Abstract
Background:
Hospital-acquired infections (HAI) contribute to prolonged hospital stays and account for a substantial economic burden to healthcare systems. Middle-income countries (MICs) experience a greater burden of HAI than developed countries. Evidence on the cost-effectiveness of interventions to reduce HAI is required to inform decision-making in these settings.
Aim:
To synthesise the evidence on cost-effectiveness as related to HAI interventions in MICs and to assess the quality of this evidence.
Methods:
A systematic review of published literature on the cost-effectiveness of interventions to reduce the incidence of HAI in MICs between 2000 and 2018 was conducted.
Results:
Six studies met the pre-determined inclusion criteria. The studies were from three countries: Thailand; India; and Vietnam. The evidence suggests that interventions to reduce HAI are cost-effective and, in most cases, cost-saving to healthcare systems. The quality of the reporting varied across studies.
Conclusions:
The implementation of HAI prevention interventions appears to be a high value use of resources in MICs. There is a need for further cost-effectiveness analyses in a wider range of MICs in order to confirm these findings. Improved standardisation and quality of reporting is required.
Keywords: Cost-effectiveness, economic evaluation, healthcare-associated infections, infection control, middle-income countries
Introduction
Hospital-acquired infections (HAIs) are potentially preventable adverse events that occur under medical care in a hospital or other healthcare facility. They include infections acquired in the healthcare facility but appearing after discharge, as well as occupational infections among healthcare workers (Sri Lanka College of Microbiologists, 2005; World Health Organization [WHO], 2002). HAIs are associated with increased risks of morbidity and mortality for affected patients (Jarvis, 1996; Stone et al., 2005). They also prolong hospital stays and generate substantial costs to the healthcare system (Cheng et al., 2018).
At any point in time, more than 1.4 million people worldwide suffer from infections acquired in hospitals (Sri Lanka College of Microbiologists, 2005). Due to the lack of reliable data in many countries, the exact prevalence of the problem is unknown (WHO, 2011). A WHO report on the global burden of endemic HAI worldwide indicated the burden of disease is substantially higher in low- and middle-income countries (WHO, 2011). A systematic review of the burden of HAIs in developing countries estimated a prevalence of 15.5 per 100 patients in 2010, substantially higher than those reported from Europe (7.1 per 100 patients) and the USA (4.5 per 100 patients) (Allegranzi et al., 2011).
There are many interventions aimed at preventing HAIs and, in turn, reducing excess mortality and costs (Graves et al., 2007). Economic evaluations, including cost-effectiveness analyses (CEA), are comparisons of the costs and health consequences of competing health programs or initiatives. They aim to assist decision makers in maximising health benefits from scarce resources (Martin, 2015). Cost utility analysis is a type of CEA where the health benefits are measured in terms of quality-adjusted life years (QALYs) (Drummond et al., 2015). There are two overarching approaches in conducting CEAs of healthcare interventions: (1) trial-based evaluation through a primary data collection exercise; and (2) modelling studies in which data on the costs and health benefits of an intervention may be obtained from multiple sources. Modelling studies are often more practical when longer-term impacts are of importance or where data are costly or impractical to collect within a single study.
Given the relatively high prevalence of HAI in middle-income countries, combined with limited resources for prevention in these settings, there is a need for evidence on the cost-effectiveness of strategies to reduce risks of HAI in this setting (Pittet et al., 2008; WHO, 2014). This study presents the first systematic review of cost-effectiveness analyses of HAI prevention strategies in MICs. We included both trial-based and model-based studies to synthesise the available evidence and assess the quality of this evidence.
Methods
A systematic review of studies reporting on the cost-effectiveness of HAI prevention interventions in MICs and published between 1 January 2000 and 24 August 2018 was conducted. We adopted the list of MICs as defined by the World Bank (World Bank, 2017) from the year 2000 up to 24 August 2018. The search strategy is shown in Box 1. Searches were conducted in MEDLINE, CHINAL, EMBASE and Cochrane Library databases. The inclusion and exclusion criteria are listed below.
Inclusion criteria
Had a full publication or manuscript for review
Written in the English language
Studies related to healthcare (hospital, outpatient, clinics or community-based clinics)
Conducted a full economic evaluation which valued both costs and health benefits
Evaluated at least a single intervention to reduce the incidence of HAI, compared to an existing practice comparator.
