Fig. 3.
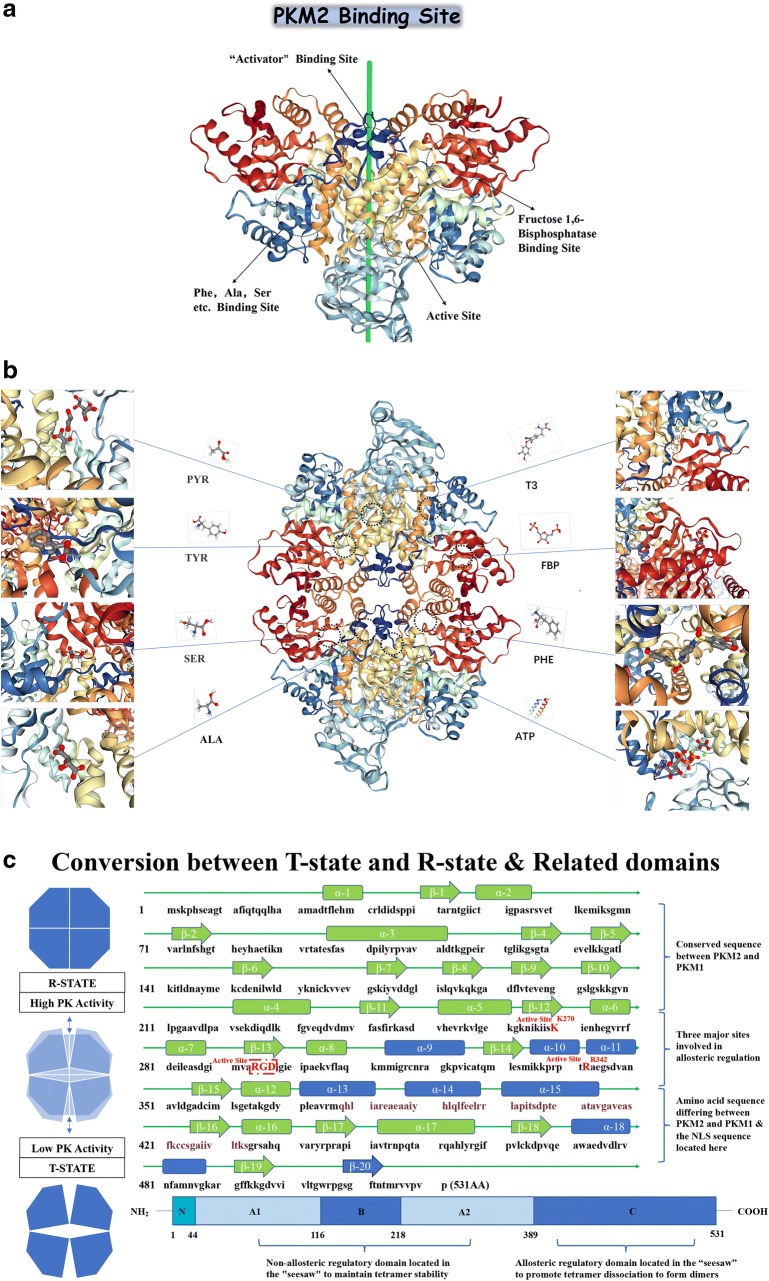
The Specific Site of PKM2 Allosteric Regulation and The Amino Acid Sequence of PKM2. It is now accepted that the PKM2 in the tetrameric state has an allosteric regulatory domain within its spatial structure, forming a pattern similar to the seesaw, when some allosteric regulators are inserted into the spatial structure involved in PKM2 allosteric regulation. After the domain (a), the tetramer PKM2 can be transferred from a compact state (R-state) to a loose state (T-state) and finally disassembled into a dimeric form [59]. When these allosteric regulators bind to PKM2, they will change the spatial conformation of PKM2, and affect the electrostatic force inside the molecule, and then affect the transition state of PKM2. The allosteric form a stable and compact PKM2 R-state to form a tetramer and perform PK enzyme activity. After allosteric adjustment the PKM2 forms a loose and unstable T-state, and eventually breaks the linked fragment in the tetramer to form a PKM2 dimer form with lower PK enzymatic activity. When PKM2 is allosteric to form a dimer, it will expose the active region inside the molecule, although the PK enzyme activity is low, it has protein activation activity [60]. In b we specifically list the each participating allosteric regulates the binding site of the small molecule and the binding site of the activator in the PKM2 protein spatial structure [61]. In Fig. 3c we simply describe the seesaw structure of PKM2 and specifically identify the specific spatial domains that participate in the seesaw pattern: α-9 and 10 and 11 and 13 and 14 and 15 and 18 and β-20 which marked with blue text in c. The residues at the active site which was mentioned in a are highlighted by red box in c. Residue Arg342 and Residue Lys342, which is responsible for active site “RGD” stabilization is colored in red. There is one point to be noted here: whether PKM2 has protein kinase activity or not, there is a negative attitude, however in some of the researchers’ experiments described later, phosphorylation was indeed found [62]