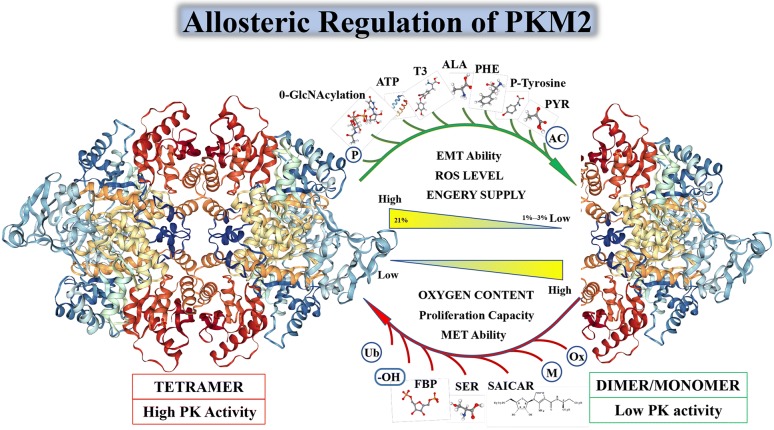
Fig. 2.
Relationship between PKM2 enzyme activity and spatial conformation. The transition between PKM2 dimers and tetramers is allosterically regulated by endogenous and exogenous activators and inhibitors. PKM2 has PK enzyme activity only when it serves as a tetramer. PKM2 is activated by the glycolytic intermediate products named fructose 1,6-bisphosphate (FBP). It can also be activated by the allosteric effects of serine and succinylaminoimidazolecarboxamide ribose-50 phosphate SDH succinate dehydrogenase (SAICAR) [54, 55]. The PK enzymatic activity of PKM2 can be inhibited by many endogenous inhibitors and cellular signaling events including 0-GlcNAcylation, pyruvate (PYR), P-tyrosine (P-TYR), phenylalanine (PHE), alanine (ALA), adenosine triphosphate (ATP), and thyroid hormone T3 [56–58]. In addition, due to the number of related molecules involved in PKM2’s post-translational modification (PTM), I will not list them in Fig. 2, but in the form of Tables 2 and 3 in the fourth part of this article “Interaction of PKM2 with other proteins”