Abstract
Cudraxanthone D (CD), derived from the root bark of Cudrania tricuspidata, is a natural xanthone compound. However, the biological activity of CD in terms of human metabolism has been barely reported to date. Autophagy is known as a self-degradation process related to cancer cell viability and metastasis. Herein, we investigated the effects of CD on human oral squamous cell carcinoma (OSCC) metastatic related cell phenotype. We confirmed that CD effectively decreased proliferation and viability in a time- and dose-dependent manner in human OSCC cells. In addition, OSCC cell migration, invasion, and EMT were inhibited by CD. To further determine the underlying mechanism of CD's inhibition of cell metastatic potential, we established the relationship between EMT and autophagy in OSCC cells. Thus, our findings indicated that CD inhibited the potential metastatic abilities of OSCC cells by attenuating autophagy.
1. Introduction
Autophagy is a biological process that protects against cellular homeostasis disruption from intracellular or environmental stresses, such as hypoxia, starvation, and cytochemism [1]. During autophagic processes, double-membrane vesicles, called autophagosomes, sequester cytoplasmic organelles and carry them to lysosomes. Lastly, autophagosome-fused lysosomes are degraded [2]. Autophagy is related to many pathological and physiological processes, such as apoptosis and cancer metastasis. The role of autophagy in tumor cells is known as a “dual-edged sword”—it may function as a tumor suppressor by protecting proteins from damage during the tumor formation stage, while in terms of tumor growth, autophagy might be a tumor promoter by distributing substrates for metabolic balance. Recently, much evidence of autophagy function in cancer has been reported such that it is key in the development of chemotherapeutic agents [3–6].
Tumor metastasis is divided into sequences of stages. Of the multistep processes, epithelial-mesenchymal transition (EMT) is concerned with local invasion [7], by which, when it is initiated, epithelial cells lose their properties and are transformed into different types of cells with mesenchymal properties, thereby increasing cell mobility and migration [8]. The interplay between autophagy and EMT has been thought for a long time to be connected. One study reported that increasing autophagy supports various cancer cells' EMT during spreading by upregulating cell motility [9–11]. On the other hand, there are data that indicate that autophagy prevents tumor cells from engaging in EMT by activating autophagic process and returning to the EMT phenotype. Synthetically, it is important to control autophagy and EMT interactions for regulation of tumor metastasis [12–14].
Cudraxanthone D (CD), a natural xanthone compound, is derived from the root bark of Cudrania tricuspidata distributed widely in Korea, Japan, and China [15]. Its compounds are used as traditional drugs for many diseases [16–19]. However, the relationship among oral squamous cell carcinoma (OSCC), autophagy, and EMT is unknown. So, in this study, we investigate what the role of autophagy is in OSCC cell metastatic cell phenotype and elucidate whether CD might be a strategically novel agent in oral cancer cell migration and invasion.
As is well known, autophagy and EMT are major biological processes in the occurrence and development of cancer, and there is a complex relationship between autophagy and EMT signaling pathways.
2. Materials and Methods
2.1. Reagents
Fetal bovine serum (FBS), Dulbecco's modified Eagle medium (DMEM), and DMEM : nutrient mixture F-12 (DMEM/F-12) were obtained from Thermo Fisher Scientific (Pittsburgh, USA). CD was purchased from ChemFace (Wuhan, China). 3-[4,5-Dimethylthiazol-2-yl]2,5-diphenyl tetrazolium bromide (MTT) and dimethyl sulfoxide (DMSO) were procured from Duchefa Biochemie (Haarlem, the Netherlands). Rabbit antibodies, E-cadherin, slug, snail, ATG5, beclin-1, and LC3B were purchased from Cell Signaling Technology (Beverly, USA). The anti-mouse HRP-conjugated secondary antibody and anti-rabbit HRP-conjugated secondary antibody were obtained from Enzo Biochem (Farmingdale, USA). Acridine orange and 3MA (3-methyladenine) were obtained from Sigma-Aldrich (St. Louis, USA).
2.2. Cell Culture
The Ca9-22, SCC25, and CAL27 cells were procured from America Type Culture Collection (Manassas, USA). HSC4 cells were provided by Professor Sung-Dae Cho of the Department of Oral Pathology, School of Dentistry, Chonbuk National University (Jeonju, Korea). Ca9-22 and CAL27 cells were cultured in DDMEM, Thermo Fisher Scientific (Pittsburgh, USA), while the SCC25 cells were cultured in DMEM/F-12. HSC4 cells were cultured in MEM with 10% FBS and 1% penicillin-streptomycin at 37°C in a humidified 5% carbon dioxide (CO2) atmosphere. CD was dissolved in DMSO at a stock solution of 10 mM and was kept at 4°C. The CD stock solution was diluted to marked concentrations with medium when we required.
2.3. Cell Viability Measurement
The cell viability of OSCC cells was determined using an MTT assay. Cells were cultured in 96-well plates and then incubated for different time periods in the presence of various concentrations of CD (0–100 μM). After terminating the treatment, the medium was removed and 100 μl of MTT (500 mg/mL) was added to each well. The cells were incubated for 3 h at 37°C to form formazan crystals. The formazan crystals were measured as described in the previous study [20].