Conducted in lower and upper MICs with GNI per capita in the range of US$996–12,055 (World Bank, 2017)
Exclusion criteria
Studies with only cost analysis
Editorials, reviews or methods articles
Studies that did not adopt a comparator
Study settings not related to healthcare
Data extraction
The quality of the methods and reporting of the studies was assessed by considering data extracted against the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) reporting checklist (Supplementary File 1) (Husereau et al., 2013). Data fields included research question, setting and location, perspective, time horizon, discount rate, structure of the economic model if applied, study population, intervention and comparator, outcome measures, incremental cost-effectiveness ratio and sensitivity analysis method.
Each item in the CHEERS checklist was scored as having met the criteria in full (‘1’), not at all (‘0’) or not applicable (NA). When items partially met the criteria, they were scored as ‘0’; no partial scores were assigned to avoid introducing subjectivity.
The quality of the data sources used for clinical outcomes, costs and utilities was assessed through the modified hierarchies of data used to inform model parameters (Coyle and Lee, 2002), (Supplementary File 2). Quality was ranked in the range of 1–6 with the highest quality of evidence ranked 1. For each study, the highest level of evidence used for each parameter was recorded.
Results
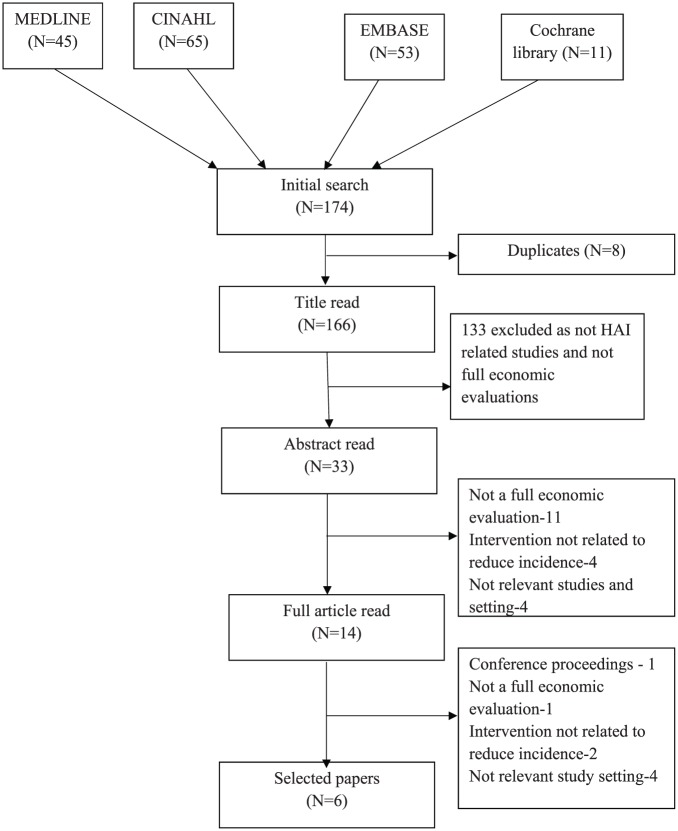
Figure 1 outlines the results of the search strategy, including how studies were excluded. Six articles met the pre-determined inclusion criteria from a total of 166 articles identified in the search (Luangasanatip et al., 2018; Maenthaisong et al., 2006; Rattanaumpawan and Thamlikitkul, 2017; Rosenthal et al., 2015; Singh et al., 2012; Thi Anh Thu et al., 2015).
Figure 1.
PRISMA flow chart for selection of articles for the review.
A summary of the key characteristics of the six included studies is provided in Table 1. Three studies were from Thailand (Luangasanatip et al., 2018; Maenthaisong et al., 2006; Rattanaumpawan and Thamlikitkul, 2017), two from India (Rosenthal et al., 2015; Singh et al., 2012) and one from Vietnam (Thi Anh Thu et al., 2015). The interventions evaluated included a hand hygiene programme in two studies (Luangasanatip et al., 2018; Thi Anh Thu et al., 2015), implementation of individualised bundling infection control measures (Rattanaumpawan and Thamlikitkul, 2017) and conducting a modular training programme (Singh et al., 2012). Regular standard infection control measures were considered as the baseline in each of these four studies. The use of chlorhexidine gluconate instead of povidone iodine as an antiseptic solution in catheter care was evaluated in the fifth study (Maenthaisong et al., 2006) and split septum and single use prefilled flushing device use for ICU patients with a centre line was compared with a three-way stopcock device in the sixth study (Rosenthal et al., 2015).
