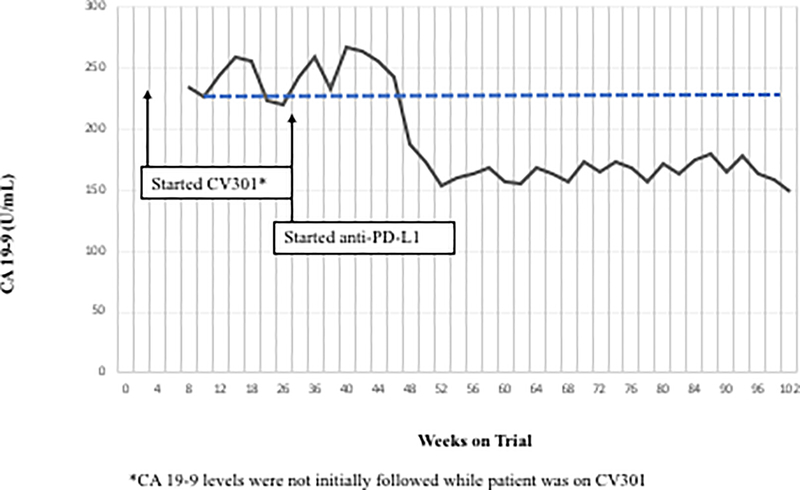
Figure 2.
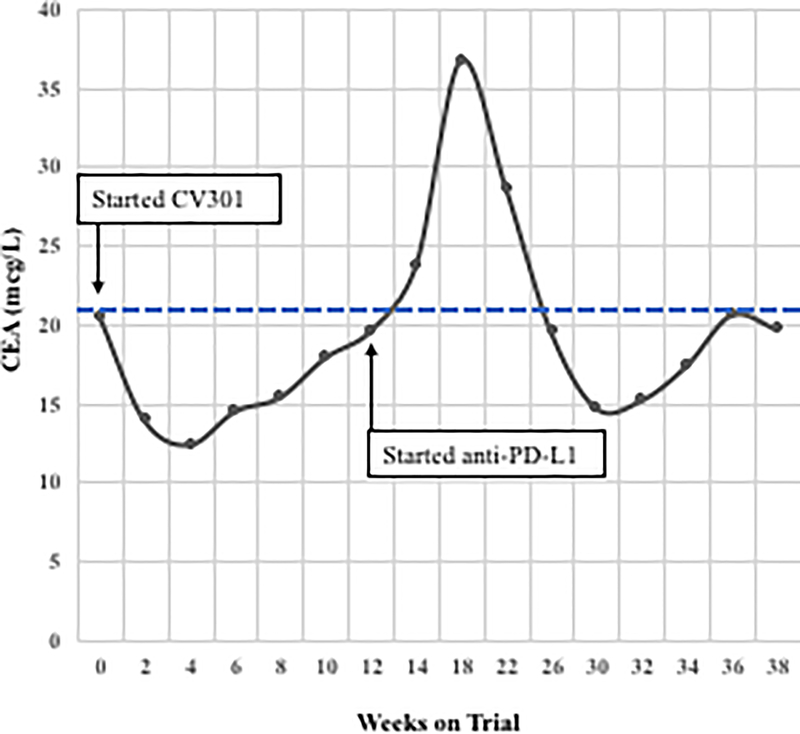
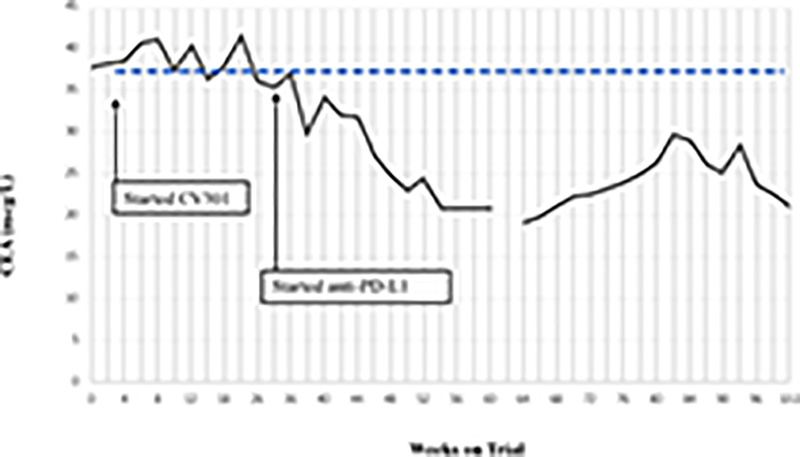
Two patients with KRAS mutation, MSS mCRC had a ≥ 35% decrease in tumor markers associated with prolonged stable disease after treatment with BN-CV301 followed by an anti-PD-L1 antibody.
