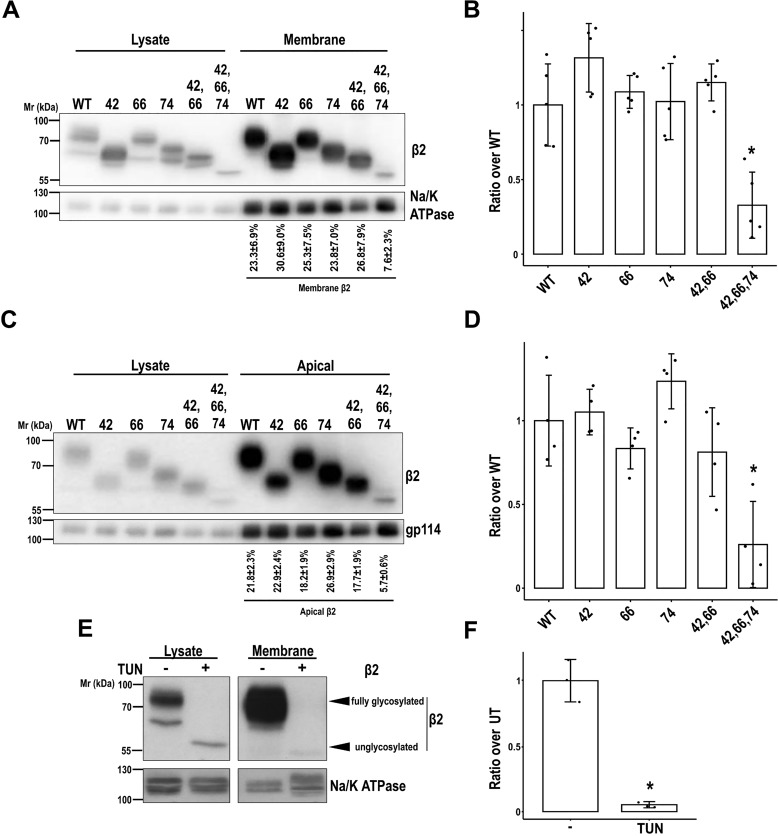
Figure 3.
N-Glycosylation is required for efficient cell surface localization of β2. MDCK cells were transiently transfected with the SCN2B-yfp vector to express WT or partially or fully unglycosylated β2. Cells were grown for 2 days in wells (A and B) or polarized in Transwells (C and D) and surface-biotinylated at 4 °C. Representative Western blots (A and C) and band quantitation (B and D) show that levels of fully unglycosylated β2 were reduced compared with the WT and partially glycosylated mutants in biotin-NeutrAvidin pulldowns, both in subconfluent and in polarized cells. One-way ANOVA with Tukey's HSD post hoc test highlighted these differences (B, *, p < 0.001; D, *, p < 0.05). A and C, values below blots show the percentage of each β2 variant at the surface over total cellular β2 protein. E and F, cells were treated with TUN to block N-glycosylation, grown for 1 day in wells, and surface-biotinylated at 4 °C (UT, untreated cells). Representative Western blots (E) and band quantitation (F) show the absence of unglycosylated β2 in pulldowns (two-tailed Student's t test shows significant differences; *, p = 0.009). The same amount of protein was used to process each lysate (∼130 μg), and the corresponding portion (nine-tenths) was subjected to overnight pulldown. Na/K-ATPase (A and E) or gp114 (C) were blotted as surface markers to correct for quantitations in pulldowns. All data are mean ± S.D. (error bars) (n ≥ 3). Molecular mass markers are in kDa. The dividing line in E separates different parts of the same blot (taken from the same exposure) conveniently put together for clear display.