Figure 3.
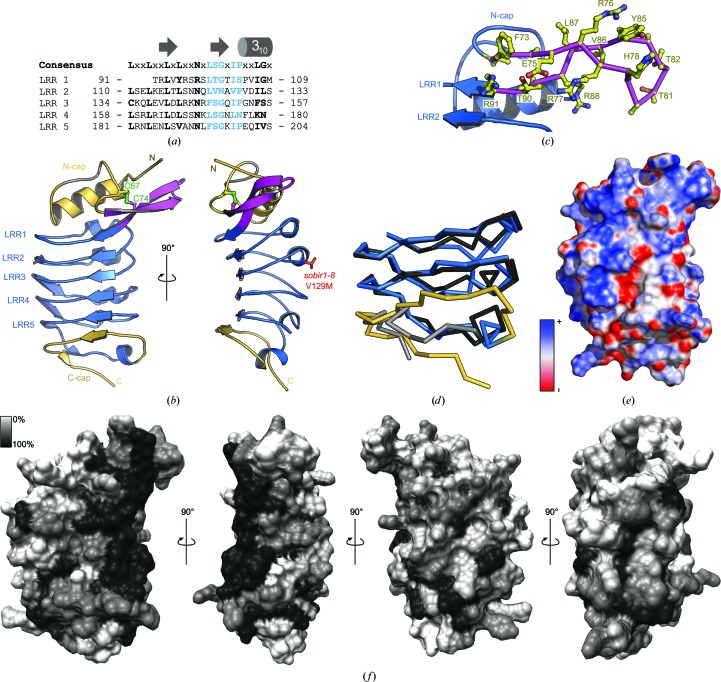
The SOBIR1 ectodomain harbours five LRRs and unusual capping domains. (a) Sequence alignment of the five SOBIR1 LRRs, with the canonical consensus sequence in black and the plant-specific LRR motif in blue. The LRR consensus sequence and a secondary-structure assignment calculated with DSSP (Kabsch & Sander, 1983 ▸) are shown alongside. (b) Front (left) and y-axis-rotated side (right) views of the SOBIR1 LRR domain (blue ribbon diagram), with N- and C-terminal capping domains shown in yellow, the SOBIR1-specific extended β-hairpin in magenta and a disordered loop in the N-terminal capping domain (residues 57–63) in grey. The disulfide bond (in green) and Val129 (in red), which is mutated to Met in sobir1-8, are shown in bond representations. (c) Close-up view of the extended β-hairpin (magenta Cα trace, with side chains shown in ball-and-stick representation in yellow) in the SOBIR1 ectodomain (blue ribbon diagram). (d) Cα traces of a structural superposition of the ectodomains of SOBIR1 (blue, C-terminal capping domain in yellow) and SERK3 (PDB entry 4mn8; Sun, Li et al., 2013 ▸; black, C-terminal capping domain in grey). (e) Surface representation of the SOBIR1 ectodomain coloured according to the electrostatic surface potential (blue, negative; red, positive). (f) Surface representation of the SOBIR1 ectodomain coloured according to SOBIR1 sequence conservation, comparing SOBIR orthologues from different plant species (sequences are shown in Fig. 4 ▸ c). Note the presence of a highly conserved patch at the outer edge of the inner surface, ranging from the N-terminal cap through LRRs 1–5.