whereas expression of remaining EP encoding genes was not significantly changed going from growth into RPMI medium to macrophage.
Figure 1.
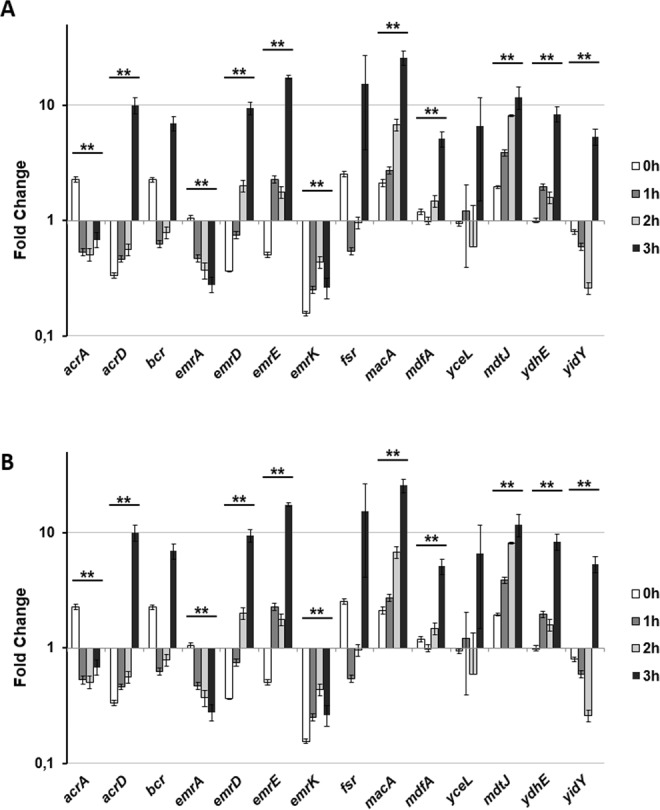
Relative efflux pumps encoding gene transcription during S. flexneri infection of (A) U937 and (B) Caco-2. Quantitative analysis of 14 efflux pump transcripts was performed by means of Q-Real Time PCR assay. Total RNA was extracted from intracellular S. flexneri M90T bacteria at various time points p.i., from 0 h (corresponding to bacterial adhesion to the target cells, see MM) up to 3 h. Each infection was repeated three times and at least three wells were run for each sample. The x axis indicates the expression fold-change (RQ value) for each gene on a logarithmic scale. The results are shown relative to the expression of each gene in bacteria grown in RPMI (A) or DMEM (B) set to 1.00. Statistical significance was determined by a one-tailed ANOVA, and p values are as follows: *p < 0.05, **p < 0.01. Error bars represent SD.
Expression level of the S. flexneri EP genes was also analysed during infection of Caco-2 epithelial cell line. Surprisingly, expression of both emrK and emrA genes was down-regulated, in contrast to what had been observed in macrophages (Fig. 1B). Expression of the macA gene was strongly induced in Caco-2 cells, whereas it had been found unaltered inside macrophages. Last, a series of genes showed the same pattern of expression in both cell lines with the mdtJ and ydhE genes being up-regulated and acrA gene being down-regulated. Also, emrD and emrE are induced in Caco-2 cells although at later time during infection.
In conclusion, transcript analyses indicated that expression of a subset of the 14 conserved EP-encoding genes varies depending upon the host cell type Shigella is growing in. Entry and subsequent killing of macrophages represents the rate limiting step of the Shigella invasive process34. Given the cell specific expression of the emrK gene, belonging to the emrKY operon, we focused our subsequent efforts on the regulation and potential role of this EP within macrophages.
Expression of emrKY in response to environmental stimuli
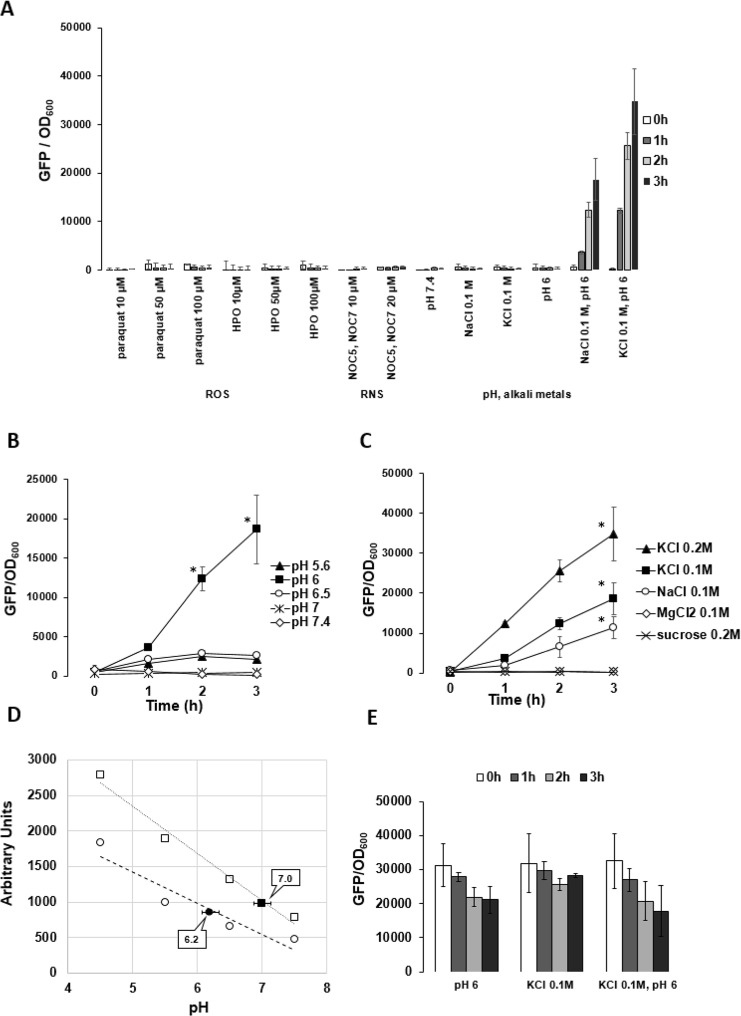
We aimed at identifying the stimuli causing emrKY induction in macrophages. Shigella infecting a macrophage faces oxidative stress, nitrosative stress and acidic environment. Therefore, we used paraquat and hydrogen peroxide (HPO) to induce the production of reactive oxygen species (ROS), and the NO-amine complexes NOC-5 and NOC-7 to induce the formation of reactive nitrogen species (RNS), while acidic conditions were achieved by adding HCl to M9 complete medium. We also evaluated whether an unbalanced composition in alkali metals (NaCl or KCl) acted as a stimulus. To efficiently monitor emrKY expression, we cloned the gfp gene under the control of the emrKY promoter and transformed the resulting plasmid pZemrK-gfp into M90T. As shown in Fig. 2A when paraquat, HPO, NOC-5 or NOC 7 was added to a culture of M90T pZemrK-gfp at OD600 0.2, expression of GFP remained close to the basal level. Similarly, neither medium acidification nor the presence of alkali metals modified the expression level of the reporter gene (Fig. 2A). In contrast, a very high level of GFP was observed when the culture was grown at pH 6.0 in M9 complete medium supplemented with KCl or NaCl (0.1 M). Considering that M9 medium already contains 0.093 M NaCl and only 0.022 M KCl, a lower final concentration of KCl is sufficient to obtain a more effective induction (Fig. 2A).