Table 1.
Summary of cost-effectiveness studies of interventions to reduce HAI incidence.
| Study and year | Country | Research question | Perspective | Time horizon | Discount rate | Structure of the economic model |
|---|---|---|---|---|---|---|
| Rattanaumpawan and Thamlikitkul (2017) | Thailand | How cost-effective is the application of individualised bundling infection control measures in a resource-limited setting | Provider’s perspective | Not mentioned explicitly; appears as the period of hospitalisation | Not considered | Simple decision tree model |
| Maenthaisong et al. (2006) | Thailand | How cost-effective is the usage of chlorhexidine gluconate as an antiseptic solution compared to povidone iodine in catheter care | Provider’s perspective | Period of hospitalisation | Not considered | Decision analytical model |
| Luangasanatip et al. (2018) | Thailand | 1. How cost-effective is the multimodal hospital intervention to
improve hand hygiene compliance at the baseline compliance level
of 10–< 20% in paediatric and adult ICUs 2. How cost-effective is the multimodal hospital intervention to improve hand hygiene compliance at the baseline compliance level of < 20–40% in paediatric and adult ICUs |
Provider’s perspective | Lifetime | 3% | Transmission dynamic models and decision analytic models |
| Thi Anh Thu et al. (2015) | Vietnam | How cost effective is the application of hand hygiene programme in reducing HAI incidence | Not explicitly stated; healthcare provider costs considered | Not mentioned explicitly; appears as the period of hospitalisation | Not considered | Not applied |
| Rosenthal et al. (2015) | India | How cost-effective is the split system and single use prefilled flushing device vs. three-way stopcock in reduction of central line associated bloodstream infection rates | Provider’s perspective | Not mentioned explicitly; appears as the period of hospitalisation | Not considered | Not applied |
| Singh et al. (2012) | India | What is the impact of modular training in infection control practices on process measures, outcomes and costs of four types of HAI | Not explicitly indicated; service provider and patient perspectives are presented | Not mentioned explicitly; appears as the period of hospitalisation | Not considered | Not applied |
ICU, intensive care unit; HAI, healthcare-acquired infections.
The study design and methods adopted in each paper, including the perspective time horizon, discount rate and model structure, are outlined in Table 1. The healthcare provider perspective was adopted in all six studies. Singh et al. (2012) considered the patient perspective in addition to the provider perspective. The only study to apply a long-term time horizon was Luangasanatip et al. (2018) which modelled cost-effectiveness over the lifetime and adopted a 3% discount rate. Maenthaisong et al. (2006) considered the period of hospitalisation as the time horizon. While not explicitly stated in the remaining studies, it can be inferred from the presentation of results that the period of hospitalisation was adopted as the time horizon. Among the six articles, three adopted a model-based economic evaluation study design. Decision analytic modelling was applied in all three modelling studies.
The cost-effectiveness outcome measures and results are presented in Table 2. Two studies adopted a cost utility analysis by estimating the QALYs gained as the measure of health benefit (Luangasanatip et al., 2018; Rosenthal et al., 2015). The remaining studies estimated health outcomes in natural units including lives saved, mortality rate reduction or reduction of HAI incidence (Maenthaisong et al., 2006; Rattanaumpawan and Thamlikitkul, 2017; Singh et al., 2012; Thi Anh Thu et al., 2015).
Table 2.
Results of cost-effectiveness studies of interventions to reduce HAI incidence included in the review.
