ABSTRACT
To meet the high calcium (Ca) demand during eggshell biomineralization (2 g of Ca per egg), laying hens develop specific metabolic regulations to maintain Ca homeostasis. The intake of Ca, its solubilization, and absorption capacity are enhanced at sexual maturity (SM). A better knowledge of the intestinal Ca transporters involved in their variations at this stage could indicate new nutritional strategies to enhance Ca digestive utilization. Transcellular Ca absorption pathway and its major player calbindin-D 28 K (CALB1) mediate a saturable transport, which has been extensively described in this model. Conversely, a contribution by the paracellular pathway involving non-saturable Ca transport through intercellular tight junction has also been suggested. The aim of the present study was to identify candidate genes of these two pathways and their patterns of expression, in immature pullets (12, 15, and 17 wk old) and mature laying hens (23 wk old) in the duodenum, jejunum, and ileum. Using RT-qPCR, this study identifies 3 new candidate genes for transcellular, and 9 for paracellular Ca transport. A total of 5 candidates of the transcellular pathway, transient receptor potential cation channels subfamily C member 1 (TRPC1) and M member 7 (TRPM7); CALB1 and ATPase plasma membrane Ca2+ transporting 1 (ATP2B1) and ATPase plasma membrane Ca2+ transporting 2 (ATP2B2) were enhanced with age or after SM in the duodenum, the jejunum or all 3 segments. A total of 4 candidates of the paracellular pathway Claudin 2 (CLDN2) and tight junction proteins 1, 2, and 3 (TJP1, TJP2 and TJP3) increased in the small intestine after SM. Additionally, CALB1, ATP2B2, and CLDN2 were overexpressed in the duodenum or the jejunum or both segments after SM. The enhanced expression of candidate genes of the paracellular Ca pathway after SM, supports that the non-saturable transport could be a mechanism of great importance when high concentrations of soluble Ca are observed in the intestinal content during eggshell formation. Both pathways may work cooperatively in the duodenum and jejunum, the main sites of Ca absorption in laying hens.
Keywords: laying hen, Ca absorption, gene expression, transcellular pathway, paracellular pathway
INTRODUCTION
In laying hens, eggshell formation represents a fast mineralization process, which takes place daily in the uterus, with the deposition of Calcium (Ca) carbonate (Nys and Le Roy, 2017). Large amounts of Ca, about 2 g per egg, must be supplied to the uterine gland, mainly during the dark period, when the hen is not consuming feed (Bar, 2009). To meet this demand, the hen adapts its daily Ca intake (4.2 to 4.6 g/d) and retention, notably by optimizing its intestinal Ca absorption during the ovulatory cycle (Hurwitz and Bar, 1965; Bar, 2009). Before the dark period, laying hens exhibit a specific appetite for Ca and crop dilatation favors enhanced acid secretion in the proventriculus, increasing Ca solubilization (Mongin and Sauveur, 1974; Sauveur and Mongin, 1983; Guinotte et al., 1995). These mechanisms facilitate the intense Ca absorption during eggshell formation (Hurwitz and Bar, 1965, 1969). Incorporation of coarse calcium carbonate into the diet can improve Ca retention by extending the supply of soluble Ca in the intestine during the dark period (Guinotte et al., 1995). When the content of soluble Ca in the intestine becomes limiting, Ca is mobilized from the medullary bone formed before the onset of laying which constitutes a labile source of Ca (Castillo et al., 1979; Sauveur and Mongin, 1983; Kerschnitzki et al., 2014). Any imbalance in Ca homeostasis impairs the quality of the eggshell or bone mineralization and strength, and Ca deficiency is the primary cause of osteoporosis. It coincides with a progressive loss of mineralized structural bone which leads to bone fragility and susceptibility to fracture (Whitehead, 2004). It represents a common problem in laying flocks causing both financial losses and welfare issues which can occur as soon as 45 wk old (Kim et al., 2012; Olgun and Aygun, 2016). Many dietary factors interacting with Ca digestibility and retention have already been studied, including phytic acid content (Jalal and Scheideler, 2001; Lim et al., 2003), Ca to Phosphorus (P) ratio (Keshavarz, 1986; Lim et al., 2003) and vitamin D3 levels (Plaimast et al., 2015). An improved knowledge about mechanisms involved in intestinal Ca absorption in laying hens could help to define new nutritional strategies to enhance its digestive utilization. In laying hens, Ca absorption occurs in the small intestine, and increases mainly in the duodenum and jejunum during eggshell formation, due to high Ca absorption rates (Hurwitz and Bar, 1965; Hurwitz and Bar, 1968). Both active (saturable) and passive (non-saturable) Ca transport have been reported during the eggshell formation period (Hurwitz and Bar, 1969; Nys and Mongin, 1982). Active Ca transport, mediated by the transcellular Ca absorption pathway, has been intensively studied and several of its components have been described, in contrast to the mechanisms of passive Ca transport. The major player in the transcellular pathway is the vitamin D3-induced Ca-binding protein, named calbindin-D 28K (CALB1), initially discovered in the chick's intestine (Wasserman and Taylor, 1966). Other members of the transcellular pathway, in charge of enterocyte Ca2+ entry Transient receptor potential cation channel subfamily V member 6 (TRPV6) and extrusion ATPase plasma membrane Ca2+ transporting 1 (ATP2B1), ATPase plasma membrane Ca2+ transporting 2 (ATP2B2), ATPase plasma membrane Ca2+ transporting 4 (ATP2B4), and Solute Carrier Family 8 Member A1 (SLC8A1) have also been detected in the duodenum of laying hens (Yang et al., 2011; Jonchère et al., 2012). However, a complete description of these candidates is lacking in the different segments of the small intestine of the laying hen.
Controversies have also been raised about TRPV6 expression in the small intestine of laying hens or chickens, suggesting that other candidates should be investigated (Yang et al., 2011; Proszkowiec-Weglarz et al., 2019).
Duodenal CALB1 concentration correlates positively to the percentage of net Ca absorption, but negatively to the daily amount of Ca absorbed (Bar et al., 1979). In addition, no variation of duodenal CALB1 concentration occurs during the ovulatory cycle (Nys et al., 1992). These observations support the hypothesis that a non-saturable transport can contribute to Ca absorption in laying hens having high Ca intake.
In mammalian species, passive Ca transport relies on the paracellular pathway. It consists of intercellular tight junctions (TJ) composed of transmembrane (claudin: CLDN, junctional adhesion molecule: JAM and occludin: OCLN) and cytosolic (tight junction proteins: TJP), proteins which form pores for the permeation of ions and water. The nature of the CLDN determines the specificity of pores for Ca transport and overexpression of Claudin 2 (CLDN2) or CLDN12 enhances Ca transfer through intestinal epithelial cells in vitro (Fujita et al., 2008). Intestinal Ca transport is mediated by the paracellular pathway when dietary Ca is high and by the transcellular pathway when dietary Ca is low (Christakos et al., 2014). Moreover both pathways may cooperate, as invalidation of Calbindin-D 9K in mice induces an overexpression of CLDN2 and 15 (Hwang et al., 2013). The paracellular pathway has not been studied so far in the laying hens. If present and functional it could participate in the intestinal Ca transport, when they exhibit high Ca intake to match the high Ca demand for eggshell mineralization. As a first step to support this hypothesis, the present study identifies candidate genes encoding transmembrane and cytosolic proteins of the TJ in the small intestine of laying hens. In the domestic hen, sexual maturity (SM) occurs with the onset of the reproductive function and the first oviposition (García-Fernández et al., 2015). This transition is characterized by a huge increase in intestinal Ca absorption capacity (up to 6-fold), and a rise of 1.25(OH)2D3 plasma levels (Castillo et al., 1979; Nys et al., 1986b, 1992). Duodenal CALB1 mRNA and protein levels increase at SM (Nys et al., 1986b, 1992). We hypothesize that genes with an increased expression profile between immature and mature hens would be strong candidates to mediate Ca absorption.
The objective of the present study was therefore to identify candidate genes of the transcellular and paracellular pathways and their patterns of expression in immature pullets (12, 15, and 17 wk of age) and mature laying hens (23 wk of age), in the 3 segments of the small intestine duodenum, jejunum and ileum.
MATERIALS AND METHODS
Animals
The experiment was conducted under the guidelines of the French Ministry of Agriculture for Animal Research at the Experimental Poultry Unit of Tours, INRA, Nouzilly, France (PEAT INRA 1295). The experimental design was approved by the Regional Ethics Committee on animal experimentation (Tours, France) and the French Ministry of Higher Education and Research (Paris, France; authorization: 10043). Animals had free access to water and feed, with diets adapted to their physiological status (Table 1). Thirty 10-wk-old pullets (Isa Brown strain, Hendrix Genetics Layers, Ploufragan, France) were raised collectively in 3 pens (10 hens per per) until their slaughter at 12, 15, and 17 wk old, following the breeder guidelines for diet and environmental conditions (Hendrix Genetics, Ploufragan, France).
Table 1.