Figure 2.
emrKY expression in S. flexneri is modulated by specific stimuli both outside and inside U937. (A) Expression of emrKY in response to the indicated stimuli in strain M90T pZemrK-gfp grown in LB or M9 complete medium. (B) Expression of emrKY in response to different pH values (from pH 5.6 to 7.4) in M9 complete medium supplemented with 0.1 M KCl. (C) Expression of emrKY in response to increasing concentration of KCl, NaCl, MgCl2 or sucrose added to M9 complete medium at pH 6. The addition of 0.05 M MgCl2 or 0.1 M sucrose (not shown) has the same effect as 0.1 M MgCl2 or 0.2 M sucrose, respectively. In A,B and C the expression of emrKY was measured as a function of GFP production in M90T pZermK-gfp strain. (D) Intracellular pH values (filled symbols) of mock (square) and M90T infected U937 cells (circle) at 3 hours p.i. pH was measured by loading cells with green pHrodo dye and quantified on the relative standard curves obtained with four different calibrated pH buffers. Fluorescence intensity was read on the Victor multilabel counter (Perkin Elmers). (E) Expression of mdtJI in response to the indicated stimuli measured as a function of GFP production in M90T pZmdtJ-gfp strain grown in M9 complete medium. GFP production was evaluated by reading fluorescence intensity on Infinite 200 (TECAN) every 15 minutes throughout 3 hours. GFP values were normalized to the bacterial cell concentration and the results shown are the mean of three independent experiments. Three independent experiments were carried out and statistical significance was determined by a one-tailed ANOVA, and p values are as follows: *p < 0.05. Error bars represent SD.
Interestingly, when the pH value of the M9 medium was adjusted at different pH values from pH 5.6 to pH 7.4, stimulatory effect on the emrKY promoter was observed only at pH 6 (Fig. 2B). The addition of KCl (0.2 M) to the M9 medium yielded a two-fold enhancement in the emrKY expression (Fig. 2C). The role played by high concentrations of K+ and Na+ ions is evidenced by observing that no induction occurs when another chloride salt, MgCl2 (0.05 M and 0.1 M) is used, thus excluding a potential involvement of Cl- ions in agreement with previous observations35. Last, the fact that addition of MgCl2 (0.1 M) or sucrose (0.1 M or 0.2 M) has no effect on emrKY induction (Fig. 2C), indicated that the emrKY up-regulation, observed with KCl or NaCl, was not due to an overall increase in osmolality.
To test whether pH 6 and high concentration of K+ ions recapitulated part of the physico-chemical environment met by Shigella in macrophages, we measured the macrophage intracellular pH (pHi) during bacterial infection. The data shown in Fig. 2D demonstrate that, upon Shigella infection U937 pHi value dropped to 6.2, whereas mock infected cells maintained a neutral pHi value. The values reported refer to 3 hours post infection (p.i.), when the infection efficiency was at its peak, ranging from 60 to 70% in the various infection experiments.
Interestingly, mdtJ gene, whose expression is induced in both macrophage and epithelial cells, was not modified in M9 complete medium supplemented with 0.1 M KCl and set at pH 6 (Fig. 2E), leading some credence to the notion that EmrKY carries on specific tasks in the macrophage compartment.
Finally, we asked whether in the macrophage environment the high expression of emrKY was linked to a reduced expression of acrAB, encoding the major E. coli EP31. Indeed, it has been suggested that the loss of a given EP might be compensated by an increased synthesis of another EP that could fulfill, at least in part, the same function36,37. We generated M90T derivatives lacking the EmrKY (M90T ΔermKY) or AcrAB (M90T ΔacrAB) systems or overproducing the AcrAB pump (M90T pACacrAB). U937 cells were infected with M90T or its derivatives, and the acrA or emrK gene transcripts analyzed at different time points p.i. As reported in Fig. S1, induction of emrK gene was influenced neither by the absence nor by the overexpression of acrAB genes. Conversely, presence or absence of ermKY has no effect on the acrA transcription level.
