Abstract
Surgical site infections (SSI) are devastating complications after surgery for femoral neck fractures. There are a lot of literature have shown a strong association between diabetic patients and SSI. This study aimed to identify diabetes as an independent risk factor of SSI, focusing on femoral neck fractures, and to investigate the other potential risk factors for SSI.
We retrospectively collected data from patients who underwent surgery for femoral neck fractures through the medical record management system at a single level 1 hospital between January 2015 and June 2016. Demographic and clinical patient factors and characteristics of SSI were recorded. The case group was defined as patients with SSI and the control group was defined as patients without SSI. Univariate and multivariate analyses were performed to determine the risk factors for SSI.
Data were provided for 692 patients, among whom 26 had SSI, representing an incidence rate of 3.67%. In the SSI group, 24 (3.47%) patients had superficial infection and 2 (0.29%) had deep infection. On multivariate analysis, diabetes (P < .001) was determined an independent risk factor of SSI, so were surgery performed between May and September (P = .04), body mass index (P = .031), corticosteroid therapy (P = .003), anemia (P = .041), and low preoperative hemoglobin levels.
Our results suggest that clinicians should recognize patients with these factors, particularly diabetes. And taking management optimally in the preoperative period will prevent the SSI after femoral neck fracture.
Keywords: femoral neck fractures, incidence, risk factors, surgical site infection
1. Introduction
Femoral neck fracture is one of the most common reasons for admission of elderly patients to the acute orthopedic ward, accounting for 48.22% of hip fractures and 3.13% of all fractures in adults.[1] These injuries generally require surgical treatment to restore the normal range of motion in order to perform activities of daily living. The mortality rate associated with a femoral neck fracture is approximately 30% during the first year after surgical repair[2]; most deaths are because of comorbidities and postoperative complications,[3] among which surgical site infections (SSI) plays an important role.[4–7] When SSI occurs, patients may require readmission and reoperation to exchange or remove the surgical hardware.[7–14] From an economic perspective, SSI can cause prosthetic loosening and the need for revision surgery, increased antibiotic use, and prolonged hospital stay, and it represents an increasing financial burden within the total healthcare expenditure.[15,16] As reported by Edwards et al, when compared with uninfected hip fractures, the cost of treating a patient with a deep wound infection represents approximately a 3 fold increase.[7]
Most previous studies have focused on specific pathomechanisms or individual clinical populations, such as microbiologic features,[17] patients with diabetes mellitus (DM),[18] and deep wound infection,[19] or the cost-effectiveness of prophylactic antibiotic use.[7] Additionally, because of the use of univariate analyses rather than multivariate analyses, the correlations between SSI and certain risk factors and comorbidities have been reported as inconclusive and their independent effects have not been confirmed.[17]
On the basis of this, the objectives of this study were to assess the incidence and risk factors including diabetes and other potential comorbidities for the development of SSI after surgical repair of femoral neck fracture by evaluating all suspected risk factors from collected clinical data. Recognition of risk factors is valuable for the accurate estimation of individual patient risk associated with femoral neck procedures, and may serve as the basis for further clinical studies evaluating prophylactic therapy.
2. Methods
2.2. Ethics statement
The study design was reviewed and approved by The Third Hospital of Hebei Medical University (THHMU) Human Research Ethics Committee (IRB number: no. KE2014-015-1).
2.3. Inclusion and exclusion criteria
This is a single center, retrospective, case–control study performed in the Department of Orthopedic Surgery at THHMU. We retrospectively evaluated patients who were admitted over a period of 18 months (January 2015–June 2016). Data on all patients aged 18 years or older with acute closed femoral neck fractures treated surgically were collected from the medical records. Old fractures (>21 days from initial injury) and pathological fractures were excluded. Patients who were admitted only for SSI treatment but without initial surgery at our hospital were not included in this analysis. Patients whose information was unavailable or incomplete were excluded. Finally, patients were divided into 2 groups according to the occurrence of SSI. The case group was defined as patients with SSI and the control group was defined as patients without SSI. The flow chart representing the selection of study participants is shown in Figure 1.
Figure 1.
