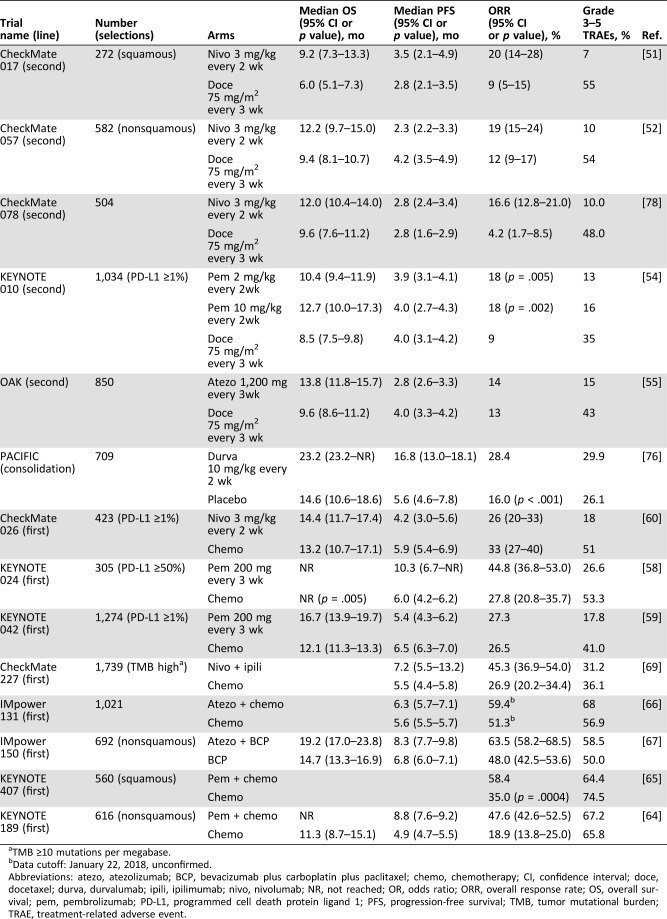
Table 2. Overview of phase III trials of PD‐1/PD‐L1 blockade therapy in advanced non‐small‐cell lung cancer as of August 2018.
TMB ≥10 mutations per megabase.
Data cutoff: January 22, 2018, unconfirmed.
Abbreviations: atezo, atezolizumab; BCP, bevacizumab plus carboplatin plus paclitaxel; chemo, chemotherapy; CI, confidence interval; doce, docetaxel; durva, durvalumab; ipili, ipilimumab; nivo, nivolumab; NR, not reached; OR, odds ratio; ORR, overall response rate; OS, overall survival; pem, pembrolizumab; PD‐L1, programmed cell death protein ligand 1; PFS, progression‐free survival; TMB, tumor mutational burden; TRAE, treatment‐related adverse event.