Shigella emrKY is regulated by EvgA both in laboratory conditions and within host cells
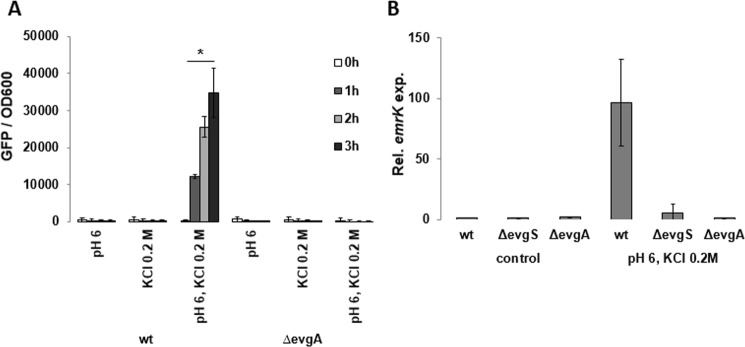
In E. coli, the emrKY operon is under the control of the EvgS/EvgA two-component system38,39 in response to mild acidic conditions and high concentrations of alkali metals35,40. Therefore, we tested whether this was also the case in Shigella both in laboratory conditions and in host cell during infection. We introduced a deletion covering the evgA gene in the M90T and monitored the emrK-gfp expression. Lack of evgA prevented emrK-gfp fusion expression under inducing conditions (pH 6.0, 0.2 M KCl) (Fig. 3A). Consistently, no emrK transcrips was evidenced in strains lacking either EvgA (M90T ΔevgA) or EvgS (M90T ΔevgS) grown in inducing conditions (Fig. 3B).
Figure 3.
Regulation of emrKY expression in S. flexneri. (A) Expression of emrKY was monitored as a function of GFP production in S. flexneri M90T and its derivative lacking the EvgA regulator (M90TΔevgA), both strains carrying the pZemrK-gfp plasmid. Strains were grown in M9 complete medium modified as indicated. GFP production was monitored by reading the fluorescence intensity on Infinite 200 (TECAN) every 15 mins throughout 3 h. (B) Quantitative analysis of the emrK transcript was performed by qRT-PCR using RNA extracted from S. flexneri M90T and its derivatives lacking the EvgA (M90T ΔevgA) or EvgS (M90T ΔevgS) proteins. All S. flexneri strains were grown at 37 °C in M9 complete medium at pH 7.4 (control) or in M9 at pH 6 supplemented with 0.2 M KCl (induction conditions). The experiment was repeated three times and at least three wells were run for each sample. The results are shown relative to the emrK transcript levels in M90T grown in control conditions set to 1.00. Statistical significance was determined by a one-tailed ANOVA, and p values are as follows: *p < 0.05. Error bars represent SD.
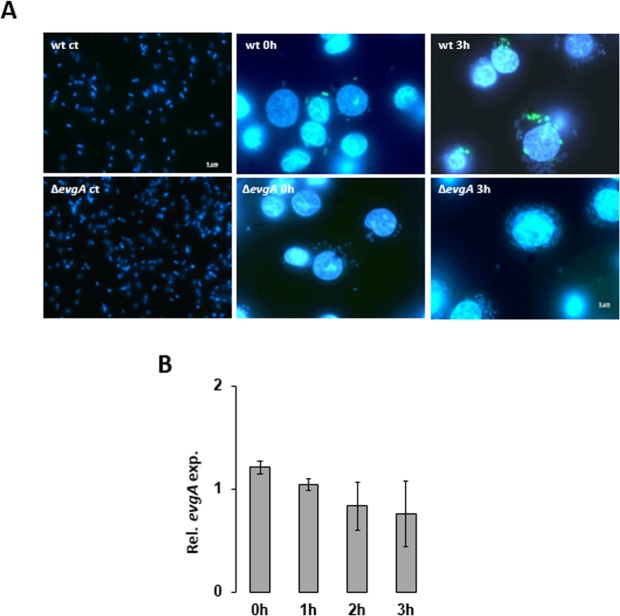
To test whether induction of emrKY is mediated by EvgA in the macrophage environment, we infected U937 cells with M90T or its ΔevgA derivative, both carrying the pZemrK-gfp plasmid. As shown in Fig. 4A expression of emrK-gfp fusion occurred very early in the M90T strain during the infection process and was maintained throughout the infection period. In contrast, the M90T ΔevgA mutant was unable to activate the emrKY promoter as indicated by the absence of intracellular green bacteria at all the time points analyzed. Importantly, evgA expression was no significantly modified in macrophage environment (Fig. 4B).
Figure 4.
EmrKY induction within macrophages is under the control of EvgA. (A) Fluorescence microscopy images of S. flexneri M90T and M90T ΔevgA strains (WT ct and ΔevgA ct), harboring the emrK-gfp fusion, grown in laboratory conditions (first column) and during the infection of U937 cells at 0 h (second column) and 3 h p.i. (third column). Bacteria and macrophage DNA was stained with DAPI. Individual pictures of the same field, taken with a DC camera, were merged using a Leica Microsystem Imaging Equipment. (B) Quantitative analysis of the evgA transcript from M90T in U937 cells at the indicated time points. The experiment was repeated three times and at least three wells were run for each sample. The results shown are relative to the evgA transcript levels in M90T grown in M9 (pH 7.4) set to 1.00. Statistical significance was analyzed by a one-tailed ANOVA. Error bars represent SD.
Altogether, these data demonstrate that the Shigella emrKY operon is under the positive control of the EvgS/EvgA two-component system in response to mild acidic and high K+ concentration conditions met in macrophage environment.
EmrKY contributes to Shigella fitness in macrophages
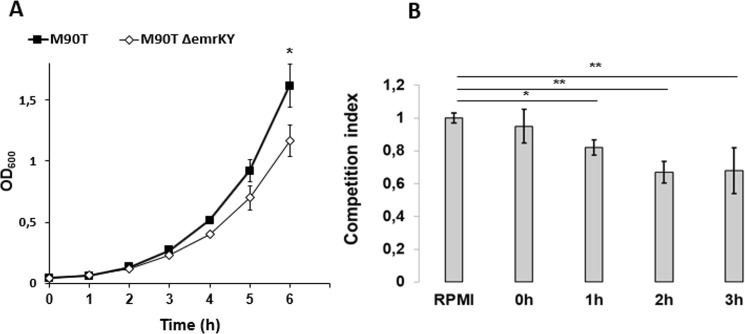
Since induction of emrKY genes is part of the bacterial response to peculiar conditions (moderate acid conditions and unbalanced alkali metals) it is reasonable that EmrKY can confer a growth advantage in those same conditions and can be required to better face the hostile environment. To verify this hypothesis, we compared the growth properties of S. flexneri M90T and its ΔemrKY derivative at pH 6.0 in M9 complete medium supplemented with 0.2 M KCl. Figure 5A shows that, actually, lack of EmrKY negatively affected the bacterial growth, suggesting that induction of this EP contributes to resist to this adverse environment (Fig. 5A). Following the same line, we asked whether EmrKY pump enhanced survival of Shigella in macrophages. To this end, we set up a competitive assay where U937 cells were co-infected with the M90T and its ΔemrKY derivative. Intracellular bacteria were recovered, counted and the competitive index was determined at various time points after infection (see Methods). The data reported in Fig. 5B clearly demonstrate that the M90T strain exhibited an intracellular growth advantage compared to its ΔemrKY derivative. Indeed, the competitive index related to the ΔemrKY strain decreased progressively reaching 0.67 at two hours p.i., a value which was not significantly affected by an additional incubation of 1 hour. This indicates that increasing expression of emrKY (Fig. 1A) triggered by U937 cytoplasm acidification (Fig. 2D) is functional for S. flexneri survival and multiplication within macrophage environment. In conclusion, these data support the notion that the massive presence of EmrKY during Shigella infection of macrophages could represent one of the multiple bacterial strategies to overcome host defenses.