2.4. Wound Healing Assay
A wound healing assay was performed to investigate the cell proliferation and migration ability of OSCC cells. Cells were seeded to roughly 80–90% confluence into six-well plates. A 1 mm pipette tip was used to formulate wounds, and debris was washed with PBS twice. Migrated cells into the wound area were visualized under an inverted microscope at 200x magnification (Olympus, Tokyo, Japan).
2.5. Migration and Invasion Assay
Cell invasion assay (transwell assay) and migration assay were conducted to examine the capacity of cell invasion and migration, as described previously. A transwell with an 8 μm pore polycarbonate membrane (Corning Costar, Cambridge, USA) was coated with 40 μl Matrigel at 200 μg/ml and incubated for 2 h. Next, OSCC cells were seeded and treated with 3MA, resveratrol, and CD. The upper chamber of the transwell was filled with serum-free medium and the lower chamber with 800 μl medium containing 10% FBS for 72 h of incubation at 37°C in a humidified 5% CO2 atmosphere. The cells were fixed in methanol and stained with hematoxylin for 30 min and were then counted under an inverted microscope (Olympus, Tokyo, Japan).
2.6. Immunofluorescent Staining
OSCC cells were cultured in DMEM with 3MA, resveratrol, and CD on a Lab-Tek™ II Chamber Slide (Nunc; Thermo Fisher Scientific, Rochester, USA). After 24 h, cells were stained with acridine orange (Sigma-Aldrich, St. Louis, USA). Fluorescent images were observed and analyzed as described in the previous study [20].
2.7. Western Blot Assay
Cells were harvested in the form of pellets, and then it was lysed in 150 μl RIPA buffer (300 mM NaCl, 50 mM Tris-Cl (pH 7.6), 0.5% Triton X-100, 2 mM PMSF, 2 μl/ml aprotinin, and 2 μl/ml leupeptin/protease inhibitor cocktail) and incubated at 4°C for 1 h. The lysate was centrifuged at 13,200 RPM for 30 minutes at 4°C. Protein quantification, electrophoresis, and detection of protein expression were performed in the same manner as described in the previous study [20].
2.8. Statistical Analysis
All data represent mean ± SD (standard deviation). Statistical analyses were conducted using one-way analysis of variance (ANOVA) followed by Dunnett's multiple comparison test. A difference of p value less than 0.05 was considered significant.
3. Results
3.1. Cudraxanthone D Affects Cell Viability in Human OSCC Cell Lines
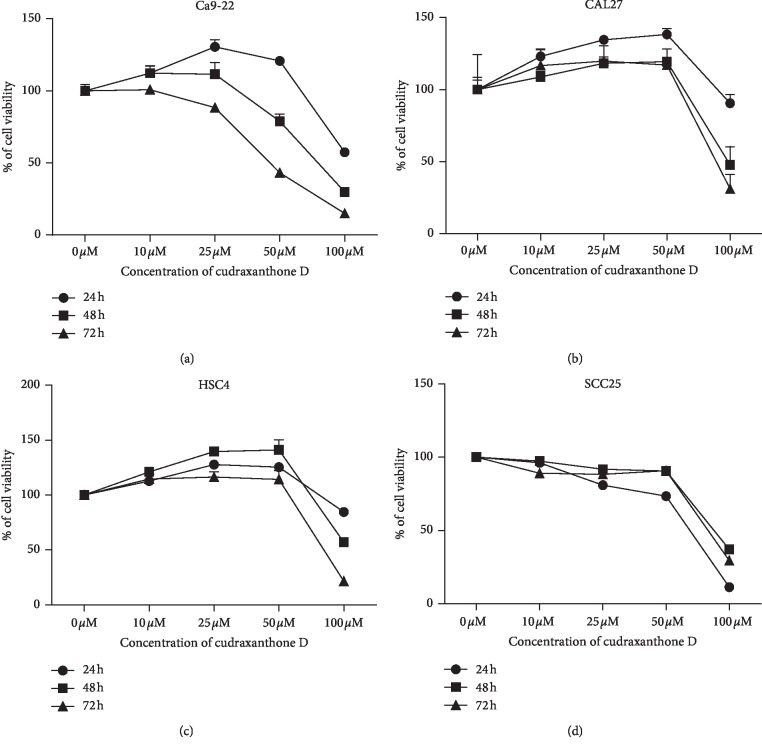
To choose proper concentrations of CD, human OSCC cell lines, specifically Ca9-22, CAL27, SCC25, and HSC4 cells, were cultured with 0–100 μM CD for 24–72 h. After treatment, cell viability was determined using an MTT assay. As shown in Figure 1, CD was cytotoxic to OSCC cells in a dose- and time-dependent manner. In particular, Ca9-22 and SCC25 cells were very sensitive to CD. So, these cells were selected and the subsequent experiments were carried out.
Figure 1.
The effect of CD on cell viability in OSCC cells. OSCC cells were treated with CD (0–100 μM) for 24–72 h. (a) Ca9-22, (b) CAL27, (c) HSC4, and (d) SCC25. Each experiment was performed in triplicate. Error bars denote mean ± SD (standard deviation). ∗p < 0.05 versus untreated cell, respectively.
