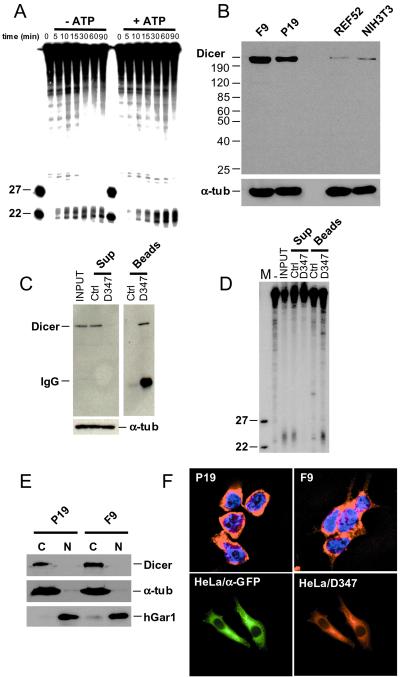
Figure 4.
In vitro cleavage of dsRNA and cellular localization of Dicer. (A) Processing of dsRNA to ≈22-nt RNAs in cytoplasmic extract of F9 cells. Reactions were incubated for the indicated time, in the presence or absence of ATP, and analyzed by PAGE-Urea. Extracts used for reactions without ATP were preincubated with hexokinase and glucose. Size markers are 22- and 27-nt oligoribonucleotides. (B) Western blot analysis of total cell extracts prepared from F9, P19, NIH 3T3, and REF52 cells. Proteins (≈75 μg) were separated in an 8% SDS/PAGE, and the blot was probed sequentially with α-Dicer D347 and anti-α-tubulin Abs. (C) Immunodepletion of Dicer from F9 cytoplasmic extract by incubation with D347 Ab-coated Protein A-Sepharose beads. As control (Ctrl), the extract was incubated with noncoated Protein A-Sepharose beads. Supernatant (Sup) and beads fractions were analyzed by Western blots, using D347 and anti-α-tubulin Abs. (D) Analysis of the dsRNA-processing activity present in the immunoprecipitation fractions described in C. dsRNA (123 bp) was used as a substrate. (E) Western blot analysis of cytoplasmic (C) and nuclear (N) fractions of EC cells using D347 Ab. Reprobing of the blot with Abs against the nuclear protein hGAR1 and the cytoplasmic α-tubulin indicated no major cross-contamination of fractions. (F Upper) Indirect immunolocalization of endogenous Dicer in F9 and P19 cells, using D347 Ab and Texas red-labeled α-rabbit secondary Ab. Nuclei are stained with 4′,6-diamidino-2-phenylindole. (Lower) Localization of the CFP-Dicer fusion protein in HeLa cells transfected with pCFP-Dicer, using α-GFP and α-Dicer D347 Abs.