Figure 5.
EmrKY confers a growth advantage to Shigella and improves its survival inside macrophages. (A) Growth curves (OD600) of M90T and its derivative lacking the EmrKY pump (M90TΔemrKY), in M9 complete medium pH 6.0 supplemented with 0.2 M KCl. (B) Competitive indices (C.Is), at the indicated time points p.i., of M90T ΔemrKY relative to M90T wt strain as normalized by their ratio in the inoculum (RPMI). M90T and M90TΔemrKY were used to coinfect U937 cells at MOI 100. The data presented are the means of three independent experiments. Statistical significance was determined by t test, and p values are as follows: *p < 0.05. Error bars represent SD.
Discussion
In the present study, we report that many of the S. flexneri EP genes have their expression differentially modulated during the intracellular life. Notably, our findings highlight how the expression of some EPs is regulated by specific cell context. A most telling case is that of the EmrKY pump, the genes of which are highly and specifically activated in Shigella-infecting U937 macrophage-like cells. We demonstrated that the Shigella emrK gene is induced in response to a combination of high K+ and mild acidic pH. Both of these physico-chemical features were found to be sensed and transduced by the EvgS/EvgA two-component system, both in laboratory conditions and in infected macrophages. Last, evidence was obtained showing that EmrKY is functional to Shigella to better overcome the macrophage obstacles, pointing out the pivotal role of an EP in Shigella invasive process.
The pathogenesis of Shigella is extremely complex and is based on its capability to cross the intestinal barrier, to kill resident macrophages and invade enterocytes, where intracellular replication and dissemination occurs19. Shigella shares a high genome homology with the commensal E. coli and on the basis of phylogenetic analysis Shigella has been included in E. coli species20,21. However, compared to E. coli, Shigella genome is extremely dynamic and subjected to an extensive gene decay process, which has targeted widely diverse housekeeping functions such as catabolic pathways or mobility24. Here we report that gene decay has targeted EP genes as only 14 out of 20 were conserved. In turn, this advocate for these 14 to have a role in Shigella physiology and adaptation, possibly in relation with its intracellular lifestyle in the human host.
Besides antibiotic resistance, bacterial EPs likely take part in several relevant processes of the microbial physiology, including bacteria-host interactions1,3,5. Involvement of EPs has been described in several pathogenic bacteria where they contribute to host colonization, intracellular survival, resistance to stress and biofilm formation3–5. In Shigella, the role of EPs in the intracellular life have been very poorly investigated. In the case of MdtJI, an EP involved in polyamine trafficking, it has been hypothesized that its activity in secreting polyamines might be associated with an increased survival within the host cells27. In the present study, transcription profile analysis of EP genes during Shigella infection showed that they are differentially expressed according to the cell environment. Of particular interest was that some of them were found up-regulated upon U937 macrophage infection. Escape from macrophage being considered as the rate limiting step in Shigella infection, the reprogramming of EP synthesis might be crucial within the overall invasive process.
EmrKY stood out as the most up-regulated EPs in Shigella-infecting macrophages. EmrKY is an efflux pump composed by an inner membrane subunit and a periplasmic adaptor protein belonging to the MF superfamily1. The structure of the EmrKY pump has not yet been solved. Comparative studies suggest that EmrKY shares homology with another MSF efflux pump, EmrAB41,42. In contrast to EmrAB, EmrKY does not contribute to antibiotic resistance but when overexpressed confers the cell resistance to bile salts31. Finally, loss of EmrKY reduces the ability of E. coli cells to form biofilm43 and increases their sensibility to DNA damaging compounds44.
In E. coli, the emrKY operon is regulated in response to K+ and mild acidic pH via the EvgS/EvgA two-component system35. Here we found the same applied to the Shigella emrKY operon. Importantly, this very regulatory circuit also takes place in the host as mutants defective in evgA were unable to activate transcription of emrK inside macrophages.
Shigella emrKY operon was found to be regulated in an opposite manner in macrophage and epithelial cells, as it was induced in the first case and repressed in the second. Taking into account the stimuli able to upregulate the emrKY operon, it can be envisaged that infected macrophage and epithelial cell environment differs markedly in some features. For what it concerns K+, it is well known that one important function of animal cells is to maintain an ion gradient across the plasma membrane and, for example, the Na+- K+ pump ensures the maintenance of high K+ and low Na+ intracellular concentration45. Thus, the relative high cytosolic levels of K+ could mirror the high K+ concentration required to induce the emrKY operon in laboratory conditions. However, this doesn’t explain the opposite expression profile of emrKY within macrophage and epithelial cells, as such an ion gradient is present in both cell types. Another explanation might stem from considering the pH factor. Shigella could face completely different cytosolic pH conditions in macrophage and epithelial cells. Indeed, Lucchini and collaborators46, analyzing genomic expression of S. flexneri during the infection of macrophage and epithelial cells, found that genes involved in acid resistance were strongly induced in U937 cells but not in HeLa cells, suggesting that, within macrophages, Shigella is exposed to an acidic cytosol. Accordingly, we were able to determine that, upon infection, U937 intracellular pH sensibly decreased. Hence, as a working hypothesis, we propose that emrKY regulation is finely tuned to respond to physico-chemical environment met by the bacterium when invading macrophages. Whether the repression observed in epithelial cells results from an active, cell-specific transcriptional repressing mechanism, i.e. actual induction of a negative regulator that respond to conditions met in epithelial cell, will pave the way for our next molecular investigations.
