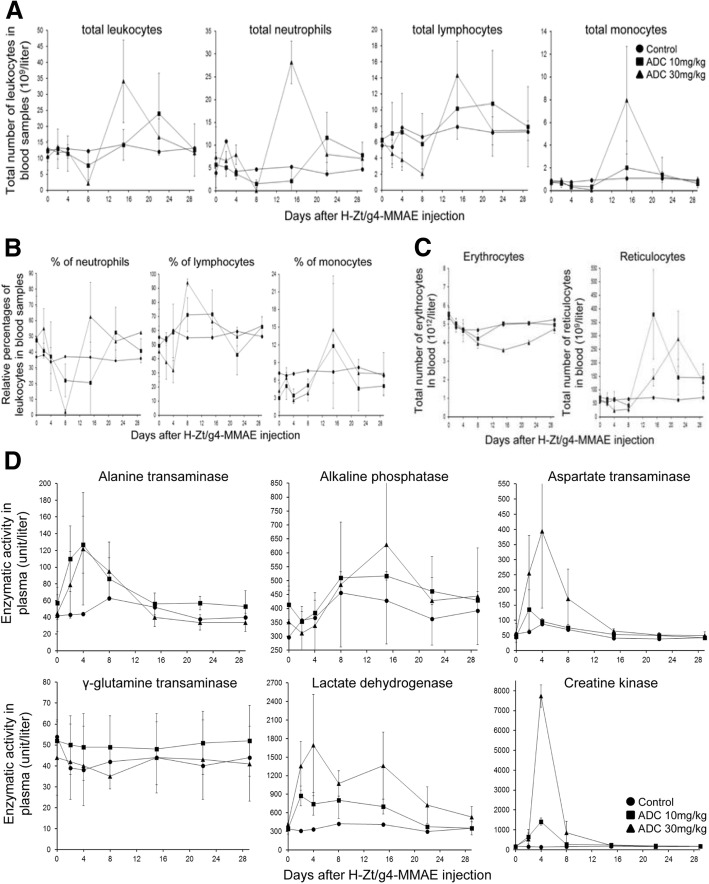
Fig. 6.
Toxicological activities of H-Zt/g4-MMAE in mouse and cynomolgus monkey. (a) and (b) Adverse activities of H-Zt/g4-MMAE in blood leukocytes in cynomolgus monkey. H-Zt/g4-MMAE at 10 or 30 mg/kg in a single dose was injected once into cynomolgus monkey. Monkeys without ADC injection served as the control. Peripheral blood samples were collected at different time intervals. Total numbers of leukocytes (a) including neutrophil, lymphocytes, and monocytes from each group were countered accordingly. The percentages of blood leukocytes (b) were also determined. (c) Adverse effect of H-Zt/g4-MMAE on blood erythrocytes and reticulocytes in cynomolgus monkey. Total numbers of erythrocytes and reticulocytes from blood samples collected from each group as described in (b) were counted accordingly. (d) Adverse effects of H-Zt/g4-MMAE on various enzymes in plasma of cynomolgus monkey. Six enzymatic activities from each group were quantitatively measured using the blood samples collected from individual monkeys as described in (b)