| Study | Study population | Intervention | Comparator | Outcome measure | ICER | Sensitivity analysis method |
|---|---|---|---|---|---|---|
| Rattanaumpawan and Thamlikitkul (2017) | Hospitalised adults with length of stays ≤ 180 days | Individualised bundling infection control measures | Standard infection control care | Lives saved | Dominant* | OW, TW |
| Maenthaisong et al. (2006) | Hospitalised patients requiring either peripheral/central vascular catheters | Using chlorhexidine gluconate as an antiseptic solution in catheter care | Using povidone iodine as an antiseptic in catheter care | 1. Reduction of incidence of CRBSI 2. Reduction of incidence of death | Dominant* | OW, scenario analysis |
| Luangasanatip et al. (2018) | Patients of one adult ICU and a paediatric ICU | Multimodal hand hygiene interventions to increase compliance of healthcare worker | Standard infection control care | QALYs gained | Base line compliance < 20% in both paediatric and adult ICUs cost-effective; base line compliance 40% in both paediatric and adult ICUs not cost-effective | Scenario analysis |
| Thi Anh Thu et al. (2015) | Patients of ICUs and CCUs | Hand hygiene program | Standard infection control care | Reduction of incidence of HAI per 100 patients | Dominant* | Scenario analysis |
| Rosenthal et al. (2015) | All patients with central line of five medical and surgical ICUs of two tertiary care hospitals | Split septum and single use prefilled flushing device use for ICU patients with central line | Three-way stopcock on central line | QALYs gained | Dominant* | Not done |
| Singh et al. (2012) | All adult patients undergoing cardiovascular surgical procedures from Jan 2009 to Dec 2010 in adult cardiovascular surgical unit in India | Two step-by-step module training on infection control | Standard infection control care | Readmission rate; incidence rate of different HAI; surgical mortality | Dominant* strategy | Not done |
The intervention is cost-saving and improves health compared to the comparator.
ICER, incremental cost-effectiveness ratio; OW, one-way analysis; TW, two-way analysis; CRBSI, catheter-related bloodstream infections; ICU, intensive care unit; CCU, critical care unit; HAI, healthcare-acquired infections.
Five out of the six studies reported that the HAI interventions were ‘dominant’, indicating the intervention was more effective and less costly than the comparator. Luangasanatip et al. (2018) reported that the intervention was cost-effective only when the baseline hand hygiene compliance was < 20%. However, this study considered methicillin-resistant Staphylococcus Aureus-associated bloodstream infection as the only outcome measure and ignored impact of the intervention on other types of HAI. Sensitivity analysis to inform the robustness of results to uncertainty was performed in four studies (Luangasanatip et al., 2018; Maenthaisong et al., 2006; Rattanaumpawan and Thamlikitkul, 2017; Thi Anh Thu et al., 2015), with two of these studies limited to scenario analysis (Luangasanatip et al., 2018; Thi Anh Thu et al., 2015). None of the studies performed probabilistic sensitivity analysis in which the effects of joint parameter uncertainty are examined.
Assessment of the quality of the economic evaluation
The quality of the economic evaluations as evidenced by compliance with the CHEERS reporting guidelines varied between studies (Table 3). All studies identified and reported on the background and objectives, target populations, study setting, comparator, choice of health outcomes and study findings. However, an explanation of some of the key components of study design including time horizon, discount rate and currency were excluded from most studies. None of the studies reported on how heterogeneity or uncertainty were characterised.
Table 3.
Quality scoring using the CHEERS criteria.
| CHEERS criterion | Number of cost-effectiveness studies meeting criterion (n = 6) | |
|---|---|---|
| 1 | Title | 5 |
| 2 | Abstract | 5 |
| 3 | Background and objectives | 6 |
| 4 | Target population and subgroups | 6 |
| 5 | Setting and location | 6 |
| 6 | Study perspective | 5 |
| 7 | Comparators | 6 |
| 8 | Time horizon | 2 |
| 9 | Discount rate | 2 |
| 10 | Choice of health outcomes | 6 |
| 11 | Measurement of effectiveness | 4 |
| 12 | Measurement and valuation of preference-based outcomes | 2 |
| 13 | Estimating resources and costs | 3 |
| 14 | Currency, price date and conversion | 2 |
| 15 | Choice of model | 3 |
| 16 | Assumptions | 3 |
| 17 | Analytical methods | 2 |
| 18 | Study parameters | 5 |
| 19 | Incremental costs and outcomes | 5 |
| 20 | Characterising uncertainty | 0 |
| 21 | Characterising heterogeneity | 0 |
| 22 | Study findings, limitations, generalisability and current knowledge | 6 |
| 23 | Source of funding | 3 |
| 24 | Conflicts of interest | 2 |
Table 4 summarises the number of CHEERS criterion met per study. Two studies met < 50% of the CHEERS criteria (Singh et al., 2012; Thi Anh Thu et al., 2015) while the highest ranked study met 22 out of 24 criteria (Luangasanatip et al., 2018).
Table 4.