Extent of emrKY operon induction observed in the macrophage environment is very high (32-fold) and is in agreement with previous transcriptomic analysis46, suggesting a potential involvement of this EP in Shigella virulence. The results obtained in infection competition assays clearly indicated that EmrKY helps Shigella to better survive inside macrophages and, possibly, to achieve a successful host infection. Why and how can EmrKY contribute to Shigella survival in the macrophage environment? It has been reported that, in E. coli, mutations affecting emrK or emrY genes cause a hypersensitive phenotype to the lethal effect of several drugs, including mitomycin C, suggesting that EmrKY might be involved in safeguarding cells from DNA damaging agents44. Does the importance of EmrKY for multiplication in macrophages associate with its capacity to eliminate DNA damaging compound produced by the infected cells? This is one of many attractive possibilities our next study will focus on.
In conclusion, our molecular and cellular investigation allowed us to identify EmrKY as a new important actor within Shigella pathogenicity program. The present study is the first dealing with and demonstrating the involvement of EPs in Shigella invasive process and, undoubtedly, open new perspectives in the analysis of the complex interactions of Shigella with its host.
Methods
Construction of strains and plasmids
Bacterial strains and plasmids used in this study are listed in Table S1. M90T is a S. flexneri serotype 5 strain (GenBank CM001474.1). E. coli DH10b has been used as recipient in cloning experiments47. Strains M90T, M90T ∆acrAB, M90T ∆evgA and M90T ∆evgS were obtained using the one-step method of gene inactivation48. pZemrK-gfp and pZmdtJ-gfp plasmids were obtained by cloning the emrKY or mdtJI regulatory regions upstream the gfp gene into pZEP08 vector49. pACacrAB has been obtained by cloning the acrAB operon under the control of the ptac promoter carried by pGIP7 vector50. Mutations and constructs obtained in this study were verified by DNA sequencing (BioFab, Rome). Sequences of the oligonucleotides, designed on the basis of M90T genome, are reported in Table S2. PCR reactions were routinely performed using DreamTaq DNA polymerase (Fermentas) or the Pfu Taq DNA polymerase (Fermentas).
Media, chemicals and growth conditions
Unless otherwise indicated, bacteria were grown aerobically in LB medium at 37 °C. When required cells were grown in M9 minimal medium supplemented with 10 μg/ml thiamine, 0.2% glucose, 0.5% casamino acids, 10 μg/ml nicotinic acid, 1 mM MgSO4 and 0.2 mM CaCl2 (M9 complete medium). Solid media contained 1.6% agar. Congo red was added (0.01% final concentration) to Trypticase soy agar to monitor the expression of the virulence phenotype prior infection assays. Antibiotics were used at the following concentrations: ampicillin 50 μg/ml; cloramphenicol 25 μg/ml; kanamycin 30 μg/ml; streptomycin 10 μg/ml. To assay emrKY and mdtJI promoter activity in response to different compounds, M90T strains carrying the gfp fusions (M90T pZemrK-gfp, M90T pZmdtJ-gfp) were grown in LB or M9 complete media at 37 °C as indicated. The overnight cultures were then washed and diluted 1:100 in the same media. The different compounds were added to cultures grown up to OD600 0.2 in a final volume of 150 µl in each well of a 96-well plate. To induce oxidative stress paraquat (Sigma-Aldrich) or hydrogen peroxide (HPO) (Sigma-Aldrich), at final concentrations of 50 µM, 100 µM and 150 µM, were added to cultures grown in LB. Nitrosative stress was induced by supplementing LB cultures with NOC-5 (3-(Aminopropyl)-1-hydroxy-3-isopropyl-2-oxo-1-triazene) or NOC-7 (3-(2-Hydroxy-1-methyl-2-nitrosohydrazino)-N-methyl-1-propanamine) at final concentrations of 10 µM and 20 µM. To test the effect of alkali metals and osmolality, NaCl (0.1 M), KCl (0.1 or 0.2 M), sucrose (0.1 M or 0.2 M) or MgCl2 (0.05 or 0.1 M) were added to bacterial cultures grown in M9 complete medium.
To test the effect of pH variation, HCl or NaOH was added to the M9 complete medium to obtain the desired pH value. The fluorescence intensity of M90T strains harboring the gfp fusions was measured on Infinite 200 (TECAN). Fluorescence and OD600 were detected every 15 minutes throughout 3 hours. Fluorescence values were divided by the absorbance at 600 nm in order to normalize to bacterial cell concentration.
