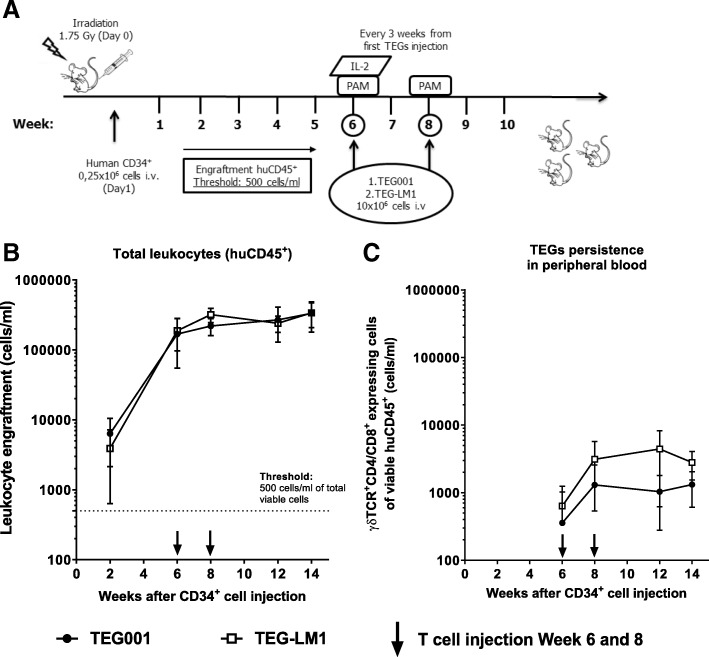
Fig. 3.
In vivo safety profile of TEG001. (a) Schematic overview of the safety experiment in healthy donor-derived xenograft (H-DX) model. NSG mice were irradiated at day 0 and engrafted with healthy cord blood-derived CD34+ progenitor cells on day 1. Engraftment was followed-up in peripheral blood by flow cytometry and when > 500 huCD45+ cells/ml were present, mice received 2 injections of therapeutic TEG001 or TEG-LM1 mock in the presence of PAM (at week 6 and 8) and IL-2 (at week 6); (b-c) In vivo safety profile of TEG001 towards healthy human hematopoietic cells. Healthy human cells engrafted in NSG mice (b) with long-term persistence of TEGs in peripheral blood (c). Data represent mean ± SD of all mice per group (n = 10)