3.2. Cudraxanthone D Suppresses Epithelial-Mesenchymal Transition in Ca9-22 and SCC25 Cells
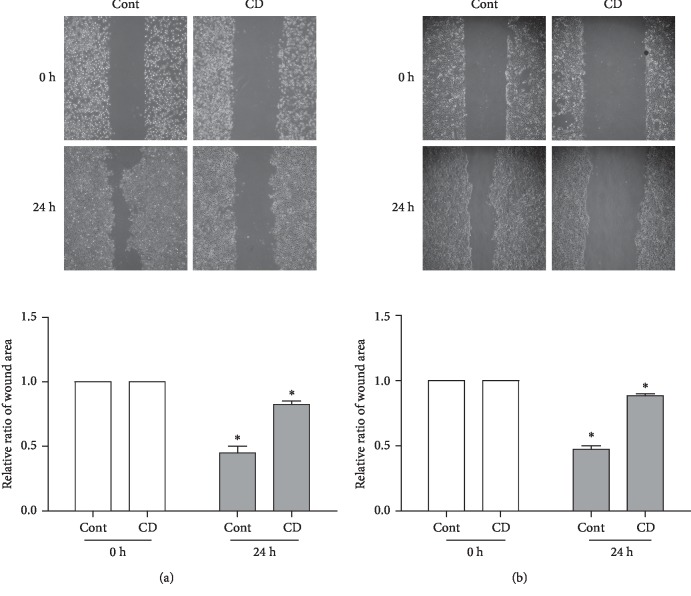
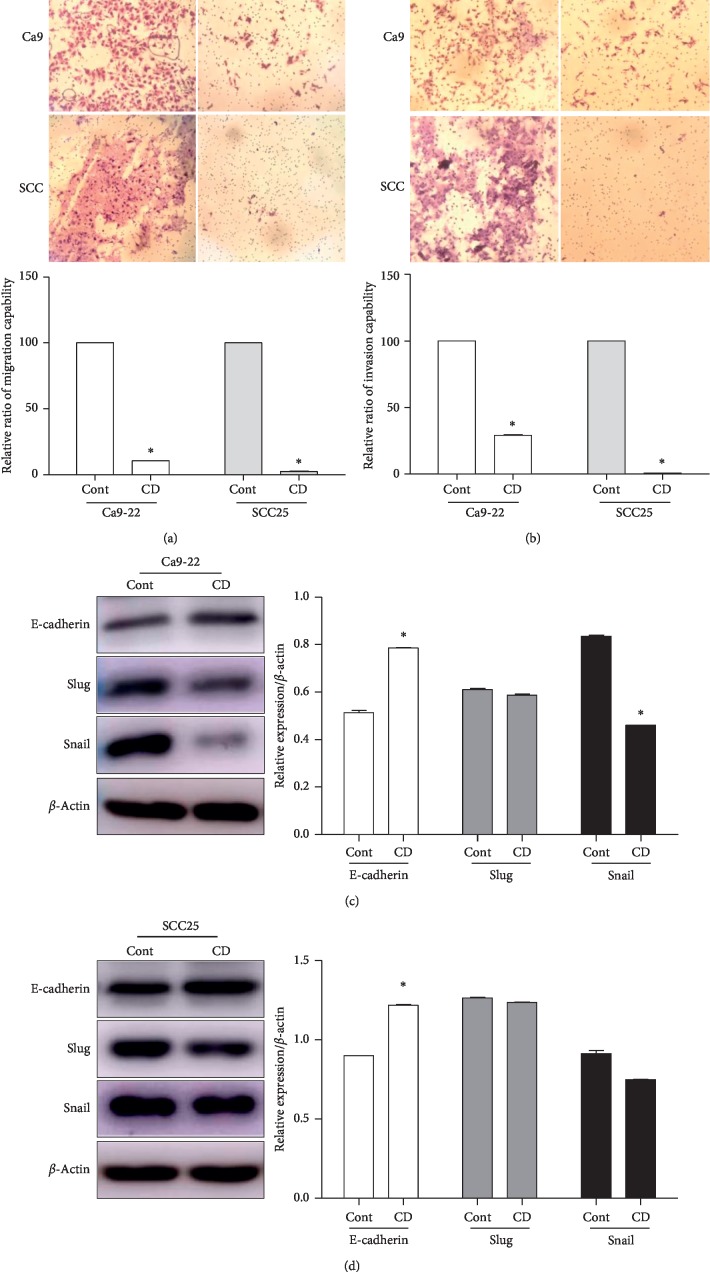
To determine whether CD affected OSCC cell cancer metastatic phenotype, we conducted a wound healing assay, transwell assay, and western blotting. Cell migration capabilities were investigated by the wound healing assay and transwell assay without Matrigel. The results demonstrated that the scratch wound area of the nontreated group was covered with proliferating Ca9-22 and SCC25 cells by approximately 55% while in the CD-treated group, the wound area covered only approximately 10% and 11% compared to prescratching, respectively (Figures 2(a) and 2(b)). In addition, we performed the transwell assay to verify migration capability in the absence of Matrigel, also known as a migration assay. First, Ca9-22 and SCC25 cell suspensions were located in the upper chamber with serum-starved media, while the lower chamber was filled with normal media. After 24 h, the nontreated group of Ca9-22 and SCC25 cells was found on the underside of the transwell filter. However, with the CD-treated group, there was a dramatic reduction in the migration capability of both cells by approximately 10% and 2%, respectively (Figure 3(a)). Similar results were found with the invasion assay, with applied Matrigel in the upper chamber. The CD-treated group exhibited inhibited invasion capabilities for both cells compared to the control groups (Figure 3(b)). Moreover, we confirmed the expression of EMT-associated proteins, such as E-cadherin, slug, and snail as shown in Figures 3(c) and 3(d). CD significantly upregulated E-cadherin, while slug expression was scarcely changed in both cells. Variation in snail expression was observed quite pronouncedly in Ca9-22. Collectively, these results suggested that CD effectively suppressed the EMT of OSCC cell lines.