Cell cultures and infections
Infection experiments were performed by using both U937 and Caco-2 cell lines. Human U937 cells (American Type Culture Collection, Manassas,VA) were grown in RF10 (RPMI 1640 [GIBCO] medium containing 10% heat-inactivated FBS [fetal bovine serum, Euroclone], 2 mM L-glutamine and PS [0.05 I.U./ml penicillin and 0.05 I.U./ml streptomycin]) at 37 °C in a humidified 5% CO2 atmosphere. For bacterial infection, cells were seeded in 6-well tissue culture plates (Falcon), at a density of 1.5 × 106 cells/well, in RF10 supplemented with phorbol myristate acetate (PMA, 80 nM; Sigma) to induce macrophage differentiation. After two days, PMA containing medium was removed and cells left for further 4 days in RF10. Two hours before bacterial infection, RF10 was replaced with fresh RPMI containing only L-glutamine. Human Caco-2 epithelial cells (American Type Culture Collection, Manassas,VA) were grown in Dulbecco modified essential medium (DMEM) (GIBCO) containing 10% heat-inactivated FBS, 2 mM L-glutamine and PS, referred to as DF10, at 37 °C in a humidified 5% CO2 atmosphere. For bacterial infection, cells were seeded in 6-well tissue culture plates (Falcon), at a density of 4 × 105 cells/well, in DF10. After 48 hours cells were serum-starved over-night in DMEM supplemented with 0.5% FBS, PS and L-glutamine (DF 0.5). Two hours before bacterial infection, DF 0.5 was replaced with fresh DMEM containing only L-glutamine. Both cell lines were infected at a MOI of 100. After addition of bacteria, plates were centrifuged for 15 min at 750 × g and incubated 30 min (U937) or 45 min (Caco-2) at 37 °C under 5% CO2 atmosphere to allow bacterial entry. Thereafter extracellular bacteria were removed by extensive washing with phosphate-buffered saline (PBS). This point was taken as time zero (T0). Fresh medium (RPMI 1640 or DMEM) containing gentamicin (100 μg/ml) was added to each plate to kill extracellular bacteria, and infected cells were incubated at 37 °C for up to 3 h. To visualize GFP expression in M90T and M90T ∆evgA harboring emrK-gfp fusion during the infection process, samples were fixed with 4% paraformaldehyde for 30 min and the DNA of bacteria and macrophages was stained with DAPI. Cells were examined using a Leica DMRE fluorescence microscope equipped with 20×, 40× and 100 × lenses. Single images were recorded on a Leica DF 420 camera and processed using the Qwin software (Leica Microsystem).
RNA isolation and Real Time PCR
To monitor the expression of emrK and/or evgA during standard bacterial growth, M90T, M90T ΔevgA or M90T ΔevgS were grown in M9 complete medium until OD600 0.2. Cultures were then supplemented with KCl and/or HCl to obtain pH 6 and RNA was extracted from strains grown until OD600 0.5 as previously described29. To monitor gene expression during host cell infection, one 6-well tissue culture plate was considered for each time point to maximize the yield of intracellular bacteria. RNA was extracted from intracellular bacteria recovered after lysing infected cells with 1% Triton X-100 (Sigma) for 5 min. The lysate was diluted 1:2 with PBS to decrease Triton concentration before proceeding with RNA extraction. Two μg of total RNA were treated with DNAse I and then retro-transcribed using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems) following the manufacturer’s instruction. qRT-PCR was performed in a 30 μl reaction mix containing 2 μl cDNA using Power SYBR Green PCR Master Mix (Applied Biosystems) on a 7300 Real-Time PCR System (Applied Biosystems). At least three wells were run for each sample. Relative quantification was performed using the comparative cycle threshold (2−ΔΔCt) method51. Primers for the nusA transcript (endogenous control) and target transcripts were designed with the aid of the Primer Express software v2.0 and experimentally validated for suitability for the 2−ΔΔCt method. All primers used are listed in Table S2. In the case of EP systems consisting of more than one protein (AcrAB, EmrAB, EmrKY, MacAB, MdtJI) encoded by genes clustered in a single operon, we monitored the transcription of the promoter-proximal gene.
Measurement of cytosolic pH
U937 cells were seeded in 96-well tissue culture plates at a density of 5 × 104 cells/well, differentiated and infected as described above. At 3 hours p.i. mock and S. flexneri M90T infected cells were loaded with pHrodo® Green AM intracellular pH indicator (ThermoFisher Scientific) according to the manufacturer’s instructions. After washing, fluorescence intensity was monitored by using the Victor multilabel counter (PerkinElmer). Afterwards, standard curves were created by incubating each sample with four different calibrated pH solutions (Intracellular pH Calibration buffer kit, ThermoFisher Scientific) and used to pinpoint the intracellular pH associated with the experimental conditions. Samples and calibrations were run in triplicate.
Competition assay
The relative survival of S. flexneri M90T and M90T ΔemrKY was tested by co-infecting U937 cells with both strains. Bacteria were grown separately at 37 °C in LB to OD600 0.6, then the two cultures were mixed, centrifuged and resuspended in RPMI 1640 containing 2mM L-glutamine. Macrophage infection was carried out as described above. Bacterial survival was monitored at time zero, and at 1, 2 and 3 hours p.i. At each time point, host cells were lysed with 1% Triton X-100, intracellular bacteria were collected, washed and resuspended in PBS. Serial dilutions of the bacterial suspension were plated on LB agar and, one day late, replicated on LB agar plates with or without 30 ug/ml kanamycin. To calculate the Competitive index (C.I.), the ratio of strains M90T ΔemrKY/M90T recovered at each time point from the infected cultures was determined and then normalized by dividing by the corresponding ratio in the initial inoculum.
Statistical analyses
The statistical differences between treatments were determined using Microsoft Excel by calculating the p values derived from a one-tailed ANOVA or, when indicated, a two-tailed t-test.
Supplementary information
Acknowledgements
This research was supported by grants from Sapienza Università di Roma and from Institut Pasteur (Paris, France) (PTR-24-16). F.B has been supported by Visiting Professor grant (VP-2015) from Sapienza University of Rome. The funders have no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author Contributions
Conceived and designed the experiments: M.P., M.G., G.P. and L.A. Performed the experiments: M.P., S.S., M.G. and L.A. Analyzed the data: M.P., M.G., G.P., F.B. and B.C. Contributed reagents/materials/analysis tools: B.C., F.B. and G.P. Wrote the paper: M.P., M.G., F.B., B.C. and G.P. All the authors review the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
5/22/2019
A correction to this article has been published and is linked from the HTML and PDF versions of this paper. The error has not been fixed in the paper.
Supplementary information
Supplementary information accompanies this paper at 10.1038/s41598-019-39749-3.