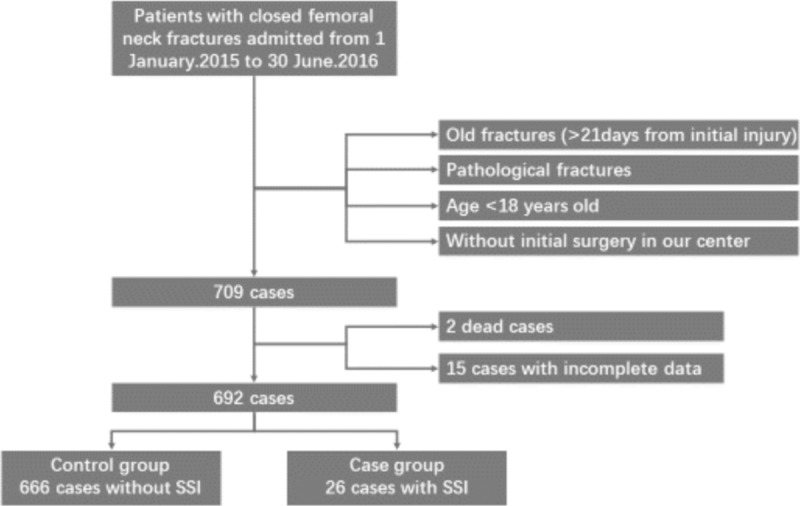
Flowchart for the selection of study participants.
To analyze risk factors, data concerning patient factors, inciting accident, fracture factors, surgical procedures, and laboratory analyses were collected from the medical record management system (MRMS) of our hospital by 2 well-trained members of the study team.
2.4. Definition of SSI and related variables
According to the Center for Disease Control standard for SSI, we defined SSI as superficial and deep. Deep infections must meet at least 1 of the following criteria: infection surpassing the deep fascia; persistent wound discharge or dehiscence; visible abscess or gangrene requiring surgical debridement; and implant removal or exchange. A superficial SSI was diagnosed if the patient received antibiotic treatment for wound signs and symptoms (redness, swelling, warmth, and pain) but did not meet the criteria for deep infection, irrespective of the microbiology result.
The following data were collected from the medical records of each patient and recorded in a standardized form. First, demographic characteristics, including age, sex, height, weight, lifestyle factors, underlying medical diagnoses, living environment (rural or urban), injury mechanism (low or high energy), injury type (closed or open), and side of injury, were collected from the admission records. Second, surgery characteristics, including data on whether operation time was between May and September, time from admission to initiation of surgery, duration of operation, fixation type, anesthesia methods (local, general, or combination), and intraoperative blood loss were collected from the surgery and anesthesia notes. Third, data on antibiotic use (preoperative, intraoperative, and/or postoperative) and application of a drainage tube postoperatively were collected from physician and nursing records. Fourth, patients’ lifestyles, underlying medical diagnoses, and long-term medications were obtained directly from past medical history in the MRMS and were expressed as “present” or “not.” These factors included smoking statusDM, hypertension, coronary heart disease, anemia, rheumatism, nephropathy, chemoradiotherapy, asthma, history of allergic, and operation in any site. Medications including immunosuppressant, corticosteroids, and others were administered. Finally, we documented laboratory values and divided them into normal range, above normal range, and below normal range. These variables included preoperative white blood cell, neutrophil, lymphocyte, monocyte, eosinophil, basophil, and red blood cell (RBC) counts; hemoglobin (Hb), platelet, serum total protein, albumin, and globulin levels; and the albumin/globulin ratio.
Body mass index (BMI) was calculated as the weight divided by the square of the height, patients were grouped according to the Chinese reference criteria: underweight, <18.5; normal, 18.5–23.9; overweight, 24–27.9; obesity, 28–31.9; morbid obesity, 32 and above. The patients were divided into 2 groups according to the volume of intraoperative blood loss: less than 400 ml and more than 400 ml. Injury mechanism was classified as low energy if fractures were caused by falls from standing height and as high energy if they were caused by falls from a greater height, traffic accidents, and other causes. Preoperative stay was defined as the interval between injury occurrence and surgery and was divided into 2 groups: <7 days and ≥7 days. Fixation types were divided into 2 groups: osteosynthesis or artificial hip joint prosthesis. Preoperative use of antibiotic was defined as antibiotic use up to 1 hour before the first scalpel cut. Postoperative use of antibiotic was defined as antibiotic use beginning the first day after the operation. We used digits to quantify patients’ preoperative coexisting injuries, with larger numbers representing worse bodily function status. We added “1” if patients sustained trauma to 1 of the following systems: musculoskeletal, abdominal, cardiothoracic, urogenital, vascular, and central nervous systems. For multiple injuries of the same system, we added only “1” to the total digits. For example, for a patient with concurrent pulmonary contusion and traumatic pleural effusion or a patient with concurrent greater Garden III fractures, we added only “1” in the tables.
