Abstract
Introduction:
Management of periprosthetic infection in total hip arthroplasties is challenging, especially when there is severe loss of proximal femoral bone stock. When a 2-stage approach is used, either a static or an articulating spacer may be considered. Static spacers leave the patient with a flail leg, which can be very difficult with massive bone loss. The purpose of this study is to report a novel technique for articulating antibiotic spacers and report our results.
Materials and Methods:
We describe a technique for an articulating hip spacer in the setting of a large amount of proximal femoral bone loss using a locked intramedullary nail, modular femoral body, and an all-polyethylene constrained acetabular component. This technique allowed for mobilization of the patient without a flail leg. Four patients underwent 2-stage reconstruction, and the case series is reported here.
Results:
No complications occurred due to the spacer, and in all cases, a second reconstruction was later carried out after treatment with intravenous antibiotics. Three of 4 patients did well after 2-stage reconstruction, with 1 patient ultimately requiring an amputation.
Discussion:
We feel this technique improves upon previously reported large spacers due to the stability and maintenance of leg length.
Conclusion:
This technique offers a modular solution to address massive bone loss of the proximal femur in the face of periprosthetic joint infection.
Keywords: periprosthetic joint infection, articulating spacer, proximal femur bone loss, revision hip replacement, reimplantation
Introduction
Periprosthetic joint infections (PJIs) associated with total hip replacement are difficult problem to treat for arthroplasty surgeons, particularly in the patient with multiple revision surgeries resulting in the loss of proximal femur bone stock. Aggressive debridement of infected bone and soft tissue must be done to ensure the success of a 2-stage reconstruction. A 2-stage treatment of PJI with use of an articulating antibiotic spacer prosthesis has shown success rates of 95.3%.1 There have been several techniques described in the literature, including the use of articulating antibiotic spacers and total hip arthroplasty. This includes hand-molded antibiotic-loaded cement constructs containing metal endoskeletons, reimplanting explanted implants after auto cleaning, and recently a combination of static and articulating total femur spacers in patients with substantial bone loss.2-4 Intramedullary nails have also been described in constructing antibiotic cement spacers.5 In this report of a small case series as a single institution, we described a technique to manage a large amount of proximal femur bone loss while still constructing and articulating antibiotic spacer that maintains leg length, prevents a flail leg, and allows partial weight bearing.
Case History
Surgical Technique
The technique used to treat these challenging cases involves placing a modular articulating antibiotic spacer (MAAS) in the setting of infection and proximal femur bone loss. After aggressive resection of nonviable proximal femur, the femoral canal is reamed and irrigated for placement of an intramedullary nail. A guidewire was placed into the femur to the depth of the expected nail. This is marked and measured to be of an appropriate length of the nail. Some flexibility is still available proximally as several different lengths of proximal body are available. An intramedullary nail (Stryker T2 Femoral Nail, Kalamazoo, Michigan) size 14 mm width, is placed with appropriate length to recreate the resected proximal femur and locked distally with interlocking screws. The locking screws are placed with fluoroscopic assistance. A modular body (LINK MP Hip Reconstruction Prosthesis, Rockaway, New Jersey) is then secured to the cephalad portion of the intramedullary nail with antibiotic-loaded cement (Figure 1A). These modular bodies have 5 different sizes to allow for added length through the proximal body. The key component in ease of building this construct is the diameters of both the distal opening of the femoral modular body and the proximal intramedullary nail being 14 mm without having any Morse taper involved in the modular body. Polymethylmethacrylate cement is placed within the proximal body opening and around the entire prosthesis (Figure 1B). This may be loaded with antibiotics to help treat the periprosthetic infection.
Figure 1.
A, Clinical image demonstrating how the intramedullary nail fits inside the modular proximal body. B, Clinical image demonstrating covering the MAAS entirely with antibiotic loaded cement. MAAS indicates modular articulating antibiotic spacer.
Appropriate femoral anteversion can be recreated while the cement is allowed to cure. An all-polyethylene constrained acetabular cup (Stryker Trident, Kalamazoo, Michigan) is cemented in place, and the hip is reduced once the cobalt chrome femoral head is placed on the neck of the modular body. Cultures are followed, and the patient is discharged with intravenous (IV) antibiotics as recommended by our infectious disease colleagues for a minimum of 6 weeks. Patients are allowed to partial weight bear (up to 50%) on the MAAS in the postoperative period. Any further medical optimization can occur during this interim period. Patients then undergo completion of the 2-stage reconstruction once the treating surgeon is satisfied that the infection is cleared. Institutional review board approval was obtained prior to review of these clinical cases.