Figure 2.
CD suppressed wound healing in OSCC cell lines. (a) Representative images and graphs of Ca9-22 cells' wound healing area treated with 50 μM CD for 24 h. (b) Representative images and graphs of SCC25 cells' wound healing area treated with 50 μM CD for 24 h. Error bars represent mean ± standard deviation (SD). ∗p < 0.05 versus 0 h respectively.
Figure 3.
CD inhibited EMT in human OSCC cell lines. Migration assay (a) and invasion assay (b) with Ca9-22 and SCC25 cells were conducted with a transwell chamber in the absence or presence of 50 μM CD for 24 h. Protein expression of EMT markers (E-cadherin, slug, and snail) was investigated after CD treatment in Ca9-22 (c) and SCC25 (d) cells. Relative protein expression levels were normalized to β-actin. Error bars represent mean ± standard deviation (SD). ∗p < 0.05 versus control, respectively.
3.3. Suppression of Resveratrol-Induced Autophagy by Cudraxanthone D Affected EMT in Ca9-22
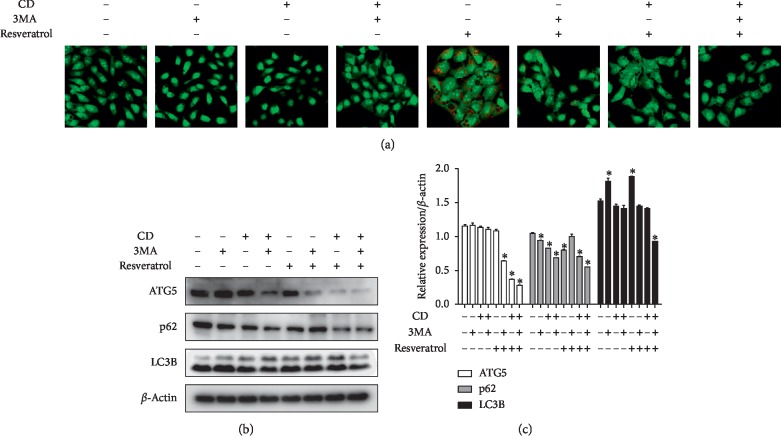
A variety of evidence indicated that autophagy induction is associated with tumor cell EMT in various cancers [5, 14]. Hence, in order to analyze the underlying mechanisms of CD-inhibited EMT, among diverse autophagy inducers, resveratrol has attracted attention, recently [21–24]. In our previous study, we confirmed that resveratrol induced autophagy in OSCC cell lines [25]. So, we employed resveratrol as an autophagy inducer and performed AO (acridine orange) staining and western blotting. Our findings demonstrated that resveratrol increased AO-positive autophagic vacuoles, and a typical autophagy inhibitor, 3MA, decreased the number of AO-positive cells. CD also remarkably diminished autophagic vacuoles compared to the resveratrol-treated group (Figure 4(a)). As shown in Figures 4(b) and 4(c), resveratrol upregulated autophagy-related proteins (ATG5, p62, and LC3B) inhibited by 3MA. CD also inhibited resveratrol-induced autophagy, with 3MA and CD cotreatment more effective against resveratrol-induced autophagy compared to single treatment. The aforementioned results suggested that CD acts as an autophagy inhibitor, while 3MA and CD dual treatment effectively suppressed resveratrol-induced autophagy.
Figure 4.
CD acts as an autophagy inhibitor in resveratrol-induced autophagy in Ca9-22 cells. (a) Cells were pretreated with 3MA (1 mM) for 2 h and then 25 μM resveratrol and 50 μM CD for 24 h. After treatment, AO-stained cells were observed under a confocal microscope. (b) Autophagy-related protein expression was confirmed by western blot. Each group was treated at the indicated concentrations and times. (c) Relative protein expression levels were normalized to β-actin. Error bars represent mean ± standard deviation (SD). ∗p < 0.05 versus control, respectively.