2.5. Statistical analysis
Data were analyzed using SPSS statistical software, version 19.0 (IBM, Armonk, NY). Categorical variables are expressed as numeric values (percentages) and compared using the χ2 test or Fisher exact test, as appropriate. Continuous variables are expressed as median values (interquartile ranges [IQRs]) and were compared using the T test or Mann–Whitney U-test depending on the data distribution (equal variance and normality or not).
Univariate logistic analysis was performed to evaluate the association between each categorical variable and SSI. The significance threshold was set at P < .05. Finally, all variables were entered into a multivariable logistic regression model to determine their independent effects on SSI occurrence. The Hosmer–Lemeshow test was used to examine the goodness-of-fit of this model, and a P value > .05 indicated acceptable fitness.
3. Results
3.2. Overall fracture characteristics
During the study period, a total of 709 patients meeting the inclusion and exclusion criteria were initially screened and 692 (692 fractures) were ultimately analyzed, after exclusion of 2 cases of patient death and 15 cases with incomplete data. All were closed fractures, including 263 males and 429 females. The mean age was 69 years (IQR, 59–78; range, 18–95) among patients without SSI and 69.5 years (IQR, 58.5–75; range, 44–88) among patients with SSI (P = .649). Over 70% of patients were aged 60 years and older, including 490 (73.6%) patients in the non-SSI group and 19 (73.1%) patients in the SSI group.
3.3. Characteristics of SSI
Twenty-six of the 692 patients developed SSI; 24 (3.47% [95% confidence interval (CI) 2.10–4.83%]) were superficial and 2 (0.29% [95% CI 0.11–0.69%]) were deep, representing an incidence rate of 3.76% (95% CI 2.34–5.17%). The earliest diagnosis of SSI occurred at the 4th postoperative day, whereas the latest presentation was at the 15th postoperative day; the median time from surgery to diagnosis of SSI was 7 days. The timing of SSI is reported in Figure 2. Demographically, there were no statistically significant differences between the SSI and non-SSI groups, although the mean age was somewhat older in the SSI groups (69.5 vs 69 years) (Table 1). Regarding preoperative variables, the SSI group did not differ from the non-SSI group in terms of preoperative stay (5 vs 4 days, P = .129), intraoperative blood loss (296 vs 279 ml, P = .380), or surgery duration (108 vs 110 minutes, P = .634). Surgery was performed a median of 4.6 days after the injury. A total of 238 patients were operated upon within 2 days; among these, 13 developed SSI. A total of 347 patients were operated upon from the 3rd to 7th day after injury, among whom 10 developed SSI. The remaining 107 patients were operated upon at least 7 days after hospital admission; among them, 3 developed SSI, including 2 cases of deep SSI (χ2 = 2.918, P = .233). The mean total duration of hospital stay was 15.0 days overall, 19.0 days in the SSI group (1 patient with deep SSI with a stay of 26 days, the remaining patients with stays of 14 days) and 14.8 days in the non-SSI group; this difference was statistically significant (P < .001).
Figure 2.
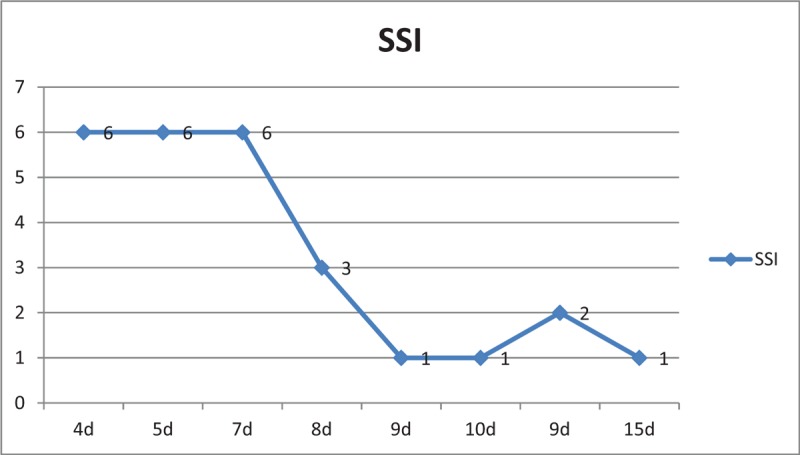
Distribution of surgical site infections over time.
Table 1.
Comparison of continuous variables in patients with and without SSI.

3.4. Univariate and multivariate analyses
In the univariate analysis, surgery between May and September (P = .003), concurrent injuries (P = .016), BMI = >28.0 (P < .001), DM (P < .001), rheumatism (P = .013), corticosteroid therapy (P < .001), asthma (P < .001), history of anemia (P = .042), intraoperative use of antibiotics (P = .037), preoperative RBC count (P = .019), and preoperative Hb (P = .017) were identified as significant risk factors for SSI. Other factors, including demographic information, other underlying diseases, preoperative factors, and other laboratory variables were not associated with SSI occurrence. Detailed information is presented in Table 2 .