References
- 1.Piddock LJ. Clinically relevant chromosomally encoded multidrug resistance efflux pumps in bacteria. Clin. Microbiol. Rev. 2006;19:382–402. doi: 10.1128/CMR.19.2.382-402.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Li XZ, Plésiat P, Nikaido H. The challenge of efflux-mediated antibiotic resistance in Gram-negative bacteria. Clin. Microbiol. Rev. 2015;28:337–418. doi: 10.1128/CMR.00117-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Piddock LJ. Multidrug - resistance efflux pumps - not just for resistance. Nat. Rev. Microbiol. 2006;4:629–636. doi: 10.1038/nrmicro1464. [DOI] [PubMed] [Google Scholar]
- 4.Martinez JL, et al. Functional role of bacterial multidrug efflux pumps in microbial natural ecosystems. FEMS Microbiol. Rev. 2009;33:430–449. doi: 10.1111/j.1574-6976.2008.00157.x. [DOI] [PubMed] [Google Scholar]
- 5.Alcalde-Rico M, Hernando-Amado S, Blanco P, Martínez JL. Multidrug efflux pumps at the crossroad between antibiotic resistance and bacterial virulence. Front. Microbiol. 2016;7:1–14. doi: 10.3389/fmicb.2016.01483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bina XR, Provenzano D, Nguyen N, Bina JE. Vibrio cholerae RND family efflux systems are required for antimicrobial resistance, optimal virulence factor production, and colonization of the infant mouse small intestine. Infect. Immun. 2008;76:3595–3605. doi: 10.1128/IAI.01620-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Quillin SJ, Schwartz KT, Leber JH. The novel Listeria monocytogenes bile sensor BrtA controls expression of the cholic acid efflux pump MdrT. Mol. Microbiol. 2011;81:129–142. doi: 10.1111/j.1365-2958.2011.07683.x. [DOI] [PubMed] [Google Scholar]
- 8.Urdaneta V, Casadesús J. Adaptation of Salmonella enterica to bile: essential role of AcrAB-mediated efflux. Environ. Microbiol. 2018;20:1405–1418. doi: 10.1111/1462-2920.14047. [DOI] [PubMed] [Google Scholar]
- 9.Buckley AM, et al. The AcrAB-TolC efflux system of Salmonella enterica serovar Typhimurium plays a role in pathogenesis. Cell. Microbiol. 2006;8:847–856. doi: 10.1111/j.1462-5822.2005.00671.x. [DOI] [PubMed] [Google Scholar]
- 10.Bogomolnaya LM, et al. The ABC-type efflux pump MacAB protects Salmonella enterica serovar typhimurium from oxidative stress. MBio. 2013;4:e00630–13. doi: 10.1128/mBio.00630-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang-Kan X, et al. Lack of AcrB efflux function confers loss of virulence on Salmonella enterica serovar Typhimurium. MBio. 2017;8:e00968–17. doi: 10.1128/mBio.00968-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Taylor DL, Bina XR, Bina JE. Vibrio cholerae vexH encodes a multiple drug efflux pump that contributes to the production of cholera toxin and the toxin co-regulated pilus. PLoS One. 2012;7:e38208. doi: 10.1371/journal.pone.0038208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hirakata Y, et al. Multidrug efflux systems play an important role in the invasiveness of Pseudomonas aeruginosa. J. Exp. Med. 2002;196:109–118. doi: 10.1084/jem.20020005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lin YT, Huang YW, Chen SJ, Chang CW, Yang TC. The SmeYZ efflux pump of Stenotrophomonas maltophilia contributes to drug resistance, virulence-related characteristics, and virulence in mice. Antimicrob. Agents Chemother. 2015;59:4067–4073. doi: 10.1128/AAC.00372-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sakhtah H, et al. The Pseudomonas aeruginosa efflux pump MexGHI-OpmD transports a natural phenazine that controls gene expression and biofilm development. Proc. Natl. Acad. Sci. USA. 2016;113:E3538–E3547. doi: 10.1073/pnas.1600424113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lin J, Sahin O, Michel LO, Zhang Q. Critical role of multidrug efflux pump CmeABC in bile resistance and in vivo colonization of Campylobacter jejuni. Infect. Immun. 2003;71:4250–4259. doi: 10.1128/IAI.71.8.4250-4259.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jerse AE, et al. Estradiol-treated female mice as surrogate hosts for Neisseria gonorrhoeae genital tract infections. Front. Microbiol. 2011;2:107. doi: 10.3389/fmicb.2011.00107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.The HC, Thanh DP, Holt KE, Thomson NR, Baker S. The genomic signatures of Shigella evolution, adaptation and geographical spread. Nat. Rev. Microbiol. 2016;14:235–250. doi: 10.1038/nrmicro.2016.10. [DOI] [PubMed] [Google Scholar]
- 19.Schroeder GN, Hilbi H. Molecular pathogenesis of Shigella spp.: controlling host cell signaling, invasion, and death by type III secretion. Clin. Microbiol. Rev. 2008;21:134–156. doi: 10.1128/CMR.00032-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pupo GM, Lan R, Reeves PR. Multiple independent origins of Shigella clones of Escherichia coli and convergent evolution of many of their characteristics. Proc. Natl. Acad. Sci. USA. 2000;97:10567–10572. doi: 10.1073/pnas.180094797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rasko DA, et al. The pangenome structure of Escherichia coli: comparative genomic analysis of E. coli commensal and pathogenic isolates. J. Bacteriol. 2008;190:6881–6893. doi: 10.1128/JB.00619-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pasqua M, et al. The intriguing evolutionary journey of enteroinvasive E. coli (EIEC) toward pathogenicity. Front. Microbiol. 2017;8:2390. doi: 10.3389/fmicb.2017.02390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Balbi KJ, Rocha EPC, Feil EJ. The temporal dynamics of slightly deleterious mutations in Escherichia coli and Shigella spp. Mol. Biol. Evol. 2009;26:345–355. doi: 10.1093/molbev/msn252. [DOI] [PubMed] [Google Scholar]
- 24.Feng Y, Chen Z, Liu SL. Gene decay in Shigella as an incipient stage of host-adaptation. PLoS One. 2011;6:e27754. doi: 10.1371/journal.pone.0027754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Prosseda G, et al. Shedding of genes that interfere with the pathogenic lifestyle: the Shigella model. Res. Microbiol. 2012;163:399–406. doi: 10.1016/j.resmic.2012.07.004. [DOI] [PubMed] [Google Scholar]