Number of CHEERS criteria met by each study.
| Study | Number of CHEERS criteria achieved (n = 24) |
|---|---|
| Rattanaumpawan and Thamlikitkul (2017) | 15 |
| Maenthaisong et al. (2006) | 18 |
| Luangasanatip et al. (2018) | 22 |
| Thi Anh Thu et al. (2015) | 10 |
| Rosenthal et al. (2015) | 12 |
| Singh et al. (2012) | 11 |
Assessment of the quality of the data used in evaluations
Three studies obtained the baseline parameters for the CEA from the published literature (Luangasanatip et al., 2018; Maenthaisong et al., 2006; Thi Anh Thu et al., 2015). Other studies sourced these data from randomised control trials (Rosenthal et al., 2015) or chart reviews from the study setting (Rattanaumpawan and Thamlikitkul, 2017; Singh et al., 2012). Baseline data were in high quality and assigned a category 1 in all studies, the highest level of data quality. The quality of cost data varied; these were gathered from administrative data bases or from published evidence from the same jurisdiction where the evaluation was conducted. For the two studies that measured QALYs, one used data derived from the same jurisdiction and it was assigned the highest level of data quality (Thi Anh Thu et al., 2015). For the studies that reported on natural outcomes such as reduction of incidents, hand hygiene compliance, reduction of mortality, these were measured directly from the same setting and were also assigned the highest level of quality.
Discussion
This is the first systematic review of cost-effectiveness studies of HAI prevention interventions in MICs. We identified and reviewed six papers published between 2000 and 2018.
A systematic audit of economic evidence linking HAIs and infection control measures between the years 1990 and 2000 identified 55 studies of relevance. Only 7% of these were from low- to middle-income countries (Stone et al., 2002). The audit reported a wide variation in both cost estimates and methods employed across studies; it concluded that increased standardisation and rigour are needed. A recent systematic review of economic evaluations of interventions for the prevention of HAIs internationally identified 27 studies published between 2009 and 2014 (Arefian et al., 2016). While the review found that prevention programs had mostly positive cost–benefit ratios, the quality of reporting was low. Only 14 articles reported more than half of CHEERS items appropriately and only four studies were considered high-quality.
Out of the six papers included in our review, three were trial-based and three were model-based. Some advantages of model-based studies over trial studies include the ability to account for long-term cost, QALY and mortality outcomes which cannot be seen in a period of a clinical trial, as well as the ability to more closely compare interventions to the status quo which often cannot be directly compared in a trial-based study, therefore increasing the generalisability of the study findings to the population. Model-based studies also enable the consideration of multiple competing interventions (Halton and Graves, 2007).
The perspective from which the benefits and costs are evaluated in an economic evaluation is important and should be explicitly stated. An evaluation containing all potential health effects and costs from a societal perspective is considered the gold standard (Jönsson, 2009) and was recommended by Stone et al. (2005) in their systematic review on economic evaluations of HAIs. With the exception of Singh et al. (2012) which considered both providers and patient perspectives, the studies identified in our review considered only the provider’s perspective. This ignores costs such as patient out-of-pocket expenditure as well as the productivity-related costs for both patients and society.
Most studies did not explicitly report on the time horizon of the analysis; however, on examination, it appears that the hospital length of stay was adopted. Only one study applied ‘lifetime’ as the time horizon. This finding is consistent with a 2005 systematic review of economic analyses of HAIs which reported that 99% of studies adopted a time horizon of ⩽ 1 year (Stone et al., 2005). This may be due to the perceived short-term nature of HAI. However, the inflammatory reactions following the short-term nosocomial infections may contribute to long-term complications after discharge. Selecting short time horizons and limited perspectives may lead to the omission of relevant costs and outcomes (Stone, 2009).
Stone et al. (2005) recommended that results of cost-effectiveness studies be reported in US dollars (USD) per QALY to enable comparisons between interventions and diseases across countries (Stone et al., 2005). While most studies in our review converted costs into USD, only two studies expressed the outcome in QALYs (Luangasanatip et al., 2018; Rosenthal et al., 2015). QALYs are a useful outcome for decision makers as it reflects both the quality and quantity of survival in a generic way that allows for comparisons between different health programmes and populations (Graves et al., 2007; Johannesson et al., 1996).
Sensitivity analysis is used to test results under plausible levels of uncertainty. Sensitivity analysis was performed in four studies but was limited to deterministic analyses including one-way, two-way and scenario analyses. It has been reported that probabilistic sensitivity analysis is the most useful method to use to prove the robustness of the results; this is an area for improvement in future studies (Cheng et al., 2018).