Table 2.
Univariate analysis of factors associated with SSI of femoral neck fractures after operative.

In the multivariate model, surgery between May and September, abnormal BMI, corticosteroid therapy, underlying disease DM, anemia, and lower preoperative Hb were independent risk factors associated with SSI, with adjusted odds ratios (ORs) of 4.255 (1.6–11.346), 1.624 (1.046–2.523), 27.966 (3.107–251.727), 5.881 (2.232–15.500), 5.087 (1.066–24.272), and 3.446 (1.438–8.261), respectively, after eliminating the effects of confounding factors. The results of the Hosmer–Lemeshow test demonstrated adequate fitness (χ2 = 10.645, P = .223). Detailed information is presented in Table 3. The power of the DM in logistic regression modal is approximate 95% using post hoc power analysis (alpha = 0.05, beta = 0.0548).
Table 2 (Continued).
Univariate analysis of factors associated with SSI of femoral neck fractures after operative.

Table 3.
Multivariate analysis of factors associated with SSI after instrumented femoral neck surgery.

4. Discussion
The SSI rate observed in this study (3.76%) was within the range of previously reported rates after surgery for femoral neck fracture (2–7%).[10,20–25] Studies published in 2000 indicated that deep SSI associated with hip fractures occurred at a rate of 1.3 to 3.6%[26] and nearly 30% of them occurred after discharge.[23] Because of a lack of follow up after discharge, some delayed and late infections may not be captured in the present study, which could partly explain why only 2 cases of deep SSI (0.29%) were identified. Therefore, the overall SSI rate calculated in our study is lower than most previous reports, especially for deep SSI.
DM has been confirmed to have a significant association with deep infection and an increased risk of mortality associated with hip fractures.[18,5,27] The present study ascertained DM was an increased risk of SSI after femoral neck fracture. There are several path mechanisms underlying SSI in patients with DM that could explain this phenomenon. When microangiopathy is present, impaired nutrition and oxygen delivery to the peripheral tissues could reduce the systemic ability to resist infection. Poor blood glucose control results in impaired leukocyte functions such as adherence,[28] chemotaxis,[29,30] phagocytosis, and intracellular elimination of pathogens.[31] Defective fibroblast proliferation and impaired red collagen synthesis could delay wound healing.[32] Neuropathy with autonomic system damage causes the skin to dry and crack, destroying its integrity and its ability to resist infection.[33] Several previous studies have found obesity to increase the odds of SSI from 2.2 to 7.1 fold, with statistical significance.[12,34–36] The algorithm used in our study divided BMI into 5 levels (normal: 18.5–23.9; underweight: <18.5; overweight: 24–27.9; obesity: 28–31.9; morbid obesity: >32) and the adjusted OR was 1.624. This indicates that BMI values both above or below the normal range are risk factors for SSI in femoral neck fractures. A survey of 30,000 wounds published by Cruse and Foord found that adiposity and malnutrition were both predisposing factors for infection.[37,38] On the basis of these results, the current authors hypothesized that the reason why abnormal BMI had a negative influence on SSI following femoral neck fracture is that 73.6% of patients in this study were aged 60 years and older and thus would have an overall poor health condition. These assumptions remain to be confirmed. Corticosteroid therapy was identified as a factor associated with SSI in the survey by Cruse and Foord because of its immunosuppressive effect.[37,38] The roles of DM, abnormal BMI, and corticosteroid therapy as independent risk factors for SSI have been discussed repeatedly, and these diagnoses represent established significant risk factors for SSI after surgical repair of femoral neck fractures.[18,5,27,37,38] The mechanisms of these associations are well-established, including vasculopathy, neuropathy, and vasospasm resulting in inhibition of wound healing and impaired immune function, thereby predisposing patients to infectious complications.[39–41] How to more effectively manage patients with these metabolic disorders and the side effects of corticosteroid therapy, in particular, the optimal timing of surgery and the most appropriate operative approach, remains a concern. It is therefore advised that DM should be well controlled before elective surgery and patients should be encouraged to lose weight, if possible, before femoral neck surgeries. If patients are unable to avoid corticosteroid therapy, more aggressive measures should be taken to reduce the risk of SSI.
