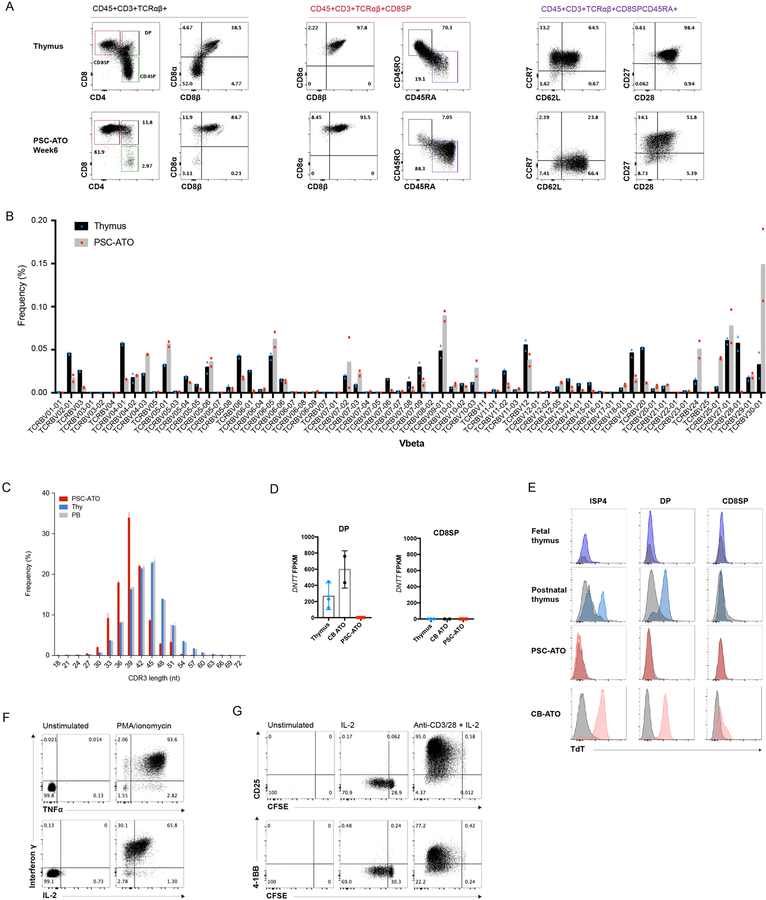
Figure 3: Maturation, TCR diversity and function of PSC derived T cells generated in PSC-ATOs.
(A) Representative flow cytometry analysis of T cell maturation markers on CD3+TCR + cells from H1 PSC-ATOs at week 7, demonstrating a conventional T cell phenotype (CD8 ) and generation of mature (CD45RA+CD45RO-) nave T cells. Parent gates are indicated above each panel. Human postnatal thymocytes are shown for comparison (top row). Data are representative of 8 independent experiments. (B) TCR V diversity in nave CD8SP T cells isolated from H1 PSC-ATOs (gray bars, n=2) compared to nave CD8SP from postnatal thymi (black bars, n=2) by deep sequencing of TCR V CDR3 regions. Frequency of cells expressing each V segment is shown. (C) TCR V CDR3 lengths in nave CD8SP T cells isolated from H1 PSC-ATOs (red bars, n=2), postnatal thymi (blue bars, n=2) and adult peripheral blood (gray bars, n=2) assessed by deep sequencing. (D) Expression of DNTT (TdT) by RNA sequencing in DP cells (left) and CD8SP T cells (right) isolated from H1 PSC-ATOs (n=3) compared to the same populations isolated from postnatal thymi (n=3) or ATOs initiated with human cord blood CD34+ HSPCs (CB-ATOs) (n=2). Gene expression is quantified as fragments per kilobase of transcript per million reads (FPKM). (E) Representative intracellular flow cytometry analysis of TdT expression gated on ISP4, DP and CD8SP populations from the human fetal and postnatal thymus (n=1 and n=2, respectively); H1 PSC-ATOs (n=3), and CB-ATOs (n=2). Isotype staining controls are shown in gray for each plot. (F) Polyfunctional cytokine production by H1 PSC-ATO derived CD8SP T cells after treatment with PMA/ionomycin. Data are representative of 3 independent experiments. (G) Proliferation (as measured by the dilution of CFSE) and upregulation of activation markers CD25 and 4–1BB by H1 PSC-ATO derived CD8SP T cells after 5 days of treatment with media only, IL-2, or anti-CD3/CD28 beads plus IL-2. Data are representative of three independent experiments.