This study has some limitations. We found a relatively small number of studies that met the inclusion criteria, with half of these from the same country. The generalisability of our findings to the broader middle-income setting is therefore limited. While we comprehensively assessed the methods reported in the identified papers, we did not attempt to determine how studies were conducted in practice. Further, we did not attempt to verify the accuracy or appropriateness of the reported estimates or findings.
Conclusion
The implementation of HAI prevention interventions appears to be a high value use of the resources in the middle-income setting. There is a need further cost-effectiveness analyses in a wider range of MICs in order to confirm these findings. There is also a need for the quality of studies to be improved.
Supplemental Material
Supplemental material, Supplementary_material_1 for Cost-effectiveness of interventions to reduce the risk of healthcare-acquired infections in middle-income countries: a systematic review by Pushpa Udayangani Gamalathge, Sanjeewa Kularatna, Hannah E Carter, Sameera Senanayake and Nicholous Graves in Journal of Infection Prevention
Supplemental Material
Supplemental material, Supplementary_table_2 for Cost-effectiveness of interventions to reduce the risk of healthcare-acquired infections in middle-income countries: a systematic review by Pushpa Udayangani Gamalathge, Sanjeewa Kularatna, Hannah E Carter, Sameera Senanayake and Nicholous Graves in Journal of Infection Prevention
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
Peer review statement: Not commissioned; blind peer-reviewed.
ORCID iD: Pushpa Udayangani Gamalathge  https://orcid.org/0000-0002-3299-9774
https://orcid.org/0000-0002-3299-9774
Supplemental material: Supplemental material for this article is available online.
References
- Allegranzi B, Nejad SB, Combescure C, Graafmans W, Attar H, Donaldson L, Pittet D. (2011) Burden of endemic health-care-associated infection in developing countries: systematic review and meta-analysis. Lancet 377: 228–241. [DOI] [PubMed] [Google Scholar]
- Arefian H, Vogel M, Kwetkat A, Hartmann M. (2016) Economic evaluation of interventions for prevention of hospital acquired infections: a systematic review. PLoS One 11: e0146381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Q, Graves N, Pacella RE. (2018) Economic evaluations of guideline-based care for chronic wounds: a systematic review. Applied Health Economics and Health Policy 16: 633–651. [DOI] [PubMed] [Google Scholar]
- Coyle D, Lee KM. (2002) Evidence-based economic evaluation: how the use of different data sources can impact results. In: Donaldson M. (ed.) Evidence-Based Health Economics: From Effectiveness to Efficiency in Systematic Review. London: BMJ Books. [Google Scholar]
- Drummond MF, Sculpher MJ, Claxton K, Stoddart GL, Torrance GW. (2015) Methods for the Economic Evaluation of Health Care Programmes. 4th ed. Oxford: Oxford University Press. [Google Scholar]
- Graves N, Halton K, Lairson D. (2007) Economics and preventing hospital-acquired infection: broadening the perspective. Infection Control & Hospital Epidemiology 28: 178–184. [DOI] [PubMed] [Google Scholar]
- Halton K, Graves N. (2007) Economic evaluation and catheter-related bloodstream infections. Emerging Infectious Diseases 13: 815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, Augustovski F, Briggs AH, Mauskopf J, Loder E. and CHEERS Task Force. (2013) Consolidated health economic evaluation reporting standards (CHEERS) statement. Cost Effectiveness and Resource Allocation 11: 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarvis WR. (1996) Selected aspects of the socioeconomic impact of nosocomial infections: morbidity, mortality, cost, and prevention. Infection Control & Hospital Epidemiology 17: 552–557. [DOI] [PubMed] [Google Scholar]
- Johannesson M, Jönsson B, Karlsson G. (1996) Outcome measurement in economic evaluation. Health Economics 5: 279–296. [DOI] [PubMed] [Google Scholar]
- Jönsson B. (2009) Ten arguments for a societal perspective in the economic evaluation of medical innovations. European Journal of Health Economics 10: 357–359. [DOI] [PubMed] [Google Scholar]