Diet composition for pullets (from 12 to 17 wk old) and laying hens (from 20 to 23 wk old).
| Diet | ||
|---|---|---|
| Item | Pullet | Laying hen |
| Ingredient, % | ||
| Corn | 47.0 | 35.5 |
| Wheat | 25 | 25 |
| Soybean meal | 18.8 | 22.7 |
| Wheat bran | 6 | 5 |
| Soybean oil | 0.3 | 1.43 |
| Corn gluten meal | 1.00 | |
| Limestone CaCo3 | 1.2 | 5.07 |
| Ground CaCO3 | 2.53 | |
| Dicalcium phosphate | 0.8 | 0.7 |
| NaCl | 0.24 | 0.29 |
| Sodium bicarbonate | 0.18 | 0.07 |
| Premix | 0.5* | 0.5† |
| DL-methionine | 0.07 | 0.15 |
| L-Thr | 0.01 | |
| Calculated energy and nutrient composition, as-fed | ||
| ME, kcal/kg | 2,886 | 2,711 |
| CP, % | 16.0 | 17.3 |
| Ca, % | 1.0 (0.99) | 3.5 (3.9) |
| Total P, % | 0.53 (0.54) | 0.50 (0.48) |
| Available P, % | 0.4 | 0.3 |
| Fat, % | 3.1 | 3.8 |
| Cellulose, % | 3.0 | 2.9 |
| Ash, % | 5.2 | 11.5 |
*Pullet's premix contains the following per kilogram: vitamin A = 10,000 IU; vitamin D3 = 2,500 IU; vitamin E = 30 IU; vitamin K = 2 mg; vitamin B1 = 2 mg; vitamin B2 = 6 mg; nicotinic acid = 40 mg; pantothenic acid = 10 mg; vitamin B6 = 3 mg; folic acid = 0.6 mg; vitamin B12 = 0.013 mg; biotin = 0.1 mg; Fe = 50 mg; Zn = 75 mg; Mn = 80 mg; Cu = 15 mg; I = 1.5 mg; and Se = 0.3 mg.
Laying hen's premix contains the following per kilogram: vitamin A = 8,000 IU; vitamin D3 = 2,000 IU; vitamin E = 20 IU; vitamin K = 1 mg; vitamin B1 = 2 mg; vitamin B2 = 4 mg; nicotinic acid = 8 mg; pantothenic acid = 20 mg; vitamin B6 = 3 mg; vitamin B12 = 0.005 mg; Fe = 18 mg; Zn = 73 mg; Mn = 90 mg; Cu = 5 mg; I = 1 mg; and Se = 0.15 mg.
(): analyzed nutritional values.
Ten mature 20-wk-old laying hens from the same strain were placed in individual cages until 23 wk old, which represents the beginning of the peak of lay. They were subjected to 14L:10D light program. The hens were euthanized 9 to 10 h after ovulation, as measured by regular monitoring of oviposition (every 10 min), and the precise stage in the ovulatory cycle was confirmed based on the weight of the dried eggshell, as previously described (Nys et al., 2010).
Tissue Sampling
Six pullets per age at 12, 15, 17 wk old, and 7 laying hens at 23 wk old at the beginning of eggshell formation (9 to 10 h post-ovulation) were submitted to blood collection in the occipital sinus using heparin–lithium coated tubes. They were then euthanized by an intravenous injection of Dolethal (182 mg/ml, pentobarbital sodium, Vetoquinol, France). Blood was centrifuged immediately at 1000 g x 15 min for plasma collection and storage at −20°C for further analysis. The oviduct was removed and weighed for immature pullets. For each hen the duodenum, jejunum, and ileum were immediately removed and thoroughly washed with a cold PBS solution. A 4-cm sample was isolated at the rostral part of the respective segments: the duodenum, the jejunum, the ileum, scraped and snap-frozen into liquid nitrogen, prior to storage at −80°C until RNA extraction.
Blood Sample Analysis
Total plasma Ca and P concentrations were measured using inductively coupled plasma optical emission spectrometry (ICP OES ThermoscientificTM iCAPTM 7200; method 990.08; AOAC International, 2006). Determination of 25(OH)D3 and 1.25(OH)2D3 concentrations were performed by chemiluminescent immunoassay (Diasorin, Saluggia, Italy, Spanaus and von Eckardstein, 2017) at the Liaison XL platform Service des Explorations Fonctionnelles (G.H. Necker Enfants Malades, 75743, Paris cedex 15, France).
Selection of Candidate Genes and Primer Design
Candidate genes were selected according to their identification in previous studies on mammals or chickens (Fujita et al., 2008; Yang et al., 2011; Jonchère et al., 2012; Gunzel and Yu, 2013; Hwang et al., 2013; Rousseau et al., 2016). For genes identified from mammals, the retrieval of the orthologous sequence was performed in Gallus_gallus-5.0 version of the chicken genome assembly using Ensembl 91 (December 2017).
Analysis of published RNAseq data on chicken gastro-intestinal tracts (Juanchich et al., 2018) allowed us to check the presence of genes previously selected from the literature and provided additional candidates. Primers were then designed with the aid of Primer-BLAST (Ye et al., 2012). The validation of primers was performed by RT-qPCR, using a LightCycler 480 Instrument II (Roche Applied Science, Meylan, France), considering primer efficiency between 80 and 120%, and sequencing of the PCR product (Genewiz, United Kingdom). We obtained a final list of 20 candidate genes and 7 housekeeping genes (HKG) with their validated primers (Tables 2, 3 and 4). All genes were named and abbreviated according to the CNGC Chicken Nomenclature genome Consortium (http://birdgenenames.org/cgnc).
Table 2.
PCR primers for candidate genes of the transcellular pathway.
| Gene symbol | Gene name | Function/location* | Forward/reverse primer | Amplicon size (bp) | Accession # |
|---|---|---|---|---|---|
| TRPV2 | Transient receptor potential cation channel subfamily V member 2 | cation channel/APM | ACTTCCCCTCTCTTTGGCTG/AGTCTTCACACCTGCCTTCA | 211 | XM_004946685.3 |
| TRPC1 | Transient receptor potential cation channel subfamily C member 1 | cation channel/APM | CATCGAGTGGCAAAGTGAAA/AGTTCGAAAGCCAAGGAGGT | 233 | NM_001004409.2 |
| TRPM7 | Transient receptor potential cation channel subfamily M member 7 | cation channel/APM | GTGTTCCCAGGAAGGCAATA/GCTTGAAGAAATGGGGTCAA | 196 | NM_001177555.1 |
| CALM1 | Calmodulin | control ion channel/C | GGTCGCTGGGTCAAAATCC/ACTCGGAATGCCTCACGGA | 163 | NM_001110364.1 |
| CALB1† | Calbindin 28K | intracellular Ca2+ binding protein/C | CAGGGTGTCAAAATGTGTGC/GCCAGTTCTGCTCGGTAAAG | 215 | NM_205513.1 |
| ATP2B1† | ATPase plasma membrane Ca2+ transporting 1 | Ca2+ /H+ exchange pump/BPM | CTGCACTGAAGAAAGCAGATGTTG/GCTGTCATATACGTTTCGTCCCC | 146 | NM_001168002.3 |
| ATP2B2† | ATPase plasma membrane Ca2+ transporting 2 | Ca2+ /H+ exchange pump/BPM | TTACTGTACTTGTGGTTGCTGTCCC/GGTTGTTAGCGTCCCTGTTTTG | 176 | XM_025154762.1 |
| ATP2B4† | ATPase plasma membrane Ca2+ transporting 4 | Ca2+/H+ exchange pump/BPM | TGCTCTGAAGAAAGCTGATGTTGG/GCTGGTGAAGTTGTCATCCGTC | 103 | XM_015298964.2 |
APM = apical plasma membrane, C = cytosol, BPM = basal plasma membrane.
Primer sequence designed by Jonchère et al. (2012).
Table 3.
PCR primers for candidate genes of the paracellular pathway.
| Gene symbol | Gene name | Function/location* | Forward/reverse primer | Amplicon size (bp) | Accession # |
|---|---|---|---|---|---|
| CLDN2 | Claudin 2 | increase permeability to Ca2+/AIS | CGCTCGTATCTCTTGCTTGG/AGAGTATGGCTGTGACGAGG | 185 | NM_001277622.1 |
| CLDN12 | Claudin 12 | increase permeability to Ca2+/AIS | ACGAGAGGAATGTGACCGTT/TTGGCACGCTTGATACGAAG | 225 | XM_025148431.1 |
| CLDN1 | Claudin 1 | decrease permeability to cations/AIS | AGATCCAGTGCAAGGTGTACG/CTGACAGACCTGCAATGATGAAG | 216 | NM_001013611.2 |
| CLDN10 | Claudin 10 | increase permeability to cations/AIS | TCCAACTGCAAGGACTTCCC/GCACAGCCCACACAGTATGA | 210 | NM_001277767.1 |
| TJP1 | Tight junction protein 1 | tight junction protein connecting transmembrane proteins/C | ACCGAGAGATGCTGGTACTG/GCACAGCCTCATTCTCATGG | 208 | XM_015278981.2 |
| TJP2 | Tight junction protein 2 | tight junction protein connecting transmembrane proteins/C | CATTGTTCGGGAGGATGCTG/AGCCAGCCAGTTTCCTAGTT | 247 | NM_204918.1 |
| TJP3 | Tight junction protein 3 | tight junction protein connecting transmembrane proteins/C | GGATACAGTGCGGCAGATTG/TGGTAGCAGTGAAGAGGTGG | 245 | XM_015299758.2 |
| OCLN | Occludin | barrier protein of tight junction/AIS and C | CCGTAACCCCGAGTTGGAT/ATTGAGGCGGTCGTTGATG | 214 | NM_205128.1 |
| JAM2 | Junctional adhesion molecule 2 | barrier protein of tight junction/AIS and C | AGACAGGAACAGGCAGTGCT/TCCAATCCCATTTGAGGCTA | 134 | NM_001006257.1 |
AIS = apical intercellular space, C = cytosol.
Table 4.