Case 1
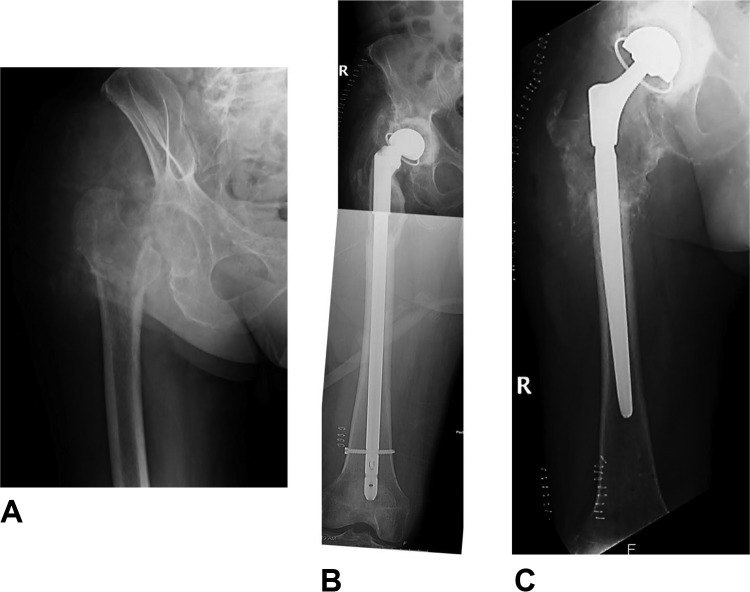
Patient 1 is a 67-year-old male who presented to our clinic 41 years after his initial hip replacement. His initial surgery was performed in 1974, with the latest revision for periprosthetic infection 2 years prior to presentation to our clinic. He had undergone 5 previous surgeries and had a history of PJI with Escherichia coli at an outside hospital. On initial presentation, the patient had loose-appearing femoral stem with severe osteolysis (Figure 2A). He had a history of hypertension, deep vein thrombosis with pulmonary embolism, and asthma. After preoperative medical optimization, the patient underwent resection of proximal femur down to the distal femur metaphysis and placement of an MAAS (Figure 2B). He was allowed to partial weight bear to his affected limb postoperatively and was treated with 6 weeks of IV ceftriaxone. Intraoperative cultures grew E coli again. He underwent completion of his 2-stage reconstruction after 17.2 weeks using a tapered revision hip prosthesis in combination with a femoral allograft. An intramedullary nail with lateral locking plate was used for allograft fixation distally (Figure 2C). Patient opted to delay his second stage reconstruction after his antibiotic holiday as he was functioning well with the spacer and had several social challenges he needed to resolve prior to reimplantation. Erythrocyte sedimentation rate and C-reactive protein normalized prior to this surgery. This construct was chosen for several reasons. First, we did not wish to proceed with total femur replacement as this was our last option and patient had a native pain-free knee. Second, other proximal femur constructs would have been technically challenging, given the short amount of femur distally along with the difficulty obtaining fixation in the metaphysis of the femur versus the diaphysis. He was allowed to touch down weight bear in the immediate postoperative period after his reimplantation and then advanced to weight bear as tolerated at a later date. At his latest follow-up 1.15 years after reimplantation, patient was ambulating with pain-free range of motion of the hip and no recurrence of infection. The allograft showed signs of incorporation with callus formation posteriorly, and the implant remained well fixed.
Figure 2.
Radiographic images of patient 1 presenting radiographs (A&B), interval placement of MAAS (C), and final reconstruction completed using a combination of femoral allograft, intramedullary nail, and bridging lateral locking plate (D).
Case 2
Patient 2 is a 52-year-old female who presented with a current diagnosis of PJI with a history of pseudotumor resection after metal-on-metal total hip arthroplasty (Figure 3A). She had 6 previous surgeries, complicated by 3 dislocations after her most recent revision. Diagnosis of PJI was confirmed with hip aspiration that grew Enterococcus faecalis. She had a past medical history of obesity (body mass index 40 kg/m2), seizure disorder, hypertension, lupus, fibromyalgia, and hypothyroidism. A MAAS (Figure 3B) was placed after aggressive resection of infected bone and soft tissue. This included removing the proximal 330 mm of femur en bloc after cutting the femur distal to the previous implant. She was allowed to partial weight bear postoperatively and was treated with 6 weeks of IV penicillin G and ciprofloxacin. Intraoperative cultures grew E faecalis and Staphylococcus epidermidis. She was reimplanted after 9.4 weeks using a proximal femoral replacement prosthesis (Figure 3C). She was allowed to weight bear as tolerated immediately after reimplantation postoperatively. Doxycycline by mouth was used prophylactically for 30 days postoperatively as prophylactic antibiotics have been shown to improve survival after the second stage reimplantation.6 At most recent follow-up 1 year after reimplantation, she was infection-free with no signs of loosening radiographically and a well-healed incision. Clinically, she was doing well, ambulating unassisted although with a Trendelenburg gait. She had no experienced any complications postoperatively, such as dislocations.