A history of anemia and lower preoperative Hb level (<120 g/L in males or <110 g/L in females) were first reported as risk factors for SSI following surgical repair of femoral neck fractures, although they were later reported to also be risk factors for SSI after spine surgery and colorectal surgery.[42–46] A low preoperative Hb level was associated with a higher risk of serious morbidity or death in patients with cardiovascular disease than in those without cardiovascular disease, and has also been reported as a significant independent predictor of mortality, length of hospital stay, and postoperative pneumonia in noncardiac surgical patients.[47] We predicted that the same adverse consequences would exist following surgery for femoral neck fractures. In this study, a history of anemia, which was present in 15.4% of the SSI group and 5.4% of the non-SSI group, and lower preoperative Hb, present in 38.5% of the SSI group and 18.8% of the non-SSI group, were significant risk factors in the final multivariate analysis (P = .041, P = .006). When comparing studies, however, differences in the definition and severity of anemia must be recognized. Preoperative anemia and a history of anemia were found to be risk factors associated with SSI in the current analysis. However, a systematic review and meta-analysis found that allogeneic blood transfusion is a risk factor for postoperative bacterial infection.[48] Therefore, how to manage and correct preoperative anemia remains a challenge for surgeons and researchers.
One potential factor contributing to an increased external risk of SSI is microbial contamination in the operating room.[49,50] Levels of CFU/m3 measured in the operating room were found to have a strong correlation with staff behavior and traffic, with increasing CFU levels for each door-opening and person present and with an increase in the duration of surgery.[50,51] Andersson et al[51] reported that traffic flow, number of persons present, and the duration of surgery explained 68% of the variance in total CFU/m3. Humans shed large amounts of particles and skin fragments and constitute the main reservoir of air contaminants in the OR.[52] Staff traffic may transfer bacteria and fungi within the operating room.[53] The Healthcare Infection Society recommended an upper limit of contamination during surgery of <180 CFU/m3.[54] Lynch et al[55] reported a mean rate of 40 door openings per hour for orthopedic total joint replacement, which must be considered in light of the strong correlation between the door opening rate and elevated CFU levels. It has been suggested that the levels be maintained at <10 CFU/m3 during implant surgery and that clinical benefits can be expected by reducing the levels to 1 CFU/m3.[56] The average temperature between May and September is above 20°C (68°) at the study location. To our knowledge, traumatic injuries occur most commonly in industrial and traffic accidents, and are significantly increased in these months because of an increased rate of dangerous daytime activities in a warm climate. Meanwhile, a large proportion of the individuals present during surgery were students or new recruits who observed surgery or were undergoing prejob training as part of their education from May to September. These 2 aspects together contributed to substantial microbial air contamination in the operating room during surgery, which correlates strongly with increased traffic flow. As a result, in the present study, surgery performed in these months represented an independent risk factor for SSI after femoral neck fracture surgery.
The strengths of this study include the analysis of consecutively treated patients over an 18-month period and the relatively large sample size. In addition, hospital care is standardized for all patients with femoral neck fracture at THHMU through the protocol-driven clinical care pathway. Limitations of the study include the retrospective design and the possible inaccuracy or misinterpretation of information abstracted from the medical records. Second, the condition of patients treated at a level 1 trauma center is more severe, and these patients do not represent the overall trend of SSI development. Finally, the lack of follow up after discharge may lead to missed cases of SSI.
In summary, we estimated an SSI incidence of 3.76% after femoral neck fracture surgery, with deep SSI representing 0.29% and superficial SSI representing 3.47% of cases. Our single-center study found that DM was an undisputed increase in the risk of SSI. And multiple and diverse factors were associated with an increased risk of SSI following surgery for femoral neck fractures, including abnormal BMI (<18.5 and >23.9), corticosteroid therapy, anemia, low preoperative Hb levels, and the performance of surgery between May and September. Preoperatively, patient treatment strategies should emphasize upon the management of patients’ underlying medical condition, controlling body weight and blood glucose levels, correcting anemia, and cessation of corticosteroid therapy. In addition, surgeons should pay attention to avoid performing surgery between May and September.
Acknowledgments
We are grateful to Y Li and L Fu of the Department of Orthopedics, and to Y Lu of the Department of statistics and applications for their kind assistance.
Author contributions
Chenni Ji and Yanbin Zhu designed the study and searched relevant studies; Fei Zhang, Song Liu, Jia Li, and Wei Chen analyzed and interpreted the data; Chenni Ji wrote the manuscript and Yingze Zhang approved the final version of the manuscript.
Conceptualization: Jia Li, Yingze Zhang.
Data curation: Chenni Ji, Yanbin Zhu, Jia Li, Fei Zhang, Wei Chen.
Formal analysis: Wei Chen.
Investigation: Fei Zhang.
Methodology: Yanbin Zhu, Song Liu, Wei Chen.