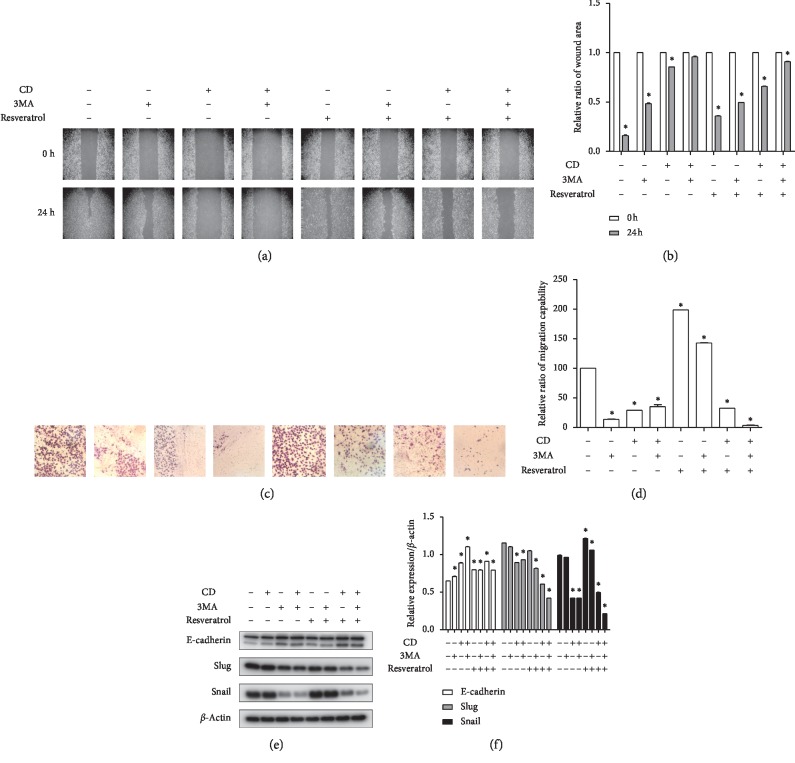
We hypothesize that CD and 3MA inhibited autophagy-affected migration and EMT in Ca9-22 cells in accordance with previous studies. To validate the hypothesis, wound healing assay and transwell assay (migration assay) were performed. The results indicated that the wound area of the resveratrol-treated group was almost filled with migrated cells. However, the wound area of the 3MA-pretreated and CD-treated groups was not reduced compared to control and resveratrol-administered groups (Figures 5(a) and 5(b)). Similarly, cell migration was induced by resveratrol treatment, and this induction was inhibited by 3MA and CD. 3MA and CD cotreatment significantly inhibited cell migration as shown in Figures 5(c) and 5(d). Next, to further elucidate the effect of CD-inhibited autophagy in the OSCC EMT, we analyzed the EMT-related proteins using a western blot assay. As shown in Figures 5(e) and 5(f), E-cadherin, an epithelial marker, was downregulated by resveratrol. Decreased E-cadherin expression was not regulated by 3MA, but E-cadherin expression increased with application of CD. Meanwhile, the expression levels of slug and snail, with mesenchymal properties, increased with resveratrol. Overexpression of slug and snail was inhibited by 3MA and CD. Additionally, dual treatment of 3MA and CD was more effective with respect to EMT protein expression inhibition than treatment alone. These data suggest that the application of CD effectively suppressed resveratrol-induced OSCC EMT and CD could be a novel autophagy inhibitor.
Figure 5.
CD and 3MA dual treatment suppressed autophagy-affected cell migration and EMT in Ca9-22 cells. (a) Cells were pretreated with 3MA (1 mM) for 2 h and then 25 μM resveratrol and 50 μM CD for 24 h. Wound area was observed under an inverted microscope by 200x magnification before or after treatment. (b) Relative ratio of healing wound area compared to before treatment. (c) Cells were treated with indicated concentrations and times, and migration capability was detected by transwell assay without Matrigel. (d) Relative ratio of migration capability compared to control. (e) The expression of EMT markers determined by western blot. (f) Relative protein expression levels compared to β-actin. Error bars represent mean ± standard deviation (SD). ∗p < 0.05 versus control, respectively.
4. Discussion
Over the past several decades, numerous dietary plant-extracted medicines have been used for human cancer. Although various evidence has indicated the advantages of many natural compounds, the OSCC five-year survival rate has been lower (approximately less than 50%) owing to many other reasons [26–28]. Among natural compounds, the Cudrania tricuspidata root bark isolated components, having a xanthone skeleton, have exhibited various biological activities [19, 29, 30]. Cudraxanthone H, with a prenylated xanthone, impacts OSCC growth inhibition and apoptosis [16]. Also, gerontoxanthone A, cudraflavone B, and gericudranin E obtained from the root bark of Cudrania tricuspidata have hepatoprotective effects [31]. Otherwise, plenty of xanthone series compounds have been used for various studies [26, 32–35]. Yet, there is no report on CD, which possesses a xanthone skeleton with a hydrogen-combined hydroxyl group and mutually ortho-located hydroxyl groups, elements of root bark of Cudrania tricuspidata in human OSCC [15]. In this study, for the first time, we focused on antimetastatic effects and chemotherapeutic potential of CD against OSCC.
First, we explored the cytotoxic effects of CD on four types of OSCC. Applications of 0 to 100 μM concentrations of CD inhibited cell growth in a dose- and time-dependent manner. Among them, Ca9-22 and SCC25 cells were more sensitive to CD than other cell lines. Recurrence and mortality rates of cancer mostly were regulated by metastasis and invasion of tumor cells, especially OSCC that chiefly metastasizes to the lymph nodes of cervical regions, fatal during prognosis [36–38]. Additional experiments such as wound healing assay, transwell assay, and western blot to detect expression levels of EMT-related protein indicated that CD remarkably suppressed cell migration, invasion, and EMT in Ca9-22 and SCC25 cells. To investigate the EMT inhibition mechanisms of CD, we concentrated on autophagy-regulated Ca9-22 cell EMT.
More research studies have reported that autophagy has a close correlation with cancer cell migration and invasion capability. A previous study suggested that compressive stress upregulates autophagy, and this status leads to enhanced migration of HeLa cells [39]. Further, Gao et al. reported that glycochenodeoxycholate supports HCC cell invasion and migration through autophagy activation [40]. Moreover, inhibition of autophagy with chloroquine attenuates metastatic ability in human non-small-cell lung adenocarcinoma A549 cells [5]. For these reasons, we formulated a hypothesis that CD-inhibited cell EMT ability is related to autophagy in OSCC cells. As an autophagy inducer, resveratrol is used on the basis of a number of earlier studies [21, 23, 41–43]. In our previous study, we evaluated the noncytotoxic concentration of resveratrol [25], where we induced autophagy by 25 μM of resveratrol.