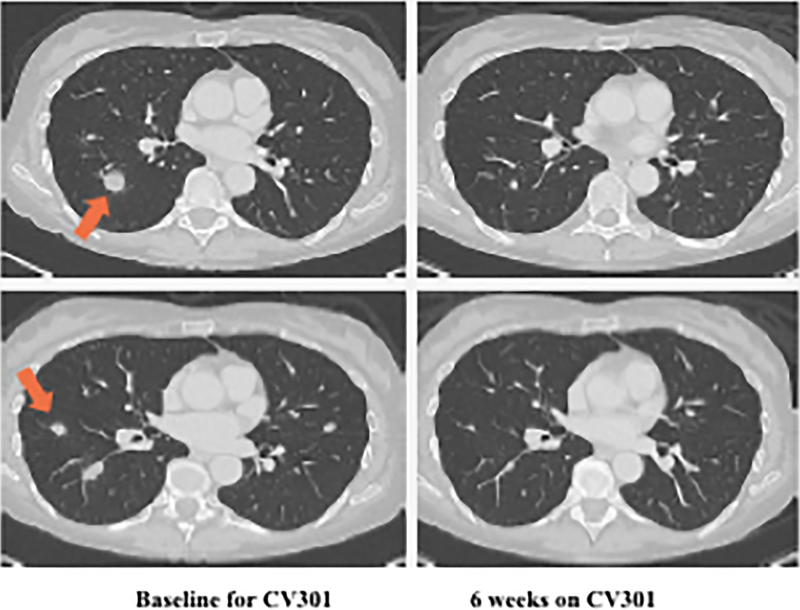
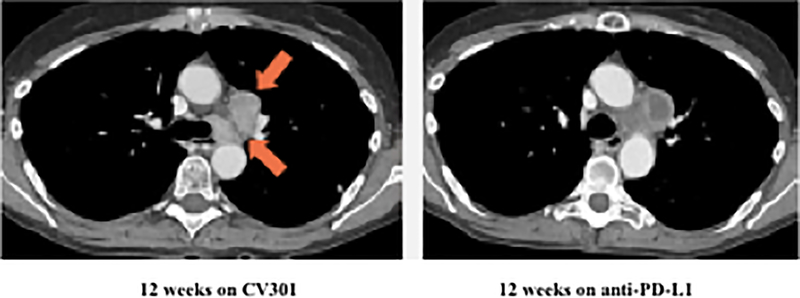
Patient #1: A 62-year-old female with KRAS MT, MSS CRC with progressive disease despite 6 prior regimens (DL=1). She had an initial decrease in CEA (A) and an unconfirmed partial response at the first restaging (6 weeks; B) followed by growth of a non-target lesion (new mediastinal adenopathy) at the 12-week restaging. The patient then enrolled on an anti-PD-L1 trial and experienced a subsequent decrease in CEA as well as a radiographic response (C) to treatment with necrosis of mediastinal adenopathy and decreasing tumor markers at week 12 of treatment with the anti-PD-L1 antibody. This patient had stable disease for 43 weeks while on an anti-PD-L1 antibody. Patient #2: A 54-year-old female with KRAS MT, MSS CRC with progressive disease despite 8 prior regimens (DL=1). While on BN-CV301 trial, CEA and tumor burden were stable but the patient eventually developed progressive disease at 26 weeks. Patient was then enrolled on an anti-PD-L1 trial and experienced a subsequent decrease in tumor markers (D and E). Radiographically the patient has continued stable disease on the anti-PD-L1 antibody ongoing at 71 weeks. Prior trials have found a median progression-free survival of 10 weeks (2.2 months) in patients with MSS, mCRC who receive an anti-PD-L1 antibody. Blue dotted lines represent baseline tumor markers.