PCR primers for vitamin D receptor, phosphorus transporters, and housekeeping genes.
| Gene symbol | Gene name | Function/location* | Forward/reverse primer | Amplicon size (bp) | Accession # |
|---|---|---|---|---|---|
| VDR | Vitamin D3 receptor | Vitamin D3 receptor/N | CGATGTTCACCTGTCCGTTC/CGATGACTTTCTGCTGCTCC | 231 | NM_205098.1 |
| SLC20A1† | Solute carrier family 20 member 1 | Sodium-phosphate symporter/APM | AGGGCAGAAAGGCGTCAA/CGAGGAAGAAGAGAACAGCAGA | 104 | XM_015297502.2 |
| SLC34A2† | Solute carrier family 34 member 2 | Sodium-phosphate symporter/APM | GTCCGTTCACTCTGTTGCCT/TGGGTCCTCTTCTTGCCTTG | 242 | NM_204474.2 |
| EIF3I | eukaryotic translation initiation factor 3 subunit I | Housekeeping gene/C | GACATGTGCTCACTGGCTCT/CACTGCTGAGCTGGTCTTCA | 95 | NM_001164395.1 |
| EIF3F | eukaryotic translation initiation factor 3 subunit F | Housekeeping gene/C | CTAACTGCTTCTCCGTCCCG/ATGTCGTGCCCTGTTGCATA | 142 | XM_421624.4 |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase | Housekeeping gene/C and N | TCTCTGTTGTTGACCTGACCTG/ATGGCTGTCACCATTGAAGTC | 155 | NM_204305.1 |
| PPIA | peptidylprolyl isomerase A | Housekeeping gene/C | CGCTGACAAGGTGCCCATAA/GTCACCACCCTGACACATGA | 124 | NM_001166326.1 |
| SDHA | succinate dehydrogenase complex flavoprotein subunit A | Housekeeping gene/C | AGATACGGGAAGGAAGGGGT/ACCGTAGGCAAAACGGGAAT | 169 | NM_001277398.1 |
| TBP | TATA-box binding protein | Housekeeping gene/N | GCGTTTTGCTGCTGTTATTATGAG/TCCTTGCTGCCAGTCTGGAC | 122 | NM_205103.1 |
| MATR3 | matrin 3 | Housekeeping gene/N | ATTCACAAGGTCATGGGCGT/CCTTCCAAGAGATGCTGGCA | 92 | NM_204147.1 |
APM = apical plasma membrane, C = cytosol, N = nucleus.
Primer sequence designed by Rousseau et al. (2016).
RNA Extraction and Reverse Transcription
Total RNA was extracted using the method of Chomczynski and Sacchi, (1987) according to the manufacturer's recommendations (RNANow, Ozyme, Saint-Quentin en Yvelines, France). Concentration and quality of the extracted RNA were assessed by spectrophotometry from 230 to 280 nm, using a Nanodrop 1000 spectrophotometer (Nanodrop Technology, Wilmington, USA). The ratios 260/280 and 260/230 were between 1.8 and 2.2. The integrity of RNA was assessed by the migration of total RNA on a 1.5% agarose gel. Total RNA samples (5 μg) were subjected to DNase (DNAfree, Invitrogen) and then reverse transcripted using RNase H-MMLV reverse transcriptase (Superscript II, Invitrogen, Cergy Pontoise, France) and random hexamers (Amersham, Orsay, France).
Quantitative RT-PCR
High throughput real-time quantitative PCR was performed using the Biomark microfluidic system from Fluidigm, in which every sample-gene combination is quantified using 96.96 Dynamic Array IFCs (BMK-M-96.96, Fluidigm). Pre-amplification of the samples, chip loading and qRT-PCR were performed according to the manufacturer's protocol. Results were analyzed using the real-time PCR analysis software v.4.1.3 (Fluidigm).
Calculation of Relative Gene Expression
The relative mRNA quantification was performed with the ΔΔCt calculation method proposed by Pfaffl (2001). The relative expression ratio (R) of a candidate gene was calculated, based on the Efficiency (E) and the cycle threshold (Ct) deviation of a cDNA sample (the duodenum, jejunum, and ileum of individuals) vs. a control (mix of all cDNA samples at similar concentration), and expressed in comparison to the geometric average of a set of HKG according to the following equation of Pfaffl (2001).
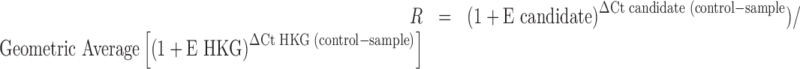 |
The 7 most stable HKG (out of 11) were chosen using GeNorm (Vandesompele et al., 2002).To facilitate the reading of the data tables and figures, the relative gene expressions were multiplied by 100.
Statistical Analyses
All statistical analyses were performed using R 3.4.0 software (R Core Team, 2017, Vienna, Austria). For plasma levels of total Ca, P, and vitamin D metabolites, the assumptions of the linear model were validated, and the data analyzed by one-way ANOVA, including the hen age as the main effect. Significant differences between means (P < 0.05) were further separated using a Tukey's test.
For mRNA expression, the normality of the data was checked by a quantile-quantile plot. Outliers, identified on the linear relationship between theoretical and sample percentiles, were removed from the dataset, resulting in a slight reduction in the number of samples for some genes in their experimental group. Changes in mRNA expression of candidate genes were analyzed with a linear mixed model (lme function) including the interaction between age and intestinal segment. The hen, considered as the experimental unit, was included as a random effect in the model. The interaction was removed from the model when it did not significantly affect mRNA expression. When significant differences were observed (P < 0.05), all pairwise comparisons were performed using the least-square means (LSMeans) method (function emmeans) and a Tukey adjustment.
RESULTS
Plasma Levels of Total Ca, P, and Vitamin D Metabolites of Immature and Mature Hens
The effect of age on plasma levels of total Ca, P, and vitamin D metabolites are shown in Table 5. All recorded plasma parameters varied between 12 and 17 wk old or between 17 and 23 wk old, after SM. plasma levels of total Ca remained stable until 17 wk old, then increased markedly from 88 to 158 mg/l (P < 0.01) between 17 and 23 wk old. This corresponds to the transition between immature and mature stages and to the increase of dietary Ca levels. Plasma levels of total P and 25(OH)D3 showed similar profiles, decreasing between 12 and 17 wk old, then remaining stable between 17 and 23 wk old (P < 0.001 and P < 0.01). Plasma levels of 1.25(OH)2D3 rose gradually between 12 (40 pg/ml) and 17 wk old (140 pg/ml) and sharply between 17 and 23 wk old (400 pg/ml; P < 0.001). Sexual development of pullets was attested by a 10-fold increase of the oviduct's weight between 15 and 17 wk old (data not shown).
Table 5.
Plasma levels of total calcium (Ca), phosphorus (P), and vitamin D metabolites in hens from 12 to 23 wk old.
| Age of hens (wk) | |||||
|---|---|---|---|---|---|
| Plasma levels | 12 | 15 | 17 | 23 | P-value |
| Total Ca (mg/dL) | 703 ± 34a | 696 ± 34a | 884 ± 106a | 1575 ± 70b | 0.007 |
| Total P (mg/dL) | 420 ± 16b | 343 ± 29a,b | 302 ± 22a | 296 ± 34a | <.0001 |
| 25(OH)D3 (ng/mL) | 70.5 ± 5.9b | 52.2 ± 4.8a,b | 40.0 ± 4.9a | 42.6 ± 4.8a | 0.002 |
| 1.25(OH)2D3 (pg/mL) | 39.3 ± 2.0a | 79.5 ± 12.7a,b | 140.0 ± 27.2b | 398.9 ± 19.3c | <.0001 |
Values are presented as LSMeans ± SEM (n = 6 to 7 per experimental group).
Means with different superscripts within the same column are significantly different (P < 0.05).
Candidate Genes of the Transcellular Pathway
The interaction between age and intestinal segment on mRNA expression of genes of the transcellular pathway is presented in Table 6. The mRNA expression of 6 candidate genes out of 8 was affected by the interaction between age and segment: transient receptor potential cation channel subfamily V member 2 (TRPV2) (P < 0.01), transient receptor potential cation channels subfamily M member 7 (TRPM7) (P < 0.05), calmodulin (CALM1) (P < 0.01), CALB1 (P < 0.001), ATP2B2 (P < 0.001), and ATP2B4 (P < 0.001). The mRNA expression of TRPV2 was higher in the jejunum at 12 and in the ileum at 17 wk old than in the duodenum at 12 wk old. It was then similar across segments from 15 to 23 wk old. Gene expression of TRPM7 increased in the jejunum between 12 and 23 wk old, and was similar between segments at 23 wk old, after SM. The mRNA expression of CALM1 was similar across segments at each age, but was higher in the duodenum at 23 than in the jejunum at 12 wk old. The mRNA expressions of CALB1 and ATP2B2 were similar across ages and segments until 17 wk old, but rose sharply between 17 and 23 wk old in the duodenum and jejunum for CALB1, and in the duodenum for ATP2B2. At 23 wk old, the expression of CALB1 was similar in the duodenum and jejunum and higher than in the ileum, and the expression of ATP2B2 was higher in the duodenum than in the other segments. The mRNA expression of ATP2B4, which was highest in the jejunum at 12 wk old, decreased in this segment between 12 and 15 wk old, while it increased in the ileum. The mRNA expression of ATP2B4 at 23 wk old was higher in the ileum than in the other segments.
Table 6.