Figure 3.
Radiographic images of patient 2 presenting radiographs (A), interval MAAS (B), and final reconstruction completed with a proximal femoral replacement (C). MAAS indicates modular articulating antibiotic spacer.
Case 3
Patient 3 is an 83-year-old male who developed a PJI after treatment of a femoral neck fracture. His initial surgery was 21 weeks prior to presentation to our clinic. He had undergone 8 surgeries in the interim. These included a 2-stage reconstruction for PJI. He presented to our clinic after removal of the previous prosthesis with a flail leg (Figure 4A). Prior to his initial surgery, he was unable to walk 50 feet by himself and mostly just transfer to and from a wheelchair. His past medical history included stage III chronic kidney disease, Parkinson disease, hypertension, and deep vein thrombosis. No fluid was obtained on hip aspiration. Magnetic resonance imaging of the hip showed osteomyelitis to the proximal 6 cm of femur. After resection, he was treated with an MAAS (Figure 4B) and was allowed to partial weight bear postoperatively. He was also treated with 6 weeks of IV ceftriaxone and vancomycin per recommendations from our infectious disease colleagues. Intraoperative cultures showed no growth with previous cultures growing methicillin-resistant Staphylococcus aureus. After allowing time to recover from 9 surgeries in 5 months, he was reimplanted after 35 weeks with a distal diaphyseal fitting modular tapered stem hip revision prosthesis (Figure 4C). At most recent follow-up 8 months postoperatively, patient was ambulating 300 feet with assisted device. There was some heterotopic ossification (Brooker class III) that formed around the proximal femur. Patient maintained hip flexion to 70° along with 30° of internal and external rotation. His range of motion was painless.
Figure 4.
Radiographic images of patient 3 presenting radiographs (A), interval MAAS (B), and final reconstruction done with a diaphyseal fitting modular tapered stem femoral component (C). MAAS indicates modular articulating antibiotic spacer.
Case 4
Patient 4 is a 43-year-old female who presented to our clinic with a left chronically infected native hip and ipsilateral infected native knee. She had undergone 2 irrigation debridements at an outside hospital prior to referral for her to our clinic. She had a past medical history of poorly controlled type 1 diabetes (hemoglobin A1c of 9.7 on presentation), treated lumbar osteomyelitis, hypertension, and hyperlipidemia. She was initially treated with standard articulating hip and static knee antibiotic spacers (Figure 5A) along with IV antibiotics. She continued to show drainage from the incisions and was found to have osteomyelitis of the femur in between the 2 spacers. We elected to treat her with an MAAS (Figure 5B) that extended to the tibia. This case is unique to the previous cases as it begins as infections in native joints as well as the entire femur was removed. The modular body was still cemented into the femoral nail, which was locked distally into the tibia. Intraoperative cultures during placement of the MAAS were negative. However, given previous positive cultures and drainage from wounds, our infectious disease colleagues recommended continuing broad-spectrum antibiotics. After 6 weeks of IV vancomycin and by mouth ciprofloxacin, the patient was reimplanted with a total femur implant (Figure 5C) 20.5 weeks after placement of the MAAS. Attempts were made to improve her glucose levels by medicine with only mild improvement in the patient’s hemoglobin A1c. Due to the amount of bone loss from her tibia, tibial stem and metaphyseal cone were used in addition to the total femur. This resulted in a cortical breach while placing the tibial stem. This was treated with a prophylactic plate. Patient was made touch down weight bearing in the postoperative period due to this cortical breach. She was taken to the operating room for open reduction of posterior hip dislocation with placement of constrained liner at 5 weeks postoperatively. She then developed a recurrence of PJI and underwent irrigation and debridement with polyethylene exchange 1 month after the open reduction. Her infection persisted; however, she underwent hip disarticulation 3 months after placement of the total femur. At last clinical follow-up, the patient did have significant phantom pain but had healed her amputation incision with no signs of infection.
Figure 5.
Radiographic images of patient 4 presenting radiographs (A), interval MAAS (B), and final reconstruction with total femur placement and prophylactic proximal tibia plate (C). MAAS indicates modular articulating antibiotic spacer.