As expected, resveratrol induced AO-positive autophagic vesicles and expressions of autophagy markers (ATG5, p62, and LC3B). 3MA, known as a typical autophagy inhibitor, suppressed resveratrol-induced autophagic activity. CD also inhibited resveratrol-induced autophagic actions. In order to elucidate the mechanism between resveratrol-induced autophagy and OSCC migration as well as invasion capability, we performed wound healing assay, transwell assay without Matrigel, and a western blot assay (EMT-related factors) under the same conditions as in Figure 4. The results demonstrated that autophagy induction enhanced cell migration ability and EMT factor expressions in Ca9-22 cells. Inhibition of autophagy by 3MA decreased Ca9-22 cell motility and EMT-related protein expression compared to the resveratrol-treated group. Suppression of EMT by CD was more effective than that by 3MA. Additionally, 3MA and CD cotreatment dramatically inhibited EMT compared with a single treatment. These results suggested that CD could be an EMT suppressor through autophagy inhibition.
5. Conclusion
The present study demonstrated that the molecular mechanism underlying the chemotherapeutic properties is that CD could inhibit metastatic related phenotype in human OSCC cells by inhibiting autophagy. We are suggesting that CD has sufficient potential for development as a new anticancer drug.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) Grant Funded by the Korea Government (nos. NRF-2017R1C1B5018034 and NRF-2018R1D1A1B07047739).
Contributor Information
Bong-Soo Park, Email: parkbs@pusan.ac.kr.
In-Ryoung Kim, Email: biowool@pusan.ac.kr.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Authors' Contributions
Bong-Soo Park and In-Ryoung Kim contributed equally to this study. All authors contributed to this study. Bong-Soo Park and In-Ryoung Kim conceived and designed the experiments. Su-Bin Yu and Hae-Mi Kang performed the experiments. Su-Bin Yu, Bong-Soo Park, and In-Ryoung Kim analyzed the data and wrote the paper. Dan-Bi Park and Su-Bin Yu contributed reagents/materials/analysis tools.
References
- 1.Wang Y., Xiong H., Liu D., et al. Autophagy inhibition specifically promotes epithelial-mesenchymal transition and invasion in RAS-mutated cancer cells. Autophagy. 2019;15(5):886–899. doi: 10.1080/15548627.2019.1569912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhang Y., Fan Y., Huang S., et al. Thymoquinone inhibits the metastasis of renal cell cancer cells by inducing autophagy via AMPK/mTOR signaling pathway. Cancer Science. 2018;109(12):3865–3873. doi: 10.1111/cas.13808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wei R., Xiao Y., Song Y., Yuan H., Luo J., Xu W. FAT4 regulates the EMT and autophagy in colorectal cancer cells in part via the PI3K-AKT signaling axis. Journal of Experimental & Clinical Cancer Research. 2019;38(1):p. 112. doi: 10.1186/s13046-019-1043-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.White E. Deconvoluting the context-dependent role for autophagy in cancer. Nature Reviews Cancer. 2012;12(6):401–410. doi: 10.1038/nrc3262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Datta S., Choudhury D., Das A., et al. Autophagy inhibition with chloroquine reverts paclitaxel resistance and attenuates metastatic potential in human nonsmall lung adenocarcinoma A549 cells via ROS mediated modulation of β-catenin pathway. Apoptosis. 2019;24(5-6):414–433. doi: 10.1007/s10495-019-01526-y. [DOI] [PubMed] [Google Scholar]
- 6.Chen H. T., Liu H., Mao M. J., et al. Crosstalk between autophagy and epithelial-mesenchymal transition and its application in cancer therapy. Molecular Cancer. 2019;18(1) doi: 10.1186/s12943-019-1030-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mowers E. E., Sharifi M. N., Macleod K. F. Autophagy in cancer metastasis. Oncogene. 2017;36(12):1619–1630. doi: 10.1038/onc.2016.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gugnoni M., Sancisi V., Gandolfi G., et al. Cadherin-6 promotes EMT and cancer metastasis by restraining autophagy. Oncogene. 2017;36(5):667–677. doi: 10.1038/onc.2016.237. [DOI] [PubMed] [Google Scholar]
- 9.Gugnoni M., Sancisi V., Manzotti G., Gandolfi G., Ciarrocchi A. Autophagy and epithelial-mesenchymal transition: an intricate interplay in cancer. Cell Death & Disease. 2016;7(12) doi: 10.1038/cddis.2016.415.e2520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Li J., Yang B., Zhou Q., et al. Autophagy promotes hepatocellular carcinoma cell invasion through activation of epithelial-mesenchymal transition. Carcinogenesis. 2013;34(6):1343–1351. doi: 10.1093/carcin/bgt063. [DOI] [PubMed] [Google Scholar]
