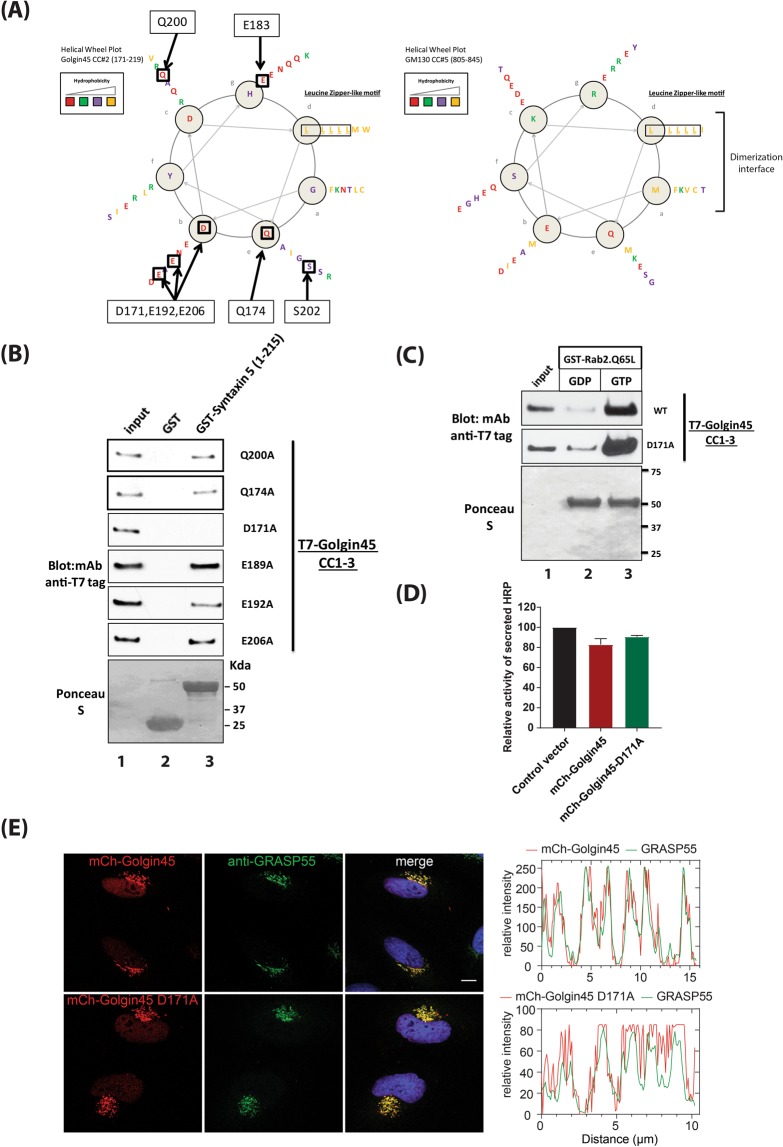
Figure 2.
Alanine scanning mutagenesis of Golgin45 Leucine zipper motif reveals that D171A mutant abrogates Golgin45 binding to the N-terminal regulatory domain of Syntaxin5, but not to Rab2-GTP. (A) Helical wheel plots showing the positions of homologous amino acid residues found in the leucine zipper-like motifs of GM130 and Golgin45. Boxed residues indicate identical or similar AA residues; (B) Alanine scanning mutagenesis of common AA residues within Leucine zipper motif of Golgin45 and GST pull-down assays were used to identify a specific point mutant of Golgin45 that fails to bind recombinant Syntaxin5. D171A mutation significantly abrogates Golgin45 CC domain binding to GST-Syntaxin5 (1–215) in pull-down assays. (C) GST-Rab2.Q65L recombinant proteins were used to test if Golgin45.D171A mutant show any change in their interaction, compared to the WT Golgin45. Both the WT and D171A.Golgin45 equally bound to Rab2 in GTP-dependent manner, suggesting Syntaxin5-binding deficient mutant is not affected in Rab2 interaction in vitro. (D) Expression of mCherry-Golgin45.D171A does not significantly affect secretion of soluble secretory cargo, ss-HRP, in HeLa cells, compared to the cells transfected with vector-transfected control, while expression of mCherry-Golgin45 WT very moderately inhibits secretion of ss-HRP. (E) D171A mutation does not affect Golgin45 targeting to the Golgi and its co-localization with endogenous GRASP55 in HeLa cells, compared to that of Golgin45 WT. Line analysis graph shows that both the WT and the mutant mCherry-Golgin45.D171A co-localize well with anti-GRASP55 stained Golgi area. Bar = 10 μm.