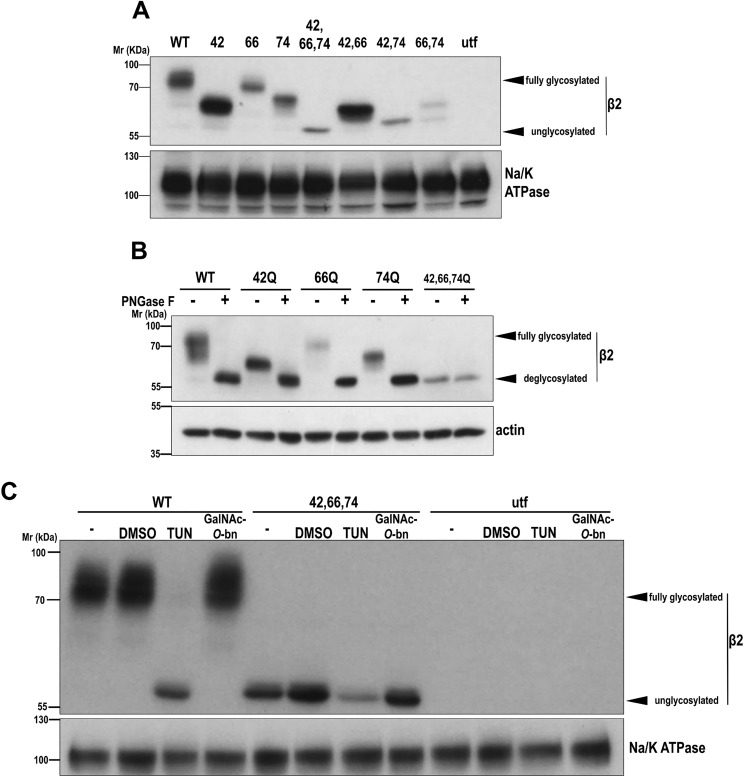
Figure 1.
β2 is N-glycosylated at positions Asn-42, Asn-66, and Asn-74. MDCK cells were transiently transfected with the SCN2B-yfp vector to express WT or partially or fully unglycosylated β2 or left untransfected (utf). A and B, cells were grown for 2 days in wells. Representative Western blots are shown with the same amount of protein lysate loaded into each lane. A, all glycosylation-defective mutants display increased electrophoretic mobility, with N42Q as the single mutant with the greatest change, and triple (fully) unglycosylated β2 showing complete shift. B, denatured protein from cell lysates was treated overnight at 37 °C with PNGase F to cleave off all N-glycans. C, cells were treated with TUN or with GalNAc-O-bn, to block N- or O-glycosylation, respectively, and grown for 1 day in wells; β2 WT remains unglycosylated only with TUN. DMSO, cells with the equivalent volume of solvent added; −, untreated cells. Blots for Na/K-ATPase or actin are included as loading controls. Molecular mass markers are in kDa.