Discussion
We present a technique to allow for an articulating spacer in cases with large femoral bone loss, including complete loss of the entire femur. Using an MAAS allows the treating surgeon to be aggressive with resection of bone without fear that he or she will not be able to place an articulating antibiotic spacer. In comparison with the previously described methods of custom articulating antibiotic spacers, there is greater stability and maintenance of leg length obtained from a locked intramedullary nail with modular body construct. Maintaining tension to the soft tissues allows for easier reimplantation once the infection has been cleared. The articulating portion of the MAAS provides less pain and increased mobility during the time from explantation to reimplantation. Furthermore, with allowance of partial weight bearing, patients may also be at decreased risk for further bone loss by way of disuse osteoporosis.4 The MAAS was well tolerated by the patients treated in this series. As seen by the variable time to reimplantation, the stability and durability of the construct allows it to be left in place for a prolonged period of time if needed. No episodes of fracture or dislocation were noted in this series. There has been a previous case report of mating a standard femoral intramedullary nail with a premade hip antibiotic cement spacer for the treatment of proximal femur bone loss.7 We feel the MAAS improves upon that construct in terms of stability and comfort using a cemented all-polyethylene acetabular component versus a molded cement spacer proximally in the case report.
Use of this construct allows for massive endoprosthesis reconstruction after explantation. Massive endoprosthesis reconstruction has shown promising results for this difficult patient population with a 2-year survival rate of 72%.8 Two of the 4 patients in this series were treated with endoprostheses, with the remaining 2 using modular diaphyseal components and allograft.
This patient population is obviously at high risk for developing complications as they all had multiple previous surgeries and/or multiple comorbidities. We have frank discussions with these patients preoperatively, specifically about the risk of amputation if the infection is unable to be cleared after placement of MAAS and IV antibiotics. We discuss with them that the hope of a 2-stage reconstruction with an MAAS is to avoid hip disarticulation, as reported ambulation rates after hip disarticulation are only as high as 43% with a prosthesis.9
Conclusion
In summary, we described the surgical technique that provides an alternative to the limited options currently available to manage this difficult problem. We believe this MAAS is a reasonable surgical option for the treatment of periprosthetic hip infections with significant proximal femur bone loss.
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Wesley Mayes, MD  https://orcid.org/0000-0002-4603-1617
https://orcid.org/0000-0002-4603-1617
References
- 1. Canham CD, Walsh CP, Incavo SJ. Antibiotic impregnated total femur spacers: a technical tip. Arthroplasty Today. 2018;4(1):65–70. doi:10.1016/j.artd.2017.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Barrack RL. Rush pin technique for temporary antibiotic-impregnated cement prosthesis for infected total hip arthroplasty. J Arthroplasty. 2002;17(5):600–603. doi:10.1054/arth.2002.32698. [DOI] [PubMed] [Google Scholar]
- 3. Ries MD, Jergesen H. An inexpensive molding method for antibiotic-impregnated cement 208 spacers in infected total hip arthroplasty. J Arthroplasty. 1999;14(6):764–765. doi:10.1016/S0883-5403(99)90234-6. [DOI] [PubMed] [Google Scholar]
- 4. Hofmann AA, Goldberg TD, Tanner AM, Cook TM. Ten-year experience using an articulating antibiotic cement hip spacer for the treatment of chronically infected total hip. J Arthroplasty. 2005;20(7):874–879. doi:10.1016/j.arth.2004.12.055. [DOI] [PubMed] [Google Scholar]
- 5. Ben-Lulu O, Farno A, Gross AE, Backstein DJ, Kosashvili Y, Safir OA. A modified cement spacer technique for infected total hip arthroplasties with significant bone loss. J Arthroplasty. 2012;27(4):613–619. doi:10.1016/j.arth.2011.06.031. [DOI] [PubMed] [Google Scholar]
- 6. Frank JM, Kayupov E, Moric M, et al. Oral antibiotics reduce reinfection after two-stage exchange: a multicenter, randomized controlled trial. Clin Orthop Relat Res. 2017;475(1):56–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Kamath AF, Austin D, Lee GC. Mating of a PROSTALAC spacer with an intramedullary nail for reconstruction of an infected interprosthetic femoral shaft fracture: a case report. J Orthop Surg (Hong Kong). 2012;20(2):263–268. [DOI] [PubMed] [Google Scholar]
- 8. Alvand A, Grammatopoulos G, de Vos F, et al. Clinical outcome of massive endoprostheses 217 used for managing periprosthetic joint infections of the hip and knee. J Arthroplasty. 2018;33:829–834. doi:10.1016/j.arth.2017.09.046. [DOI] [PubMed] [Google Scholar]
- 9. Kralovec M, Houdek M, Andrews K, Shives T, Rose P, Sim F. Prosthetic rehabilitation 220 after hip disarticulation or hemipelvectomy. Am J Phys Med Rehab. 2015;94(12):1035–1040. doi:10.1097/PHM.0000000000000292. [DOI] [PubMed] [Google Scholar]