Resources: Song Liu, Jia Li, Yingze Zhang.
Software: Song Liu, Jia Li, Wei Chen.
Supervision: Yingze Zhang.
Validation: Yingze Zhang.
Writing – review and editing: Chenni Ji, Yanbin Zhu.
Footnotes
Abbreviations: BMI = body mass index, DM = diabetes mellitus, Hb = hemoglobin, IQR = interquartile range, MRMS = medical record management system, OR = odds ratio, RBC = red blood cell, SSI = surgical site infection, THHMU = The Third Hospital of Hebei Medical University.
CJ contributed to this manuscript.
Funding support was provided by the Support Program for the Top Young Talents for Hebei Province.
All the authors declare that they have no conflict of interest.
References
- [1].Thieme, Zhang Y, Su Y, Hao J, et al. Clinical Epidemiology of Orthopedic Trauma. 2012;177–193. [Google Scholar]
- [2].Parker M, Johansen A. Hip fracture. BMJ 2006;333:27–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Roche JJ, Wenn RT, Sahota O, et al. Effect of comorbidities and postoperative complications on mortality after hip fracture in elderly people: prospective observational cohort study. BMJ 2005;331:1374–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Pollard TC, Newman JE, Barlow NJ, et al. Deep wound infection after proximal femoral fracture: consequences and costs. J Hosp Infect 2006;63:133–9. [DOI] [PubMed] [Google Scholar]
- [5].Partanen J, Syrjala H, Vahanikkila H, et al. Impact of deep infection after hip fracture surgery on function and mortality. J Hosp Infect 2006;62:44–9. [DOI] [PubMed] [Google Scholar]
- [6].Fox HJ, Pooler J, Prothero D, et al. Factors affecting the outcome after proximal femoral fractures. Injury 1994;25:297–300. [DOI] [PubMed] [Google Scholar]
- [7].Edwards C, Counsell A, Boulton C, et al. Early infection after hip fracture surgery:riskfactors, costs and outcome. J Bone Joint Surg Br 2008;90:770–7. [DOI] [PubMed] [Google Scholar]
- [8].De Boer AS, Mintjes-de Groot AJ, Severijnen AJ, et al. Risk assessment for surgical-site infections in orthopedic patients. Infect Control Hosp Epidemiol 1999;20:402–7. [DOI] [PubMed] [Google Scholar]
- [9].Huotari K, Lyytikainen O. Impact of postdischarge surveillance on the rate of surgical site infection after orthopedic surgery. Infect Control Hosp Epidemiol 2006;27:1324–9. [DOI] [PubMed] [Google Scholar]
- [10].Kalmeijer MD, van Nieuwland-Bollen E, Bogaers-Hofman D, et al. Nasal carriage of Staphylococcus aureus is a major risk factor for surgical-site infections in orthopedic surgery. Infect Control Hosp Epidemiol 2000;21:319–23. [DOI] [PubMed] [Google Scholar]
- [11].Merrer J, Girou E, Lortat-Jacob A, et al. Surgical site infection after surgery to repair femoral neck fracture: a French multicenter retrospective study. Infect Control Hosp Epidemiol 2007;28:1169–74. [DOI] [PubMed] [Google Scholar]
- [12].Olsen MA, Nepple JJ, Riew KD, et al. Risk factors for surgical site infection following orthopaedic spinal operations. J Bone Joint Surg Am 2008;90:62–9. [DOI] [PubMed] [Google Scholar]
- [13].SooHoo NF, Krenek L, Eagan MJ, et al. Complication rates following open reduction and internal fixation of ankle fractures. J Bone Joint Surg Am 2009;91:1042–9. [DOI] [PubMed] [Google Scholar]
- [14].Wimmer C, Gluch H, Franzreb M, et al. Predisposing factors for infection in spine surgery: a survey of 850 spinal procedures. J Spinal Disord 1998;11:124–8. [PubMed] [Google Scholar]
- [15].Green JW, Wenzel RP. Postoperative wound infection: a controlled study of the increased duration of hospital stay and direct cost of hospitalization. Ann Surg 1977;185:264–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Metsemakers W, Smeets B, Nijs S, et al. Infection after fracture fixation of the tibia: analyse of healthcare utilisation and relater cosets. Bone Joint J 2016;98:78–178. [DOI] [PubMed] [Google Scholar]