Effect of interaction between age and intestinal segment on the relative mRNA expression of candidate genes of the transcellular pathway.
| Gene | TRPV2 | TRPM7 | CALM1 | CALB1 | ATP2B2 | ATP2B4 | |
|---|---|---|---|---|---|---|---|
| Age (wk) | Segment | ||||||
| 12 | Duodenum | 42b | 61a–c | 71a,b | 16b | 46b | 19e |
| (16) | (8) | (6) | (11) | (38) | (6) | ||
| Jejunum | 118a | 38c | 65b | 1b | 64b | 104a | |
| (15) | (8) | (6) | (11) | (38) | (6) | ||
| Ileum | 54a,b | 56a–c | 92a,b | 1b | 39b | 51c–e | |
| (15) | (8) | (6) | (11) | (38) | (6) | ||
| 15 | Duodenum | 92a,b | 101a | 81a,b | 15b | 94b | 29c–e |
| (16) | (9) | (6) | (11) | (41) | (6) | ||
| Jejunum | 53a,b | 68a–c | 78a,b | 3b | 35b | 58c,d | |
| (15) | (8) | (6) | (11) | (38) | (6) | ||
| Ileum | 95a,b | 85a,b | 74a,b | 1b | 87b | 98a,b | |
| (15) | (8) | (6) | (11) | (38) | (6) | ||
| 17 | Duodenum | 65a,b | 89a,b | 82a,b | 43b | 123b | 25d,e |
| (16) | (8) | (6) | (11) | (38) | (6) | ||
| Jejunum | 52a,b | 60a–c | 83a,b | 13b | 53b | 46c–f | |
| (15) | (8) | (6) | (11) | (38) | (6) | ||
| Ileum | 110a | 53b,c | 63b | 5b | 83b | 71b,c | |
| (15) | (8) | (6) | (11) | (38) | (6) | ||
| 23 | Duodenum | 74a,b | 88a,b | 96a | 143a | 381a | 31c–e |
| (14) | (8) | (5) | (10) | (35) | (5) | ||
| Jejunum | 70a,b | 86a,b | 90a,b | 109a | 53b | 54c–e | |
| (14) | (8) | (5) | (11) | (35) | (5) | ||
| Ileum | 61a,b | 57a–c | 85a,b | 47b | 75b | 91a,b | |
| (14) | (8) | (5) | (10) | (35) | (5) | ||
| Source of variation | |||||||
| Age | 0.860 | 0.002 | 0.011 | <.0001 | 0.005 | 0.020 | |
| Segment | 0.599 | 0.000 | 0.548 | <.0001 | <.0001 | <.0001 | |
| Age x Segment | 0.001 | 0.038 | 0.006 | 0.000 | 0.000 | <.0001 | |
Relative expression values, expressed as arbitrary units are presented as LSMeans with (SEM) with n = 5 to 7 per experimental group.
Means with different superscripts within the same column are significantly different (P < 0.05).
TRPV2 = Transient receptor potential cation channel subfamily V member 2; TRPM7 = Transient receptor potential cation channel subfamily M member 7; CALM1 = Calmodulin; CALB1 = Calbindin 28K; ATP2B2 = ATPase plasma membrane Ca2+ transporting 2; and ATP2B4 = ATPase plasma membrane Ca2+ transporting 4.
For the remaining three candidates of the transcellular pathway, no interaction between age and segment was observed. The main effects of age and intestinal segment are presented in Figure 1. The mRNA expression of transient receptor potential cation channels subfamily C member 1 (TRPC1) increased between 12 and 15 wk old (P < 0.05), and then remained elevated (Figure 1A). The mRNA expression of ATP2B1 rose between 17 and 23 wk old (P < 0.05), and its expression was higher in the duodenum compared to the other segments (P < 0.05; Figure 1B and 1C).
Figure 1.
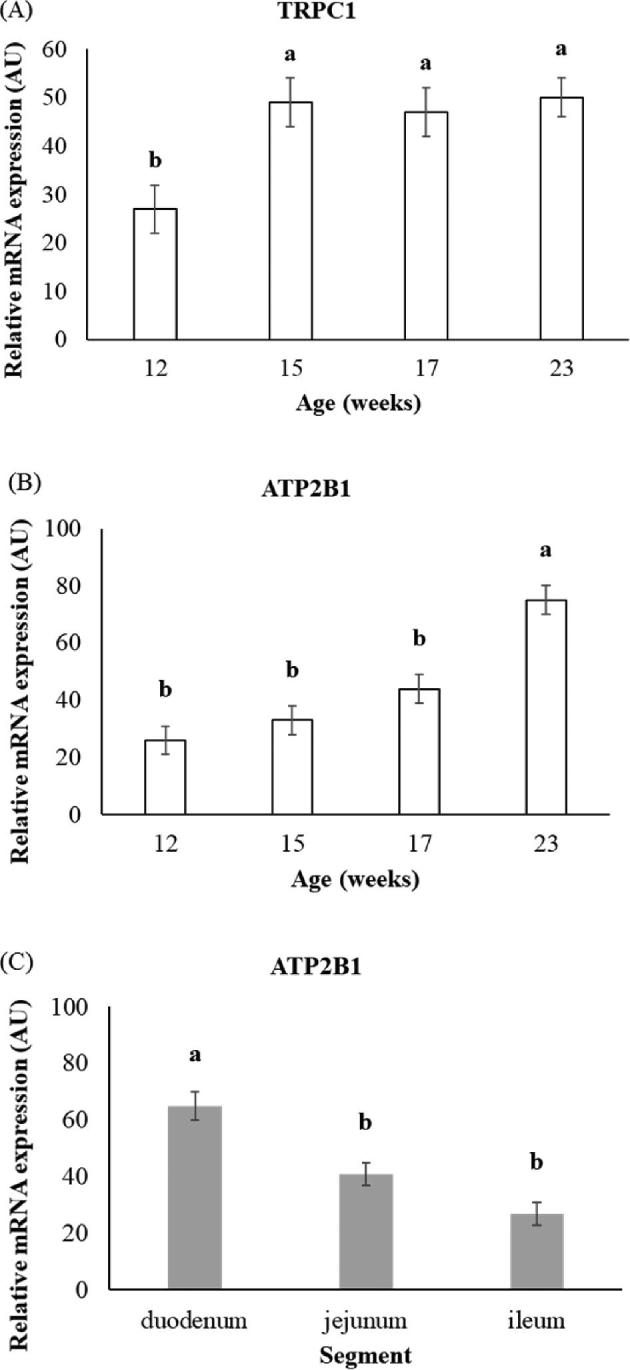
Effect of age (A-B) or segment (C) on the relative mRNA expression of candidate genes of the transcellular pathway. (A) Transient receptor potential cation channel subfamily C member 1 (TRPC1), (B, C) ATPase plasma membrane Ca2+ transporting 1 (ATP2B1). Values expressed as arbitrary units (AU) are LSMeans ± SEM (n = 17 to 21 per age and n = 23 to 25 per segment). a-bMeans with different superscripts within the graph are significantly different (P < 0.05).
Candidate Genes of the Paracellular Pathway
The interaction between age and intestinal segment on mRNA expression of genes of the paracellular pathway is presented in Table 7. The mRNA expression of 4 candidate genes out of 9 was affected by the interaction between age and segment: CLDN2 (P < 0.01), CLDN10 (P < 0.001), OCLN (P < 0.05) and JAM2 (P < 0.001). The mRNA expression of CLDN2 increased in the duodenum between 12 and 23 wk old, while it was low in the jejunum until 17 wk old, and increased between 17 and 23 wk old. As a result, the expression of CLDN2 was higher in the jejunum than in the ileum at 23 wk old. Gene expression of CLDN10, OCLN, and JAM2 decreased with age in 1 or 2 segments. The mRNA expression of CLDN10 decreased in the duodenum between 12 and 17 wk old. In the jejunum, it decreased between 17 and 23 wk old and in the ileum between 12 and 23 wk old. As a result, CLDN10 was expressed at similar levels in the 3 segments after SM. The mRNA expression of OCLN was similar across the 3 segments, except at 15 wk old, when it was higher in the ileum than in the other segments. Gene expression of JAM2 was higher in the ileum than in the other 2 segments at 15 and 17 wk old, and subsequently decreased in the ileum. As a result similar mRNA expression between segments was observed after SM.
Table 7.
Effect of interaction between age and intestinal segment on the relative mRNA expression of candidate genes of the paracellular pathway.
| Gene | CLDN2 | CLDN10 | OCLN | JAM2 | |
|---|---|---|---|---|---|
| Age (wk) | Segment | ||||
| 12 | Duodenum | 17c–h | 339a | 72b,c | 83b,c |
| (11) | (21) | (9) | (243) | ||
| Jejunum | 5g,h | 181b,c | 99b,c | 884a–c | |
| (11) | (21) | (9) | (142) | ||
| Ileum | 9e–h | 250a,b | 105a–c | 435a–c | |
| (11) | (21) | (9) | (155) | ||
| 15 | Duodenum | 53a–g | 235a,b | 67c | 421b,c |
| (11) | (23) | (10) | (155) | ||
| Jejunum | 38b–h | 305a | 87b,c | 240c | |
| (11) | (21) | (9) | (142) | ||
| Ileum | 14d–h | 179b,c | 149a | 1174a | |
| (11) | (21) | (9) | (142) | ||
| 17 | Duodenum | 68a–d | 172b,c | 82b,c | 355c |
| (11) | (21) | (9) | (142) | ||
| Jejunum | 42b–h | 237a,b | 76b,c | 334c | |
| (11) | (21) | (9) | (142) | ||
| Ileum | 28e–h | 147b–d | 119a,b | 1078a,b | |
| (11) | (21) | (9) | (142) | ||
| 23 | Duodenum | 87a,b | 50d | 87b,c | 228c |
| (10) | (20) | (9) | (131) | ||
| Jejunum | 99a | 108c,d | 85b,c | 160c | |
| (10) | (20) | (9) | (141) | ||
| Ileum | 59b–f | 113c,d | 110a–c | 357c | |
| (10) | (20) | (9) | (131) | ||
| Source of variation | |||||
| Age | 0.000 | <.0001 | 0.462 | 0.024 | |
| Segment | <.0001 | 0.047 | <.0001 | 0.000 | |
| Age x Segment | 0.005 | <.0001 | 0.015 | 0.000 | |
Relative expression values, expressed as arbitrary units, are presented as LSMeans with (SEM) with n = 4 to 7 per experimental group.
Means with different superscripts within the same column are significantly different (P < 0.05).
CLDN2 = Claudin 2; CLDN10 = Claudin 10; OCLN = Occludin; and JAM2 = Junctional adhesion molecule 2.