- Luangasanatip N, Hongsuwan M, Lubell Y, Limmathurotsakul D, Srisamang P, Day NPJ, Graves N, Cooper BS. (2018) Cost-effectiveness of interventions to improve hand hygiene in healthcare workers in middle-income hospital settings: a model-based analysis. Journal of Hospital Infection 100: 165–175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maenthaisong R, Chaiyakunapruk N, Thamlikitkul V. (2006) Cost-effectiveness analysis of chlorhexidine gluconate compared with povidone-iodine solution for catheter-site care in Siriraj Hospital, Thailand. Journal of the Medical Association of Thailand = Chotmaihet Thangphaet 89: S94–S101. [PubMed] [Google Scholar]
- Martin E. (2015) Rationing in healthcare. In: Australian Healthcare and Hospitals Association (ed.) Issues Brief. Canberra, ACT: Deeble Institute. [Google Scholar]
- Pittet D, Allegranzi B, Storr J, Bagheri Nejad S, Dziekan G, Leotsakos A, Donaldson L. (2008) Infection control as a major World Health Organization priority for developing countries. Journal of Hospital Infection 68: 285–292. [DOI] [PubMed] [Google Scholar]
- Rattanaumpawan P, Thamlikitkul V. (2017) Epidemiology and economic impact of health care–associated infections and cost-effectiveness of infection control measures at a Thai university hospital. American Journal of Infection Control 45: 145–150. [DOI] [PubMed] [Google Scholar]
- Rosenthal VD, Udwadia FE, Kumar S, Poojary A, Sankar R, Orellano PW, Durgad S, Thulasiraman M, Bahirune S, Kumbhar S, Patil P. (2015) Clinical impact and cost-effectiveness of split-septum and single-use prefilled flushing device vs 3-way stopcock on central line–associated bloodstream infection rates in India: a randomized clinical trial conducted by the International Nosocomial Infection Control Consortium (INICC). American Journal of Infection Control 43: 1040–1045. [DOI] [PubMed] [Google Scholar]
- Singh S, Kumar RK, Sundaram KR, Kanjilal B, Nair P. (2012) Improving outcomes and reducing costs by modular training in infection control in a resource-limited setting. International Journal for Quality in Health Care 24: 641–648. [DOI] [PubMed] [Google Scholar]
- Sri Lanka College of Microbiologists. (2005) Hospital Infection Control Manual. Colombo: Sri Lanka College of Microbiologists. [Google Scholar]
- Stone PW. (2009) Economic burden of healthcare-associated infections: an American perspective. Expert Review of Pharmacoeconomics & Outcomes Research 9: 417–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stone PW, Braccia D, Larson E. (2005) Systematic review of economic analyses of health care-associated infections. American Journal of Infection Control 33: 501–509. [DOI] [PubMed] [Google Scholar]
- Stone PW, Larson E, Kawar LN. (2002) A systematic audit of economic evidence linking nosocomial infections and infection control interventions: 1990–2000. American Journal of Infection Control 30: 145–152. [DOI] [PubMed] [Google Scholar]
- Thi Anh, Thu L, Thi Hong Thoa V, Thi Van Trang D, Phuc Tien N, Thuy Van D, Thi Kim Anh L, Wertheim HF, Truong Son N. (2015) Cost-effectiveness of a hand hygiene program on health care-associated infections in intensive care patients at a tertiary care hospital in Vietnam. American Journal of Infection Control 43: e93–e99. [DOI] [PubMed] [Google Scholar]
- World Bank. (2017) List of Low, Lower-Middle, and Upper-Middle income economies according to the World Bank. 38th Annual Conference of the International Society for Clinical Biostatistics Vigo, Spain, 9-13 July 2017. [Google Scholar]
- World Health Organization. (2002) Prevention of hospital-acquired infections: a practical guide. 2nd ed. Geneva: WHO. [Google Scholar]
- World Health Organization. (2011) Report on the burden of endemic health care-associated infection worldwide. Geneva: WHO. [Google Scholar]
- World Health Organization. (2014) WHO Global Health Expenditure Atlas. Geneva: WHO. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, Supplementary_material_1 for Cost-effectiveness of interventions to reduce the risk of healthcare-acquired infections in middle-income countries: a systematic review by Pushpa Udayangani Gamalathge, Sanjeewa Kularatna, Hannah E Carter, Sameera Senanayake and Nicholous Graves in Journal of Infection Prevention
Supplemental material, Supplementary_table_2 for Cost-effectiveness of interventions to reduce the risk of healthcare-acquired infections in middle-income countries: a systematic review by Pushpa Udayangani Gamalathge, Sanjeewa Kularatna, Hannah E Carter, Sameera Senanayake and Nicholous Graves in Journal of Infection Prevention