- [17].Jacques M, Emmanuelle G, Alain LJ, et al. Surgical site infection after surgery to repair femoral neck fracture: a French multicenter retrospective study. Infect Control Hosp Epidemiol 2007;28:1169–74. [DOI] [PubMed] [Google Scholar]
- [18].Gulcelik NE, Bayraktar M, Caglar O, et al. Mortality after hip fracture in diabetic patients. Exp Clin Endocrinol DM 2011;119:414–8. [DOI] [PubMed] [Google Scholar]
- [19].Mackay DC, Harrison WJ, Bates JHJ, et al. Audit of deep wound infection following hip fracture surgery. J R Coll Surg Edinb 2000;45:56–9. [PubMed] [Google Scholar]
- [20].Khan OA, Weston VC, Scammell BE. Methicillin-resistant Staphylococcus aureus incidence and outcome in patients with neck of femur fractures. J Hosp Infect 2002;51:185–8. [DOI] [PubMed] [Google Scholar]
- [21].Ridgeway S, Wilson J, Charlet A, et al. Infection of the surgical site after arthroplasty of the hip. J Bone Joint Surg Br 2005;87:844–50. [DOI] [PubMed] [Google Scholar]
- [22].UK Health Protection Agency. Mandatory Surveillance of Surgical Site Infection in Orthopaedic Surgery. Report of Data Collected Between April 2004 and December 2005. Available at: http://www.hpa.org.uk/infections/topics_az/hai/SSI_mandatory_0405report.pdf [Accessed on January 7, 2007]. [Google Scholar]
- [23].Mannien J, Wille JC, Snoeren RL, et al. Impact of postdischarge surveillance on surgical site infection rates for several surgical procedures: results from the nosocomial surveillance network in The Netherlands. Infect Control Hosp Epidemiol 2006;27:809–16. [DOI] [PubMed] [Google Scholar]
- [24].Soriano A, Popescu D, Garcia S, et al. Usefulness of teicoplanin for preventing methicillin-resistant Staphylococcus aureus infections in orthopedic surgery. Eur J Clin Microbiol Infect Dis 2006;25:35–8. [DOI] [PubMed] [Google Scholar]
- [25].Merrer J, Desbouchages L, Serazin V, et al. Comparison of routine prophylaxis with vancomycin or cefazolin for femoral neck fracture surgery: microbiological and clinical outcomes. Infect Control Hosp Epidemiol 2006;27:1366–71. [DOI] [PubMed] [Google Scholar]
- [26].Mackay DC, Harrison WJ, Bates JH, et al. Audit of deep wound infection following hip fracture surgery. J R Coll Surg Edinb 2000;45:56–9. [PubMed] [Google Scholar]
- [27].Enstone JE, Humphreys H. Monitoring infective complications following hip fracture. J Hosp Infect 1998;38:1–9. [DOI] [PubMed] [Google Scholar]
- [28].Bagdade JD, Stewart M, Walters E. Impaired granulocyte adherence. A reversible defect in host defense in patients with poorly controlled DM. DM 1978;27:677–81. [DOI] [PubMed] [Google Scholar]
- [29].Delamaire M, Maugendre D, Moreno M, et al. Impaired leucocyte functions in diabetic patients. Diabet Med 1997;14:29–34. [DOI] [PubMed] [Google Scholar]
- [30].Mowat A, Baum J. Chemotaxis of polymorphonuclear leukocytes from patients with DM mellitus. N Engl J Med 1971;284:621–7. [DOI] [PubMed] [Google Scholar]
- [31].Sima AA, O’Neill SJ, Naimark D, et al. Bacterial phagocytosis and intracellular killing by alveolar macrophages in BB rats. DM 1988;37:544–9. [DOI] [PubMed] [Google Scholar]
- [32].Twigg SM, Chen MM, Joly AH, et al. Advanced glycosylation end products up-regulate connective tissue growth factor (insulin-like growth factor-binding protein-related protein 2) in human fibroblasts: a potential mechanism for expansion of extracellular matrix in DM mellitus. Endocrinology 2001;142:1760–9. [DOI] [PubMed] [Google Scholar]
- [33].Laing P. Diabetic foot ulcers. Am J Surg 1994;167:31S–6S. [DOI] [PubMed] [Google Scholar]
- [34].Friedman ND, Sexton DJ, Connelly SM, et al. Risk factors for surgical site infection complicating laminectomy. Infect Control Hosp Epidemiol 2007;28:1060–5. [DOI] [PubMed] [Google Scholar]
- [35].Maragakis LL, Cosgrove SE, Martinez EA, et al. Intraoperative fraction of inspired oxygen is a modifiable risk factor for surgical site infection after spinal surgery. Anesthesiology 2009;110:556–62. [DOI] [PubMed] [Google Scholar]