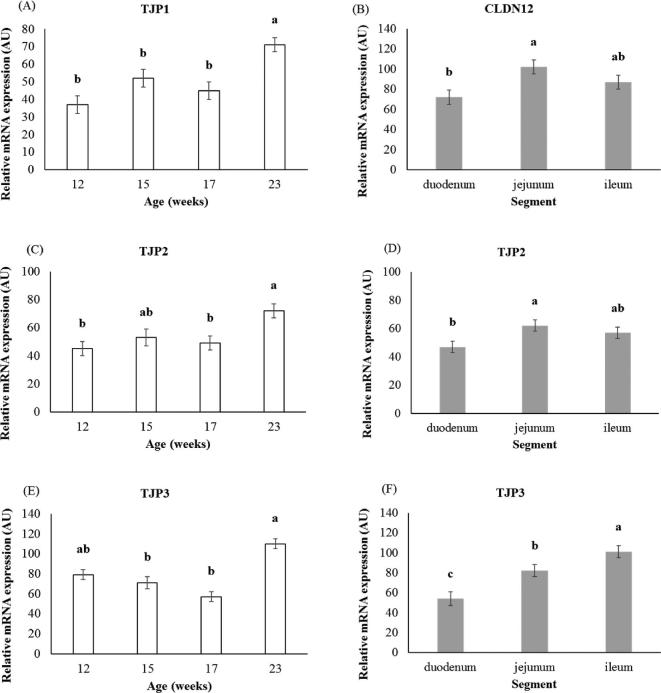
For the remaining 5 candidates of the paracellular pathway, no interaction between age and segment was observed. The effects of age and intestinal segment are shown in Figure 2. While CLDN1 expression neither varied with age, nor with segment (data not shown), CLDN12 expression varied with the segment (P < 0.05), tight junction proteins 1 (TJP1) expression with age, tight junction proteins 2 (TJP2), and tight junction proteins 3 (TJP3) expression with both age and segment (P < 0.05). The mRNA expression of CLDN12 was higher in the jejunum than in the duodenum, but intermediate in the ileum (Figure 2B). Gene expression of TJP1, TJP2, and TJP3 increased between 17 and 23 wk old (Figure 2A, 2C and 2E). The mRNA expression of TJP2 was higher in the jejunum than in the duodenum, but intermediate in the ileum (Figure 2D). Gene expression of TJP3 increased from the duodenum to the ileum (Figure 2F).
Figure 2.
Effect of age (A-C-E) or segment (B-D-F) on the relative mRNA expression of candidate genes of the paracellular pathway (A, C) Tight junction protein 1 (TJP1), (B) Claudin 12 (CLDN12), (C; D) Tight junction protein 2 (TJP2), (E, F) Tight junction protein 3 (TJP3). Values expressed as arbitrary units (AU) are LSMeans ± SEM (n = 17 to 21 per age and n = 24 to 25 per segment). a-cMeans with different superscripts within the graph are significantly different (P < 0.05).
Vitamin D Receptor and P Transporters
The interactions between age and intestinal segment on mRNA expression of vitamin D receptor (VDR) and P transporters are shown in Table 8. The mRNA expression of VDR increased in the jejunum between 12 and 23 wk old (P < 0.01), but was not affected by age in the other segments. In addition, it was similar for segments at each of the four ages. The mRNA expression of solute carrier family 20 member 1 (SLC20A1) was higher in the jejunum than in the duodenum between 12 and 17 wk old, and higher in the ileum than in the duodenum at 23 wk old, following a significant decrease in the jejunum between 15 and 23 wk old (P < 0.001). Gene expression of solute carrier family 34 member 2 (SLC34A2) was higher in the duodenum than in the jejunum and ileum, until 17 wk old. It subsequently increased in the jejunum between 17 and 23 wk old. Consequently its expression was similar in the duodenum and jejunum at 23 wk old, and was higher than in the ileum (P < 0.001).
Table 8.
Effect of interaction between age and intestinal segment on the relative mRNA expression of candidate genes for vitamin D receptor and transcellular phosphorus apical entry.
| Gene | VDR | SLC20A1 | SLC34A2 | |
|---|---|---|---|---|
| Age (wk) | Segment | |||
| 12 | Duodenum | 78a–d | 72d–f | 357a |
| (13) | (24) | (30) | ||
| Jejunum | 23d | 189a–c | 1c | |
| (13) | (24) | (30) | ||
| Ileum | 56b–d | 268a,b | 59c | |
| (13) | (26) | (30) | ||
| 15 | Duodenum | 96b–d | 70c–f | 257a,b |
| (14) | (26) | (33) | ||
| Jejunum | 102a,b | 288a | 93c | |
| (13) | (24) | (30) | ||
| Ileum | 50b–d | 162b–d | 2c | |
| (13) | (24) | (30) | ||
| 17 | Duodenum | 69a–d | 34e,f | 329a |
| (13) | (24) | (30) | ||
| Jejunum | 74a–d | 182a–d | 108b,c | |
| (13) | (24) | (30) | ||
| Ileum | 31c,d | 115c–f | 3c | |
| (13) | (24) | (30) | ||
| 23 | Duodenum | 103a,b | 24f | 315a |
| (12) | (22) | (28) | ||
| Jejunum | 119a | 110c–f | 265a | |
| (12) | (22) | (28) | ||
| Ileum | 86a–d | 152b–e | 54c | |
| (12) | (22) | (28) | ||
| Source of variation | P-value | |||
| Age | 0.001 | 0.003 | 0.003 | |
| Segment | 0.001 | <.0001 | <.0001 | |
| Age x Segment | 0.007 | 0.000 | 0.000 | |
Relative expression values, expressed as arbitrary units, are presented as LSMeans with (SEM) with n = 5 to 7 per experimental group.
Means with different superscripts within the same column are significantly different (P < 0.05).
VDR = Vitamin D receptor; SLC20A1 = Solute carrier family 20 member 1; SLC34A2 = Solute carrier family 34 member 2.
DISCUSSION
In the present study, RT-qPCR was used to quantify mRNA expression of candidate genes of the transcellular and the paracellular Ca absorption pathways in the small intestine of laying hens. An integrative description of genes involved in Ca and P absorption in mature laying hens is provided in the Figure 3. Concerning the transcellular pathway, we identified three additional candidate genes for Ca2+ entry. We also confirmed that CALB1 increased in the duodenum of mature laying hens, and observed that it also increased in the jejunum. Two candidate genes for Ca extrusion ATP2B1 and ATP2B2 rose respectively, in all 3 segments or in the duodenum of laying hens. This study also detected the expression of candidate genes of the paracellular pathway for the first time. Gene expression of CLDN2, CLDN12, TJP1, TJP2, and TJP3 increased with age or only after SM, or in 1 or 2 segments of the small intestine. The up regulation of CLDN2 and CLDN12, which increase Ca2+ permeability in intestinal epithelial cells, supports the hypothesis that the paracellular pathway may be involved in Ca absorption in laying hens.
Figure 3.
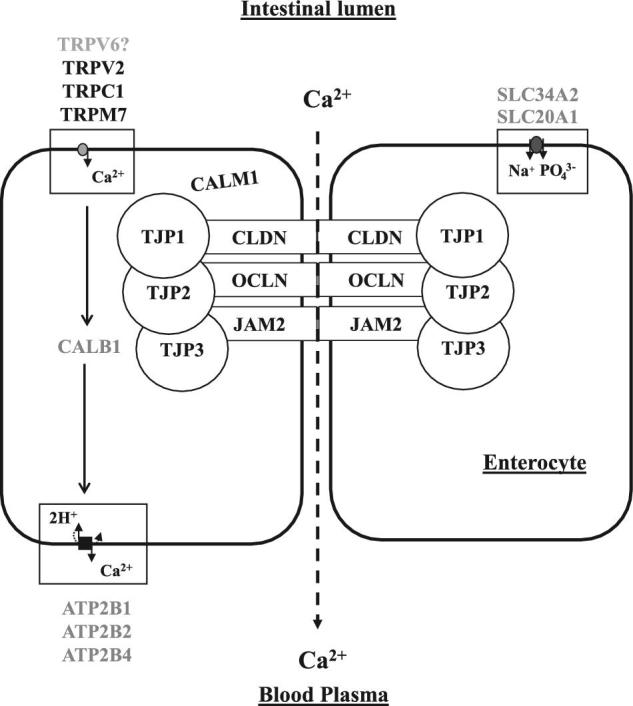
Hypothetical model of ionic calcium (Ca) and phosphate (P) transfer across the intestinal epithelial barrier of the laying hen. Rectangles represent the transmembrane proteins, junctional adhesion molecule (JAM), claudins (CLDN: CLDN1, 2, 10, or 12), or occludin (OCLN), while the circles represent the tight junction proteins (TJP: TJP1, 2, or 3). Black arrows indicate the transcellular pathway and dotted arrows the paracellular pathway. Genes in grey represent those previously identified in the small intestine of laying hens, while genes in black were those identified in the present study. Ca2+ refers to ionic calcium, PO43− to the phosphate ion, H+ to the proton ion and Na+ to the sodium ion. TRPV6 = Transient receptor potential cation channel subfamily V member 6; TRPV2 = Transient receptor potential cation channel subfamily V member 2; TRPC1 = Transient receptor potential cation channel subfamily C member 1; TRPM7 = Transient receptor potential cation channel subfamily M member 7; CALM1 = Calmodulin; CALB1 = Calbindin 28K; ATP2B1 = ATPase plasma membrane Ca2+ transporting 1; ATP2B2 = ATPase plasma membrane Ca2+ transporting 2; ATP2B4 = ATPase plasma membrane Ca2+ transporting 4; CLDN2 = Claudin 2; CLDN12 = Claudin 12; CLDN10 = Claudin 10; CLDN1 = Claudin 1; OCLN = Occludin; JAM2 = Junctional adhesion molecule 2; TJP1 = Tight junction protein 1; TJP2 = Tight junction protein 2; TJP3 = Tight junction protein 3; VDR = Vitamin D receptor; SLC20A1 = Solute carrier family 20 member 1; and SLC34A2 = Solute carrier family 34 member 2.