- 11.Galavotti S., Bartesaghi S., Faccenda D., et al. The autophagy-associated factors DRAM1 and p62 regulate cell migration and invasion in glioblastoma stem cells. Oncogene. 2013;32(6):699–712. doi: 10.1038/onc.2012.111. [DOI] [PubMed] [Google Scholar]
- 12.Lv Q., Wang W., Xue J., et al. DEDD interacts with PI3KC3 to activate autophagy and attenuate epithelial-mesenchymal transition in human breast cancer. Cancer Research. 2012;72(13):3238–3250. doi: 10.1158/0008-5472.can-11-3832. [DOI] [PubMed] [Google Scholar]
- 13.Li G., Li C.-X., Xia M., et al. Enhanced epithelial-to-mesenchymal transition associated with lysosome dysfunction in podocytes: role of p62/Sequestosome 1 as a signaling hub. Cellular Physiology and Biochemistry. 2015;35(5):1773–1786. doi: 10.1159/000373989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Catalano M., D’Alessandro G., Lepore F., et al. Autophagy induction impairs migration and invasion by reversing EMT in glioblastoma cells. Molecular Oncology. 2015;9(8):1612–1625. doi: 10.1016/j.molonc.2015.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fujimoto T., Hano Y., Nomura T., Uzawa J. Components of root bark of Cudrania tricuspidata 2. Structures of two new isoprenylated flavones, cudraflavones a and B. Planta Medica. 1984;50(2):161–163. doi: 10.1055/s-2007-969660. [DOI] [PubMed] [Google Scholar]
- 16.Lee H.-J., Jue S.-S., Kang S.-K., Bae W.-J., Kim Y.-C., Kim E.-C. Cudraxanthone H induces growth inhibition and apoptosis in oral cancer cells via NF-κB and PIN1 pathways. The American Journal of Chinese Medicine. 2015;43(7):1439–1452. doi: 10.1142/s0192415x15500810. [DOI] [PubMed] [Google Scholar]
- 17.Shin S. Y., Yong Y., Kim C. G., Lee Y. H., Lim Y. Deoxypodophyllotoxin induces G2/M cell cycle arrest and apoptosis in HeLa cells. Cancer Letters. 2010;287(2):231–239. doi: 10.1016/j.canlet.2009.06.019. [DOI] [PubMed] [Google Scholar]
- 18.Jeong G.-S., An R.-B., Pae H.-O., et al. Cudratricusxanthone A protects mouse hippocampal cells against glutamate-induced neurotoxicity via the induction of heme oxygenase-1. Planta Medica. 2008;74(11):1368–1373. doi: 10.1055/s-2008-1081315. [DOI] [PubMed] [Google Scholar]
- 19.Park K. H., Park Y.-D., Han J.-M., et al. Anti-atherosclerotic and anti-inflammatory activities of catecholic xanthones and flavonoids isolated from Cudrania tricuspidata. Bioorganic & Medicinal Chemistry Letters. 2006;16(21):5580–5583. doi: 10.1016/j.bmcl.2006.08.032. [DOI] [PubMed] [Google Scholar]
- 20.Yu S.-B., Kim H.-J., Kang H.-M., Park B.-S., Lee J.-H., Kim I.-R. Cordycepin accelerates osteoblast mineralization and attenuates osteoclast differentiation in vitro. Evidence-Based Complementary and Alternative Medicine. 2018;2018:10. doi: 10.1155/2018/5892957.5892957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wang J., Li J., Cao N., Li Z., Han J., Li L. Resveratrol, an activator of SIRT1, induces protective autophagy in non-small-cell lung cancer via inhibiting Akt/mTOR and activating p38-MAPK. OncoTargets and Therapy. 2018;11:7777–7786. doi: 10.2147/ott.s159095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang B., Yin X., Sui S. Resveratrol inhibited the progression of human hepatocellular carcinoma by inducing autophagy via regulating p53 and the phosphoinositide 3-kinase/protein kinase B pathway. Oncology Reports. 2018;40(5):2758–2765. doi: 10.3892/or.2018.6648. [DOI] [PubMed] [Google Scholar]
- 23.Fan Y., Chiu J. F., Liu J., et al. Resveratrol induces autophagy-dependent apoptosis in HL-60 cells. BMC Cancer. 2018;18(1):p. 581. doi: 10.1186/s12885-018-4504-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gao J. L., Zhang Q. Y., Song L. Resveratrol enhances matrix biosynthesis of nucleus pulposus cells through activating autophagy via the PI3K/Akt pathway under oxidative damage. Bioscience Reports. 2018;38(4) doi: 10.1042/bsr20180544. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 25.Kim S.-E., Shin S.-H., Lee J.-Y., et al. Resveratrol induces mitochondrial apoptosis and inhibits epithelial-mesenchymal transition in oral squamous cell carcinoma cells. Nutrition and Cancer. 2018;70(1):125–135. doi: 10.1080/01635581.2018.1397708. [DOI] [PubMed] [Google Scholar]