- [36].Fang A, Hu SS, Endres N, et al. Risk factors for infection after spinal surgery. Spine (Phila Pa 1976) 2005;30:1460–5. [DOI] [PubMed] [Google Scholar]
- [37].Cruse PJ. Polk HC, Stone HH. Some factors determining wound infection: a prospective study of 30,000 wounds. Hospital-acquired infectious surgery.. Baltimore: University Park Press; 1997. 75–105. [Google Scholar]
- [38].Cruse PJ, Foord R. A five year prospective study of 23469 surgical wounds. Arch Surg 1973;107:206–12. [DOI] [PubMed] [Google Scholar]
- [39].Bybee JD, Rogers DE. The phagocytic activity of polymorphonuclear leukocytes obtained from patients with DM mellitus. J Lab Clin Med 1964;64:1–3. [PubMed] [Google Scholar]
- [40].Chaudhary SB, Liporace FA, Gandhi A, et al. Complications of ankle fracture in patients with DM. J Am Acad Orthop Surg 2008;16:159–70. [DOI] [PubMed] [Google Scholar]
- [41].Wukich DK, Kline AJ. The management of ankle fractures in patients with DM. J Bone Joint Surg Am 2008;90:1570–8. [DOI] [PubMed] [Google Scholar]
- [42].Walter WP, Zwahlen M, Reck S, et al. The association of preoperative anemia and perioperative allogeneic blood transfusion with the risk of surgical site infection. Transfusion 2009;49:1964–70. [DOI] [PubMed] [Google Scholar]
- [43].Chavan DR, Kullolli G, Metan BB, et al. Primary actinomycosis of breast in post-menopausal woman: a case report. J Evol Med Dental Sci 2014;3:5279–82. [Google Scholar]
- [44].Horzic M, Kopljar M. Postoperative infections in colorectal cancer patients. Hepatogastroenterology 2005;52:101–4. [PubMed] [Google Scholar]
- [45].Ojo OA, Owolabi BS, Oseni AW, et al. Surgical site infection in posterior spine surgery. Nigerian J Clin Pract 2016;19:821–6. [DOI] [PubMed] [Google Scholar]
- [46].Amir Abdul-Jabbar, Steven Takemoto, Michael H. Weber, et al. Surgical Site Infection in Spinal Surgery. Spine surgery 37; (15):1340-1345. [DOI] [PubMed] [Google Scholar]
- [47].Dunne JR, Malone D, Tracy JK, et al. Perioperative anemia: an independent risk factor for infection, mortality, and resource utilization in surgery. J Surg Res 2002;102:237–44. [DOI] [PubMed] [Google Scholar]
- [48].Hill GE, Frawley WH, Griffith KE, et al. Allogeneic blood transfusion increases the risk of postoperative bacterial infection: a meta-analysis. J Trauma 2003;54:908–14. [DOI] [PubMed] [Google Scholar]
- [49].Best JT. Revision total hip and total knee arthroplasty. Orthop Nurs 2005;24:174–9. [DOI] [PubMed] [Google Scholar]
- [50].Lidwell OM, Lowbury EJL, Whyte W. Airborne contamination of wounds in joint replacement operations: the relationship to sepsis rates. J Hosp Infect 1983;4:111–31. [DOI] [PubMed] [Google Scholar]
- [51].Andersson AE, Bergh I, Karlsson J, et al. Traffic flow in the operating room: an explorative and descriptive study on air quality during orthopedic trauma implant surgery. Am J Infect Control 2012;8:750–5. [DOI] [PubMed] [Google Scholar]
- [52].Hambraeus A. Aerobiology in the operating room: a review. J Hosp Infect 1988;11Suppl A:68–76. [DOI] [PubMed] [Google Scholar]
- [53].Pryor F, Messmer PR. The effect of traffic patterns in the OR on surgical site infections. AORNJ 1998;68:649–60. [DOI] [PubMed] [Google Scholar]
- [54].Stauning MT, Bediako-Bowan A, Andersen LP, et al. Traffic flow and microbial air contamination in operating rooms at a major teaching hospital in Ghana. J Hosp Infect 2018;99:263–70. [DOI] [PubMed] [Google Scholar]
- [55].Lynch RJ, Englesbe MJ, Sturm L, et al. Measurement of foot traffic in the operating foom: implications for infection control. Am J Med Qual 2009;24:45–52. [DOI] [PubMed] [Google Scholar]
- [56].Lidwell OM, Lowbury EJL, Whyte W. Effect of ultraclean air in operating rooms on deep sepsis in the joint after total hip or knee replacement: a randomised study. BMJ 1982;285:10–4. [DOI] [PMC free article] [PubMed] [Google Scholar]