Sexual maturity in birds is a period of profound physiological modifications, linked to the onset of the reproductive function and marked by the first oviposition (García-Fernández et al., 2015). In layer production units, this transition period requires appropriate environmental conditions and adequate nutrition, which were respected in the present study (Dawson et al., 2001). The activity of the gonadotropic axis results in estrogen synthesis by the ovary which in turn promotes the development and growth of various target organs, like the oviduct (Pearce, 1974; Johnson, 2000). In the present study, although steroids were not assayed, the increase of oviduct weight between 12 and 17 wk old was the hallmark of their effects (Munro and Kosin, 1943; Dougherty and Sanders, 2005). Plasma parameters were modified during this transition period from immature to mature hens. The huge rise in total plasma Ca observed after SM is consistent with previous observations. It results from the feed transition with higher dietary Ca levels (from 1 to 3.5%) and increased vitellogenin synthesis, an egg yolk precursor containing binding sites for Ca (Nys et al., 2011). A decrease in total plasma P was observed at 17 wk old, when medullary bone was first detected, and coincided with the increase of 1.25(OH)2D3, previously described by Castillo et al. (1979). We also observed a decrease in 25(OH)D3 at 17 wk old, in contrast to a previous study (Nys et al., 1986a). This might reflect a limiting activity of the enzyme converting dietary cholecalciferol to 25(OH)D3 (25-hydroxylase), rather than a limiting plasma level of vitamin D binding protein, or insufficient vitamin D dietary supply high at this stage (Nys et al., 1986a). In the present study, CALB1 mRNA expression was enhanced in the duodenum after SM, as observed previously for CALB1 protein and mRNA levels, which correlated with the increased Ca absorption capacity in laying hens (Nys et al., 1986b, 1992). These observations indicate that the present experiment is particularly suited to the identification of new candidate genes of the transcellular and paracellular Ca absorption pathways in laying hens.
The present experiment provides the expression profile of 7 candidate genes of the transcellular Ca transport in addition to CALB1, in the small intestine of pullets at several ages and of laying hens, for the first time. The intestinal transcellular pathway occurs in 3 steps: apical Ca2+ entry, intracellular Ca2+ transport and basal active Ca2+ extrusion (Wasserman and Taylor, 1966; Coty and Conkey, 1982). We did not detect TRPV6 in the current study at any age, although previous studies have reported its expression in the intestine of the laying hen (Yang et al., 2011; Jonchère et al., 2012; Li et al., 2018). This observation is in agreement with the absence of a TRPV6 expression in the chicken RNAseq data and in growing broiler chickens, despite the detection of an immunoreactive TRPV6 protein in the chicken's small intestine (Proszkowiec-Weglarz and Angel, 2013; Huber et al., 2015; Juanchich et al., 2018; Proszkowiec-Weglarz et al., 2019). Therefore, as previously discussed further studies are necessary to conclude about TRPV6 expression in the hen's small intestine (Proszkowiec-Weglarz et al., 2019). Out of the 4 candidate genes for apical Ca2+ entry studied, 3 were detected. The first candidate, TRPV2 is a Ca2+ channel (Owsianik et al., 2006). It varied neither with age, nor with the segment. TRPM7 is a divalent cation channel (Chubanov et al., 2004; Owsianik et al., 2006). It was enhanced in the jejunum between 12 and 23 wk old. TRPC1 is known to facilitate Ca2+ entry (Ambudkar et al., 2007). Its expression increased in all 3 segments between 12 and 15 wk old. We hypothesize that these 2 new candidates (TRPM7 and TRPC1) could compensate for the absence of TRPV6 expression in the intestine of laying hens. CALM1 is a ubiquitous Ca2+ binding protein, acting as an inhibitor of TRP channel activities (Zheng, 2013). It did not vary consistently between segments or ages. Although none of the candidates for Ca entry was expressed differentially between 17 and 23 wk old, this does not exclude variations at protein level or activity.
The high absorption of Ca in the small intestine of laying hens implies mechanisms for maintaining a low free intracellular concentration of Ca2+ (Carafoli, 1987). It has been proposed that CALB1, discovered in chick intestinal epithelial cells, acts as a buffer through intracellular Ca2+ binding (Wasserman and Taylor, 1966). The increased expression of CALB1 previously described in the duodenum after SM, is extended to the jejunum in the present study, which further highlights the importance of this candidate in the saturable Ca transport in these two segments (Nys et al., 1992).
The output of Ca2+ from the enterocyte occurs against a concentration gradient and involves plasma membrane Ca2+ ATPases (Bar, 2009). Four isoforms have been characterized in mammals (ATP2B1, B2, B3 and B4), but only 3 are maintained in birds ATP2B1, B2, and B4 and these have already been detected in the hen's duodenum (Strehler and Zacharias, 2001; Jonchère et al., 2012). On the one hand, the results may indicate that ATP2B1, ATP2B2, and CALB1 are favored partners of the transcellular Ca absorption pathway in the duodenum of laying hens, where they are highly expressed. On the other hand, ATP2B4 could be a favored partner to mediate Ca2+ output in the ileum of the laying hens, where it is highly expressed, and could compensate for the comparatively lower expression of ATP2B1 and ATP2B2. Due to the higher pH in the ileum, which lowers Ca solubility, its absorption rate is lower than the upper segments (Hurwitz and Bar, 1968). It remains, however the high expression of ATP2B4 combined with a low expression of CALB1 could contribute to this difference. Although several studies and reviews proposed that non-saturable transport could contribute to intestinal Ca absorption in birds, candidate genes of the paracellular pathway have been given little attention so far (Hurwitz and Bar, 1969; Nys and Mongin, 1982). The TJ are the functional units of this pathway, composed of transmembrane proteins CLDN, JAM, OCLN, and cytosolic proteins TJP, forming pores for the permeation of ions and water. The expression data showed for the first time, that all candidates were expressed in the hens’ small intestine, supporting the hypothesis that this pathway could contribute to Ca transfer in the laying hens’ intestine (Hurwitz and Bar, 1969; Nys and Mongin, 1982). CLDN1 decreases cation permeability (Gunzel and Yu, 2013). It did not show any variation in this experiment. CLDN2 and CLDN12 are known to increase intestinal Ca2+ permeability (Fujita et al., 2008). In the present trial, CLDN12 expression was the highest in the jejunum across all ages studied, whereas CLDN2 only appeared after SM in this segment. In the duodenum, CLDN2 expression increased with age. Both segments support the increase of intestinal Ca absorption during shell formation, when high contents of soluble Ca are present in the intestine (Hurwitz and Bar, 1965, 1969; Sauveur and Mongin, 1983; Guinotte et al., 1995). These segments are the main Ca absorption sites, in contrast to the ileum, and their high rate of Ca absorption may largely result from a non-saturable Ca transport, because CALB1 concentration does not vary during the daily ovulatory cycle (Hurwitz and Bar, 1966, 1968; Nys et al., 1992). In contrast, CLDN10 decreased with age across all 3 segments. Two splice variants of CLDN10 were initially described in mice with opposite charge selectivity, and 4 additional ones were reported (Van Itallie and Anderson, 2006; Gunzel et al., 2009). At least 2 splice variants exist in Gallus gallus which cannot be distinguished in the present study. The reduction in CLDN10 expression with age may suggest a role that is distinct from that of CLDN2. Although the precise role of TJP proteins for Ca transport is unknown, these proteins anchor the TJ (Suzuki, 2013). The candidate TJP2 was most expressed in the jejunum and TJP3 in the ileum. All three TJPs were up-regulated after SM, suggesting their importance in laying hens. The expression of JAM2 and OCLN remained at similar levels between intestinal segments after SM. Although these two candidate genes contribute to overall intestinal permeability, further research is needed to understand their role in Ca absorption (Laukoetter et al., 2007). Based on their regulation pattern across ages and segments, CLDN2, CLDN12, TJP1, TJP2, and TJP3 are strong candidate genes, which may contribute to paracellular Ca absorption in the intestine of the sexually mature hen.
Phosphorus and Ca metabolisms are tightly linked and regulated by the same hormones (Kuro-o and Moe, 2017). An optimal absorption of P is crucial during the formation of the medullary bone before the onset of laying and later during the daily ovulatory cycle when an intense bone remodeling occurs (Castillo et al., 1979; Sauveur and Mongin, 1983; Kerschnitzki et al., 2014). In the jejunum, the expression of SLC20A1 decreased, but that of SLC34A2 increased with age. These results pointed to SLC20A1 as the main P transporter in the ileum, and SLC34A2 as the main transporter in the duodenum and jejunum after SM. However, the jejunum exhibited an intermediate level of expression of both transporters which could contribute to the relatively high absorption of P in this segment (Hurwitz and Bar, 1965).
The nuclear VDR was expressed in all 3 parts of the small intestine and at similar levels across ages. The increase in 1.25(OH)2D3 with age and after SM triggers the expression of target genes in the small intestine by activating its receptor (Dusso et al., 2005; Nys and Le Roy, 2017). Gene expression of CALB1 increased after SM, when 1.25(OH)2D3 rose sharply. This is consistent with the presence of a vitamin D responsive element on the gene sequence of CALB1 (Dusso et al., 2005; Nys and Le Roy, 2017). Several other genes, which were induced (TRPM7, TRPC1, ATP2B1, ATP2B2, ATP2B4, CLDN2, TJP1, TJP2, TJP3, and SLC34A2) or repressed (ATP2B4, CLDN10, JAM2, and SLC20A1) with age or after SM, could also be regulated by vitamin D. Some have already been shown to be responsive to vitamin D, like ATP2B1, SLC34A2, CLDN2, and CLDN12 (Cai et al., 1993; Fujita et al., 2008; Forster et al., 2013).
