Figure 4.
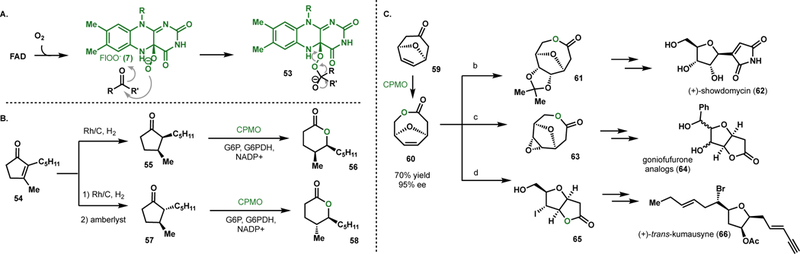
Reactions with BVMOs. (A) General mechanism for BVMOs. (B) Aerangis lactone synthesis featuring a stereoselective BV oxidation by cyclododecanone monooxygenase (CDMO) or cycopentanone monooxygenase (CPMO). (C) Chemoenzymatic synthesis of tetrahydrofuran-based natural products via a chiral intermediate generated by CPMO-mediated Baeyer-Villager oxidation a) OsO4, NMO, DCM, rt, then acetone–AlCl3, 0 °C to 40 °C, 47%; b) mCPBA, DCM, reflux, 98%; c) MeCN, H2O, KOH, rt, then I2–KI, 40 °C, dark, 75%.