- 26.Kwak H. H., Kim I. R., Kim H. J., Park B. S., Yu S. B. α-mangostin induces apoptosis and cell cycle arrest in oral squamous cell carcinoma cell. Evidence-Based Complementary and Alternative Medicine. 2016;2016:10. doi: 10.1155/2016/5352412.5352412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bu W., Wang Z., Meng L., et al. Disulfiram inhibits epithelial-mesenchymal transition through TGF-β-ERK-Snail pathway independently of Smad4 to decrease oral squamous cell carcinoma metastasis. Cancer Management and Research. 2019;11:4617–4618. doi: 10.2147/cmar.s214609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.He Y., Tai S., Deng M., et al. Metformin and 4SC-202 synergistically promote intrinsic cell apoptosis by accelerating ∆Np63 ubiquitination and degradation in oral squamous cell carcinoma. Cancer Medicine. 2019;8(7):3479–3490. doi: 10.1002/cam4.2206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Seo W. G., Pae H. O., Oh G. S., et al. Ethyl acetate extract of the stem bark of Cudrania tricuspidata induces apoptosis in human leukemia HL-60 cells. The American Journal of Chinese Medicine. 2001;29(2):313–320. doi: 10.1142/s0192415x01000332. [DOI] [PubMed] [Google Scholar]
- 30.Zou Y. S., Hou A. J., Zhu G. F., Chen Y. F., Sun H. D., Zhao Q. S. Cytotoxic isoprenylated xanthones from Cudrania tricuspidata. Bioorganic & Medicinal Chemistry. 2004;12(8):1947–1953. doi: 10.1016/j.bmc.2004.01.030. [DOI] [PubMed] [Google Scholar]
- 31.An R. B., Sohn D. H., Kim Y. C. Hepatoprotective compounds of the roots of Cudrania tricuspidata on tacrine-induced cytotoxicity in Hep G2 cells. Biological & Pharmaceutical Bulletin. 2006;29(4):838–840. doi: 10.1248/bpb.29.838. [DOI] [PubMed] [Google Scholar]
- 32.Hwang J. H., Hong S. S., Han X. H., et al. Prenylated xanthones from the root bark of Cudrania tricuspidata. Journal of Natural Products. 2007;70(7):1207–1209. doi: 10.1021/np070059k. [DOI] [PubMed] [Google Scholar]
- 33.Nakashima K., Tanaka T., Murata H., Kaburagi K., Inoue M. Xanthones from the roots of Maclura cochinchinensis var. gerontogea and their retinoic acid receptor-α agonistic activity. Bioorganic & Medicinal Chemistry Letters. 2015;25(9):1998–2001. doi: 10.1016/j.bmcl.2015.02.075. [DOI] [PubMed] [Google Scholar]
- 34.Park S. J., Park B. S., Yu S. B., Kang H. M., Kim H. J., Kim I. R. Induction of apoptosis and inhibition of epithelial mesenchymal transition by α-mangostin in MG-63 cell lines. Evidence-Based Complementary and Alternative Medicine. 2018;2018:10. doi: 10.1155/2018/3985082.3985082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fukai T., Oku Y., Hou A. J., Yonekawa M., Terada S. Antimicrobial activity of hydrophobic xanthones from Cudrania cochinchinensis against Bacillus subtilis and methicillin-resistant Staphylococcus aureus. Chemistry & Biodiversity. 2004;1(9):1385–1390. doi: 10.1002/cbdv.200490101. [DOI] [PubMed] [Google Scholar]
- 36.Tang D., Tao D., Fang Y., Deng C., Xu Q., Zhou J. TNF-α promotes invasion and metastasis via NF-κB pathway in oral squamous cell carcinoma. Medical Science Monitor Basic Research. 2017;23:141–149. doi: 10.12659/msmbr.903910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang M., Niu W., Qi M., et al. Nicotine promotes cervical metastasis of oral cancer by regulating peroxiredoxin 1 and epithelial-mesenchymal transition in mice. OncoTargets and Therapy. 2019;12:3327–3338. doi: 10.2147/ott.s194129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen F., Qi S., Zhang X., Wu J., Yang X., Wang R. lncRNA PLAC2 activated by H3K27 acetylation promotes cell proliferation and invasion via the activation of Wnt/β-catenin pathway in oral squamous cell carcinoma. International Journal of Oncology. 2019;54(4):1183–1194. doi: 10.3892/ijo.2019.4707. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 39.Das J., Agarwal T., Chakraborty S., Maiti T. K. Compressive stress-induced autophagy promotes invasion of HeLa cells by facilitating protein turnover in vitro. Experimental Cell Research. 2019;381(2):201–207. doi: 10.1016/j.yexcr.2019.04.037. [DOI] [PubMed] [Google Scholar]
- 40.Gao L., Lv G., Li R., et al. Glycochenodeoxycholate promotes hepatocellular carcinoma invasion and migration by AMPK/mTOR dependent autophagy activation. Cancer Letters. 2019;454:215–223. doi: 10.1016/j.canlet.2019.04.009. [DOI] [PubMed] [Google Scholar]
- 41.Chang C. H., Lee C. Y., Lu C. C., et al. Resveratrol-induced autophagy and apoptosis in cisplatin-resistant human oral cancer CAR cells: a key role of AMPK and Akt/mTOR signaling. International Journal of Oncology. 2017;50(3):873–882. doi: 10.3892/ijo.2017.3866. [DOI] [PubMed] [Google Scholar]
- 42.Du L., Chen E., Wu T., Ruan Y., Wu S. Resveratrol attenuates hydrogen peroxide-induced aging through upregulation of autophagy in human umbilical vein endothelial cells. Drug Design, Development and Therapy. 2019;13:747–755. doi: 10.2147/dddt.s179894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhao H., Chen S., Gao K., et al. Resveratrol protects against spinal cord injury by activating autophagy and inhibiting apoptosis mediated by the SIRT1/AMPK signaling pathway. Neuroscience. 2017;348:241–251. doi: 10.1016/j.neuroscience.2017.02.027. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.