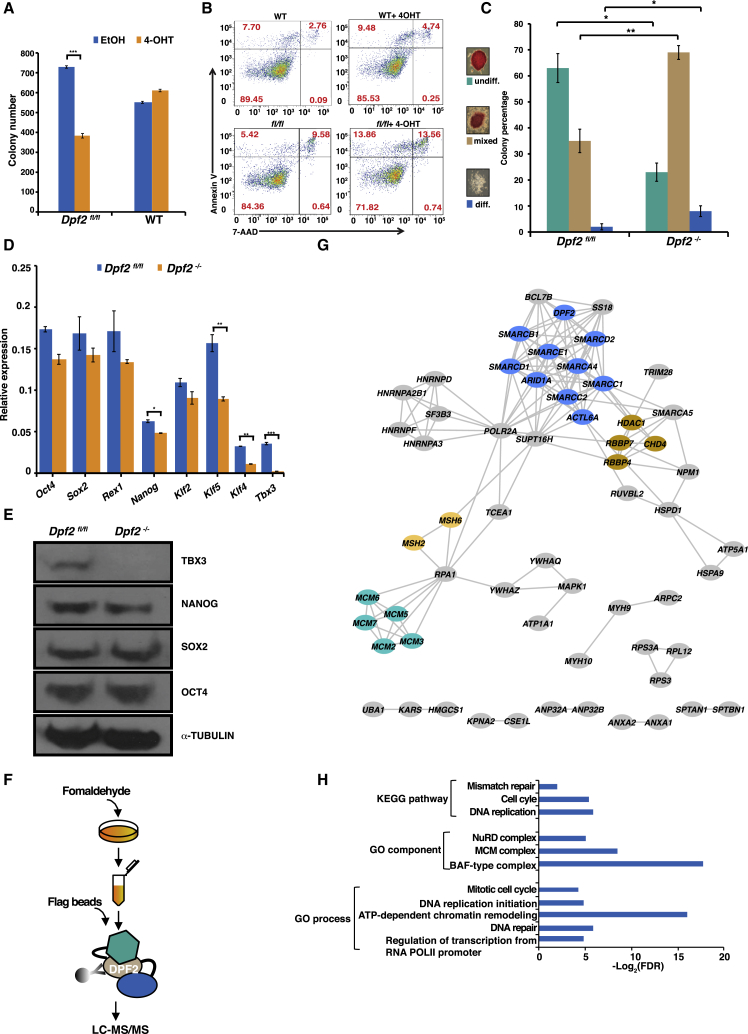
Figure 1.
Loss of Dpf2 Affects ESC Self-Renewal and Leads to Increased Apoptosis and Cell-Cycle Defects
(A) Quantification of a colony-formation assay for WT, Dpf2fl/fl, and Dpf2−/− mouse ESCs. Given is the mean of three replicates and the SD. ∗∗∗p < 0.001.
(B) Representative fluorescence-activated cell sorting (FACS) plots of Annexin V and 7-aminoactinomycin D (7-AAD) levels in Dpf2fl/fl and WT control ESCs. Percentages of cells with different apoptosis marker levels are indicated in brackets.
(C) Alkaline phosphatase (AP) staining assay for Dpf2fl/fl and Dpf2−/− ESCs. Colonies were scored as undifferentiated (undiff), mixed, and differentiated (diff). The mean and SD of three replicates is displayed. ∗p < 0.05, ∗∗p < 0.01.
(D) Transcript levels of pluripotency-associated genes in Dpf2fl/fl and Dpf2−/− ESCs based on qPCR.
(E) Western blot for OCT4, SOX2, NANOG, and TBX3 protein levels in Dpf2fl/fl and Dpf2−/− ESCs; α-TUBB served as a loading control.
(F) Schematic of the affinity purification of FLAG-tagged DPF2 in ESCs and the MS procedure.
(G) DPF2-interacting proteins annotated in the STRING (Search Tool for the Retrieval of Interacting Genes/Proteins) database. Subunits of the BAF (blue), NuRD (tan), MCM (green), and MSH complexes (yellow) are highlighted.
(H) GO term and KEGG (Kyoto Encyclopedia of Genes and Genomes) pathway enrichment of DPF2-interacting proteins. Selected terms are shown. FDR, false discovery rate.