In birds, most dietary Ca is absorbed in the upper parts of the small intestine (the duodenum and jejunum), while the main site of absorption in mammals is the ileum (Bar, 2009). This could be attributed to the comparatively shorter length of the entire small intestine, the faster passage rates and the intrinsic Ca absorption rates of the different compartments in birds (Hurwitz and Bar, 1966, 1968; Caviedes-Vidal et al., 2007). The present study suggests that molecular mechanisms underlie these differences. Indeed CLDN2, CLDN12, CALB1, ATP2B1, and ATP2B2 were enhanced after SM in the duodenum or jejunum. The transcellular pathway cannot be sufficient to support Ca absorption in laying hens, as it is saturable. Our results highlight that 4 candidates of the paracellular pathway (CLDN2, TJP1, TJP2, and TJP3), as well as 3 candidates of the transcellular pathway (CALB1, ATP2B1, and ATP2B2) are enhanced after SM in the duodenum or the jejunum or the 3 intestinal segments. Therefore, we believe that both pathways work cooperatively to support the high rate of Ca absorption, when a high content of soluble Ca is present in the intestine during eggshell formation (Hurwitz and Bar, 1968; Sauveur and Mongin, 1983; Hwang et al., 2013). It would therefore be interesting to sustain higher intestinal levels of soluble Ca during the last third of eggshell formation. This would increase the non-saturable transport of Ca, and thereby Ca retention, a pre-requisite for reducing bone mobilization and P losses. Understanding the regulation of candidate genes of both pathways by 1.25(OH)2D3 could also be of interest in older hens, as a deficiency in the vitamin D metabolite conversion was suggested (Abe et al., 1982; Frost and Roland, 1990). This deficiency could possibly be alleviated by enhancing the expression of genes involved in both Ca absorption pathways, through a dietary supply of vitamin D3 metabolites.
To conclude, this study identified 17 candidate genes of the transcellular and paracellular Ca transport pathways in the 3 distinct parts of the laying hen's small intestine at different ages, before and after SM, and suggested the importance of the paracellular pathway in Ca absorption. The present expression data sets the basis for future studies providing the final proof of their physiological relevance, though the detection of the corresponding proteins and the measure of their activity. Both transcellular and paracellular pathways may work cooperatively in the duodenum and the jejunum, the main sites of Ca absorption in laying hens. The non-saturable transport through the paracellular Ca pathway could be a mechanism of great importance in laying hens due to the high amount of soluble Ca in the intestine. The results also provide new insights to explain the differences in Ca absorption rates between the duodenum, jejunum and ileum in laying hens, through adaptations of the molecular repertoire of Ca transporters.
ACKNOWLEDGMENTS
The authors are grateful to Nathalie Même and Maryse Leconte (UMR BOA INRA Université de Tours, France) for their technical assistance, to Philippe Didier (UE PEAT INRA, France) for the care of experimental birds and Christelle Hennequet-Antier (UMR BOA INRA Université de Tours, France) for expert advice on statistical analysis. The research was supported by a research grant from Institut Carnot “France Futur Elevage”, as part of the Audrey Gloux's PhD project.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
REFERENCES
- Abe E., Horikawa H., Masumura T., Sugahara M., Kubota M., Suda T.. 1982. Disorders of cholecaiciferol metabolism in old egg-laying hens. J. Nutr. 112:436–446. [DOI] [PubMed] [Google Scholar]
- Ambudkar I. S., Ong H. L., Liu X., Bandyopadhyay B., Cheng K. T.. 2007. TRPC1: the link between functionally distinct store-operated calcium channels. Cell Calcium. 42:213–223. [DOI] [PubMed] [Google Scholar]
- AOAC International. 2006. Method 990.08: Metals in solid wastes. In: Official Methods of Analysis of AOAC International. Arlington, VA: Association of Official Analytical Chemists: 2006. [Google Scholar]
- Bar A. 2009. Calcium transport in strongly calcifying laying birds: mechanisms and regulation. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 152:447–469. [DOI] [PubMed] [Google Scholar]
- Bar A., Moaz A., Hurwitz S.. 1979. Relationship of intestinal and plasma calcium-binding protein to intestinal calcium absorption. FEBS Lett. 102:79–81. [DOI] [PubMed] [Google Scholar]
- Cai Q., Chandler J. S., Wasserman R. H., Kumar R., Penniston J. T.. 1993. Vitamin D and adaptation to dietary calcium and phosphate deficiencies increase intestinal plasma membrane calcium pump gene expression.. Proc. Natl. Acad. Sci. 90:1345–1349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carafoli E. 1987. Intracellular calcium homeostasis. Annu. Rev. Biochem. 56:395–433. [DOI] [PubMed] [Google Scholar]
- Castillo L., Tanaka Y., Wineland M. J., Jowsey J. O., Deluca H. F.. 1979. Production of 1,25-dihydroxyvitamin D3 and formation of medullary bone in the egg-laying hen. Endocrinology. 104:1598–1601. [DOI] [PubMed] [Google Scholar]
- Caviedes-Vidal E., McWhorter T. J., Lavin S. R., Chediack J. G., Tracy C. R., Karasov W. H.. 2007. The digestive adaptation of flying vertebrates: high intestinal paracellular absorption compensates for smaller guts. Proc. Natl. Acad. Sci. 104:19132–19137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chomczynski P., Sacchi N.. 1987. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162:156–159. [DOI] [PubMed] [Google Scholar]
- Christakos S., Lieben L., Masuyama R., Carmeliet G.. 2014. Vitamin D endocrine system and the intestine. Bonekey Rep. 3:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chubanov V., Waldegger S., Schnitzler Mm., Vitzthum H., Sassen M. C., Seyberth H. W., Konrad M., Gudermann T.. 2004. Disruption of TRPM6/TRPM7 complex formation by a mutation in the TRPM6 gene causes hypomagnesemia with secondary hypocalcemia. Proc. Natl. Acad. Sci. 101:2894–2899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coty A., Conkey C.. 1982. A high-affinity calcium-stimulated ATPase activity in the hen oviduct shell gland. Arch. Biochem. Biophys. 219:444–453. [DOI] [PubMed] [Google Scholar]
- Dawson A., King V. M., Bentley G. E., Ball G. F.. 2001. Photoperiodic control of seasonality in birds. J. Biol. Rhythms. 16:365–380. [DOI] [PubMed] [Google Scholar]
- Dougherty D. C., Sanders M. M.. 2005. Estrogen action: revitalization of the chick oviduct model. Trends Endocrinol. Metab. 16:414–419. [DOI] [PubMed] [Google Scholar]
- Dusso A. S., Brown A. J., Slatopolsky E.. 2005. Vitamin D. Am. J. Physiol. Physiol. 289:F8–F28. [DOI] [PubMed] [Google Scholar]
- Forster I. C., Hernando N., Biber J., Murer H.. 2013. Phosphate transporters of the SLC20 and SLC34 families. Mol. Aspects Med. 34:386–395. [DOI] [PubMed] [Google Scholar]
- Frost T., Roland D.. 1990. Influence of vitamin D3, 1 alpha-hydroxyvitamin D3, and 1,25-dihydroxyvitamin D3 on eggshell quality, tibia strength, and various production parameters in commercial laying hens. Poult. Sci. 69:2008–2016. [DOI] [PubMed] [Google Scholar]
- Fujita H., Sugimoto K., Inatomi S., Maeda T., Osanai M., Uchiyama Y., Yamashita T.. 2008. Tight junction proteins claudin-2 and -12 are critical for vitamin D-dependent Ca2+ absorption between enterocytes. Mol. Biol. Cell. 19:1912–1921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Fernández J. M., Cernuda-Cernuda R., Davies W. I. L., Rodgers J., Turton M., Peirson S. N., Follett B. K., Halford S., Hughes S., Hankins M. W., Foster R. G.. 2015. The hypothalamic photoreceptors regulating seasonal reproduction in birds: a prime role for VA opsin. Front. Neuroendocrinol. 37:13–28. [DOI] [PubMed] [Google Scholar]
- Guinotte F., Gautron J., Nys Y., Soumarmon A.. 1995. Calcium solubilization and retention in the gastrointestinal tract in chicks (Gallus domesticus) as a function of gastric acid secretion inhibition and of calcium carbonate particle size. Br. J. Nutr. 73:125–139. [DOI] [PubMed] [Google Scholar]
- Gunzel D., Stuiver M., Kausalya P. J., Haisch L., Krug S. M., Rosenthal R., Meij I. C., Hunziker W., Fromm M., Muller D.. 2009. Claudin-10 exists in six alternatively spliced isoforms that exhibit distinct localization and function. J. Cell Sci. 122:1507–1517. [DOI] [PubMed] [Google Scholar]
- Gunzel D., Yu A. S. L.. 2013. Claudins and the modulation of tight junction permeability. Physiol. Rev. 93:525–569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huber K., Zeller E., Rodehutscord M.. 2015. Modulation of small intestinal phosphate transporter by dietary supplements of mineral phosphorus and phytase in broilers. Poult. Sci. 94:1009–1017. [DOI] [PubMed] [Google Scholar]
- Hurwitz S., Bar A.. 1965. Absorption of calcium and phosphorus along the gastrointestinal tract of the laying fowl as influenced by dietary calcium and egg shell formation. J. Nutr. 86:433–438. [DOI] [PubMed] [Google Scholar]