- 26.Leuzzi A, et al. Role of the SRRz/Rz1lambdoid lysis cassette in the pathoadaptive evolution of Shigella. Int. J. Med. Microbiol. 2017;307:268–275. doi: 10.1016/j.ijmm.2017.03.002. [DOI] [PubMed] [Google Scholar]
- 27.Leuzzi A, et al. Multifactor regulation of the MdtJI polyamine transporter in Shigella. PLoS One. 2015;10:e0136744. doi: 10.1371/journal.pone.0136744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nickerson KP, et al. Analysis of Shigella flexneri resistance, biofilm formation, and transcriptional profile in response to bile salts. Infect. Immun. 2017;85:e01067–16. doi: 10.1128/IAI.01067-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Di Martino ML, Romilly C, Wagner EG, Colonna B, Prosseda G. One gene and two proteins: a leaderless mRNA supports the translation of a shorter form of the Shigella VirF regulator. MBio. 2016;7:e01860–16. doi: 10.1128/mBio.01860-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Di Martino ML, et al. Polyamines: emerging players in bacteria-host interactions. Int. J. Med. Microbiol. 2013;303:484–491. doi: 10.1016/j.ijmm.2013.06.008. [DOI] [PubMed] [Google Scholar]
- 31.Nishino K, Yamaguchi A. Analysis of a complete library of putative drug transporter genes in. Escherichia coli. J. Bacteriol. 2001;183:5803–5812. doi: 10.1128/JB.183.20.5803-5812.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kobayashi A, Hirakawa H, Hirata T, Nishino K, Yamaguchi A. Growth phase-dependent expression of drug exporters in Escherichia coli and its contribution to drug tolerance. J. Bacteriol. 2006;188:5693–5703. doi: 10.1128/JB.00217-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sansonetti PJ, Kopecko DJ. & Formal, S. B. Involvement of a plasmid in the invasive ability of Shigella flexneri. Infect. Immun. 1982;35:852–860. doi: 10.1128/iai.35.3.852-860.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ashida H, et al. Shigella deploy multiple countermeasures against host innate immune responses. Curr. Opin. Microbiol. 2011;14:16–23. doi: 10.1016/j.mib.2010.08.014. [DOI] [PubMed] [Google Scholar]
- 35.Eguchi Y, Utsumi R. Alkali metals in addition to acidic pH activate the EvgS histidine kinase sensor in. Escherichia coli. J. Bacteriol. 2014;196:3140–3149. doi: 10.1128/JB.01742-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Eaves DJ, Ricci V, Piddock LJ. Expression of acrB, acrF, acrD, marA, and soxS in Salmonella enterica serovar Typhimurium: role in multiple antibiotic resistance. Antimicrob. Agent Chemother. 2004;48:1145–1150. doi: 10.1128/AAC.48.4.1145-1150.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Blair JM, et al. Expression of homologous RND efflux pump genes is dependent upon AcrB expression: implications for efflux and virulence inhibitor design. J. Antimicrob. Chemother. 2015;70:424–431. doi: 10.1093/jac/dku380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kato A, et al. Transcription of emrKY is regulated by the EvgA-EvgS two-component system in Escherichia coli K-12. Biosci. Biotechnol. Biochem. 2000;64:1203–1209. doi: 10.1271/bbb.64.1203. [DOI] [PubMed] [Google Scholar]
- 39.Eguchi Y, et al. Transcriptional regulation of drug efflux genes by EvgAS, two-component system in Escherichia coli. Microbiology. 2003;149:2819–2828. doi: 10.1099/mic.0.26460-0. [DOI] [PubMed] [Google Scholar]
- 40.Roggiani M, Yadavalli SS, Goulian M. Natural variation of a sensor kinase controlling a conserved stress response pathway in Escherichia coli. PLoS Genet. 2017;13:e1007101. doi: 10.1371/journal.pgen.1007101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tanabe H, et al. Growth phase-dependent transcription of emrKY, a homolog of multidrug efflux emrAB genes of Escherichia coli, is induced by tetracycline. J Gen Appl Microbiol. 1997;43:257–263. doi: 10.2323/jgam.43.257. [DOI] [PubMed] [Google Scholar]
- 42.Hinchliffe P, et al. Structure of the periplasmic adaptor protein from a major facilitator superfamily (MFS) multidrug efflux pump. FEBS Lett. 2014;588:3147–3153. doi: 10.1016/j.febslet.2014.06.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Matsumura K, Furukawa S, Ogihara H, Morinaga Y. Roles of multidrug efflux pumps on the biofilm formation of Escherichia coli K-12. Biocontrol Sci. 2011;16:69–72. doi: 10.4265/bio.16.69. [DOI] [PubMed] [Google Scholar]
- 44.Han X, et al. Escherichia coli genes that reduce the lethal effects of stress. BMC Microbiol. 2010;10:35. doi: 10.1186/1471-2180-10-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Offner FF. Ion flow through membranes and the resting potential of cells. J Membr Biol. 1991;123:171–82. doi: 10.1007/BF01998087. [DOI] [PubMed] [Google Scholar]
- 46.Lucchini S, Liu H, Jin Q, Hinton JC, Yu J. Transcriptional Adaptation of Shigella flexneri during infection of macrophages and epithelial cells: insights into the strategies of a cytosolic bacterial pathogen. Infect Immun. 2005;73:88–102. doi: 10.1128/IAI.73.1.88-102.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Durfee T, et al. The complete genome sequence of Escherichia coli DH10B: insights into the biology of a laboratory workhorse. J Bacteriol. 2008;190:2597–2606. doi: 10.1128/JB.01695-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Datsenko KA, Wanner BL. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA. 2000;97:6640–5. doi: 10.1073/pnas.120163297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hautefort I, Proença MJ, Hinton JC. Single-copy green fluorescent protein gene fusions allow accurate measurement of Salmonella gene expression in vitro and during infection of mammalian cells. Appl. Environ. Microbiol. 2003;69:7480–7491. doi: 10.1128/AEM.69.12.7480-7491.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Barbagallo M, et al. A new piece of the Shigella pathogenicity puzzle: spermidine accumulation by silencing of the speG gene. PLoS One. 2011;6:e27226. doi: 10.1371/journal.pone.0027226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.