Table 2.
Pharmacological characteristics of CDK4/6 inhibitors
| Palbociclib (pd-0332991; ibrance, pfizer) | Abemaciclib (ly2835219; verzenio, lilly) | Ribociclib (lee011; kisquali, novartis) | |
|---|---|---|---|
| Chemical structure |
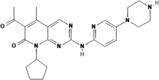
|
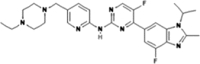
|
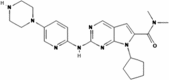
|
| Ic50 (nm) | |||
| Cdk4-cyclin d1 | 11 | 2 | 10 |
| Cdk6-cyclin d1-2-3 | 15 | 10 | 39 |
| Absorption | Increased with high-fat, high-calorie food | NR | NR |
| Distribution | 2583 L | 690.3 L | 1090 L |
| Metabolism | Liver (cyp3a and sult2a1) | Liver (cyp3a4) | Liver (cyp3a4) |
| Excretion | Feces (~74%) | Feces (~81%) | Feces (~69%) |
| Urine (~18%) | Urine (~3%) | Urine (~23%) | |
| Bioavailability | 46% | 45% | NR |
| Time to peak (hours) | 6–12 | 8 | 1–4 |
| Half-life elimination (hours) | 29 ± 5 | 18.3 | 30–55 |
| Protein binding | ~85% | 93–98% | ~70% |
| Mtd/rp2d | 125/125 mg/day on a 21-of-28-day schedule | 200 mg twice daily | 900/600 mg/day on a 21-of-28-day schedule |
| Dlts | Neutropenia | Fatigue | Neutropenia, asymptomatic thrombocytopenia, mucositis, pulmonary embolism, hyponatremia, QTcF, prolongation (> 500 ms), increased creatinine |
| Route of administration | Oral | Oral | Oral |
| Recommended dose | 125 mg once daily for 21 days, followed by 7 days off, repeat every 28 days | 150 mg twice daily | 600 mg once daily for 21 days, followed by 7 days off, repeat every 28 days |
| Dose modifications | |||
| Renal impairment | |||
| Crcl > 15 ml/min | No dosage adjustament | No dosage adjustament | No dosage adjustament |
| Crcl ≤ 15 ml/min | NR | NR | NR |
| Esrd | NR | NR | NR |
| Hepatic impairment* | |||
| Mild/moderate | No dosage adjustament | No dosage adjustament | No dosage adjustament |
| Severe | Reduce dose to 75 mg | Reduce dose to once daily | Reduce dose to 400 mg |
Chemical structures are available online at: https://pubchem.ncbi.nlm.nih.gov. Data about pharmacological characteristics of the three drugs are available online at: https://www.drugs.com
DLT dose-limiting toxicity, ESRD end-stage renal disease, IC50 half maximal inhibitory concentration, MTD maximum tolerated dose, NR not reported, RP2D recommended phase II dose
*Mild, moderate, and severe hepatic impairment refers to Child-Pugh classes A, B, and C, respectively