- Hurwitz S., Bar A.. 1966. Rate of passage of calcium-45 and Yttrium-91 along the intestine, and calcium absorption in the laying fowl. J. Nutr. 89:311–316. [DOI] [PubMed] [Google Scholar]
- Hurwitz S., Bar A.. 1968. Activity, concentration, and lumen-blood electrochemical potential difference of calcium in the intestine of the laying hen. J. Nutr. 95:647–654. [DOI] [PubMed] [Google Scholar]
- Hurwitz S., Bar A.. 1969. Intestinal calcium absorption in the laying fowl and its importance in calcium homeostasis. Am. J. Clin. Nutr. 22:391–395. [DOI] [PubMed] [Google Scholar]
- Hwang I., Yang H., Kang H. S., Ahn C., Hong E. J., An B. S., Jeung E. B.. 2013. Alteration of tight junction gene expression by calcium and vitamin D-deficient diet in the duodenum of calbindin-null mice. Int. J. Mol. Sci. 14:22997–23010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Itallie C. M., Anderson J. M.. 2006. Two splice variants of claudin-10 in the kidney create paracellular pores with different ion selectivities. Am. J. Physiol. Renal Physiol. 291:F1288–F1299. [DOI] [PubMed] [Google Scholar]
- Jalal M. A., Scheideler S. E.. 2001. Effect of supplementation of two different sources of phytase on egg production parameters in laying hens and nutrient digestiblity. Poult. Sci. 80:1463–1471. [DOI] [PubMed] [Google Scholar]
- Johnson A. 2000. Reproduction in the female. Pages 569–596 in Sturkie's Avian Physiology. Reproduction in the female. 5th ed. Scanes Colin G., ed. Academic Press, USA. [Google Scholar]
- Jonchère V., Brionne A., Gautron J., Nys Y.. 2012. Identification of uterine ion transporters for mineralisation precursors of the avian eggshell. BMC Physiol.. 12:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juanchich A., Hennequet-Antier C., Cabau C., Le Bihan-Duval E., Duclos M. J., Mignon-Grasteau S., Narcy A.. 2018. Functional genomics of the digestive tract in broilers. BMC Genomics. 19:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerschnitzki M., Zander T., Zaslansky P., Fratzl P., Shahar R., Wagermaier W.. 2014. Rapid alterations of avian medullary bone material during the daily egg-laying cycle. Bone. 69:109–117. [DOI] [PubMed] [Google Scholar]
- Keshavarz K. 1986. The effect of dietary levels of calcium and phosphorus on performance and retention of these nutrients by laying hens. Poult. Sci. 65:114–121. [Google Scholar]
- Kim W. K., Bloomfield S. A., Ricke S.. 2012. Concepts and methods for understanding bone metabolism in laying hens. Worlds Poult. Sci. J. 68:71–82. [Google Scholar]
- Kuro-o M., Moe O. W.. 2017. FGF23-αKlotho as a paradigm for a kidney-bone network. Bone. 100:4–18. [DOI] [PubMed] [Google Scholar]
- Laukoetter M. G., Nava P., Lee W. Y., Severson E. A., Capaldo C. T., Babbin B. A., Williams I. R., Koval M., Peatman E., Campbell J. A., Dermody T. S., Nusrat A., Parkos C. A.. 2007. JAM-A regulates permeability and inflammation in the intestine in vivo. J. Exp. Med. 204:3067–3076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Q., Zhao X., Wang S., Zhou Z.. 2018. Letrozole induced low estrogen levels affected the expressions of duodenal and renal calcium-processing gene in laying hens. Gen. Comp. Endocrinol. 255:49–55. [DOI] [PubMed] [Google Scholar]
- Lim H. S., Namkung H., Paik I. K.. 2003. Effects of phytase supplementation on the performance, egg quality, and phosphorous excretion of laying hens fed different levels of dietary calcium and nonphytate phosphorous. Poult. Sci. 82:92–99. [DOI] [PubMed] [Google Scholar]
- Mongin P., Sauveur B.. 1974. Voluntary food and calcium intake by the laying hen. Br. Poult. Sci. 15:349–359. [DOI] [PubMed] [Google Scholar]
- Munro S. S., Kosin I. L.. 1943. Dramatic response of the chick oviduct to estrogen. Poult. Sci. 22:330–331. [Google Scholar]
- Nys Y., Bain M., Van Immerseel F.. 2011. Egg formation and chemistry. Pages 83–132 in Improving the Safety and Quality of Eggs and Egg Products. Woodhead Publishing, Sawston, Cambridge UK. [Google Scholar]
- Nys Y., Baker K., Bouillon R., Van Baelen H., Lawson D. E. M.. 1992. Regulation of calbindin D 28K and its mRNA in the intestine of the domestic hen. Gen. Comp. Endocrinol. 86:460–468. [DOI] [PubMed] [Google Scholar]
- Nys Y., Bouillon R., Van Baelen H., Williams J.. 1986a. Ontogeny and oestradiol dependence of vitamin D-binding protein blood levels in chickens. J. Endocrinol. 108:81–87. [DOI] [PubMed] [Google Scholar]
- Nys Y., Hincke M. T., Hernandez-Hernandez A., Rodriguez-Navarro A. B., Gomez-Morales J., Jonchère V., Garcia-Ruiz J. M., Gautron J.. 2010. Eggshell ultrastructure, properties and the process of mineralization: involvement of organic matrix in the eggshell fabric. INRA Prod. Anim. 23:143–154. [Google Scholar]
- Nys Y., Mongin P.. 1982. Transport of electrolytes and water in the upper jejunum of the fowl in vivo perfusion. Pflugers Arch. 392:251–256. [DOI] [PubMed] [Google Scholar]
- Nys Y., Parkes C., Thomasset M.. 1986. Effects of suppression and resumption of shell formation and parathyroid hormone on uterine calcium-binding protein, carbonic anhydrase activity, and intestinal calcium absorption in hens. Gen. Comp. Endocrinol. 64:293–299. [DOI] [PubMed] [Google Scholar]
- Nys Y., Le Roy N.. 2017. Calcium homeostasis and eggshell biomineralization in female chicken. Pages 361–382 in Vitamin D. 4th ed.Elsevier Inc, Stanford, California, USA. [Google Scholar]
- Olgun O., Aygun A.. 2016. Nutritional factors affecting the breaking strength of bone in laying hens. Worlds Poult. Sci. J. 72:821–832. [Google Scholar]
- Owsianik G., Talavera K., Voets T., Nilius B.. 2006. Permeation and selectivity of TRP channels. Annu. Rev. Physiol. 68:685–717. [DOI] [PubMed] [Google Scholar]
- Pearce J. 1974. A study of changes in the specific activities of enzymes of lipid and carbohydrate metabolism in the liver of the domestic fowl with the onset of sexual maturity. Int. J. Biochem. 5:457–462. [Google Scholar]
- Pfaffl M. W. 2001. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic. Acids. Res. 29:45e–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plaimast H., Kijparkorn S., Ittitanawong P.. 2015. Effects of vitamin D3 and calcium on productive performance, egg quality and vitamin D3 content in egg of second production cycle hens. Thai J. Vet. Med. 45:189–195. [Google Scholar]
- Proszkowiec-Weglarz M., Angel R.. 2013. Calcium and phosphorus metabolism in broilers: effect of homeostatic mechanism on calcium and phosphorus digestibility. J. Appl. Poult. Res. 22:609–627. [Google Scholar]
- Proszkowiec-Weglarz M., Schreier L. L., Miska K. B., Angel R., Kahl S., Russell B.. 2019. Effect of early neonatal development and delayed feeding post-hatch on jejunal and ileal calcium and phosphorus transporter genes expression in broiler chickens. Poult. Sci. 1:1–11. [DOI] [PubMed] [Google Scholar]
- Rousseau X., Valable A. S., Létourneau-Montminy M. P., Même N., Godet E., Magnin M., Nys Y., Duclos M. J., Narcy A.. 2016. Adaptive response of broilers to dietary phosphorus and calcium restrictions. Poult. Sci. 95:2849–2860. [DOI] [PubMed] [Google Scholar]
- Sauveur B., Mongin P.. 1983. Plasma inorganic phosphorus concentration during eggshell formation. II. - inverse relationships with intestinal calcium content and eggshell weight. Reprod. Nutr. Dev. 23:755–764. [Google Scholar]
- Spanaus K., von Eckardstein A.. 2017. Evaluation of two fully automated immunoassay based tests for the measurement of 1α, 25-dihydroxyvitamin D in human serum and comparison with LC-MS/MS. Clin. Chem. Lab. Med. 55:1305–1314. [DOI] [PubMed] [Google Scholar]
- Strehler E. E., Zacharias D. A.. 2001. Role of alternative splicing in generating isoform diversity among plasma membrane calcium pumps. Physiol. Rev. 81:21–50. [DOI] [PubMed] [Google Scholar]
- Suzuki T. 2013. Regulation of intestinal epithelial permeability by tight junctions. Cell. Mol. Life Sci. 70:631–659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandesompele J., De Preter K., Pattyn F., Poppe B., Van Roy N., De Paepe A., Speleman F.. 2002. Accurate normalization of real-time quantitative RT-PCR data. Genome Biol.. 3:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wasserman R. H., Taylor A. N.. 1966. Vitamin D3-induced calcium-binding protein in chick intestinal mucosa. Science. 152:791–793. [DOI] [PubMed] [Google Scholar]
- Whitehead C. C. 2004. Overview of bone biology in the egg-laying hen. Poult. Sci. 83:193–199. [DOI] [PubMed] [Google Scholar]
- Yang J. H., Hou J. F., Farquharson C., Zhou Z. L., Deng Y. F., Wang L., Yu Y.. 2011. Localisation and expression of TRPV6 in all intestinal segments and kidney of laying hens. Br. Poult. Sci. 52:507–516. [DOI] [PubMed] [Google Scholar]
- Ye J., Coulouris G., Zaretskaya I., Cutcutache I., Rozen S., Madden T. L.. 2012. Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics. 13:134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng J. 2013. Molecular mechanism of TRP channels. Compr. Physiol. 3:221–242. [DOI] [PMC free article] [PubMed] [Google Scholar]