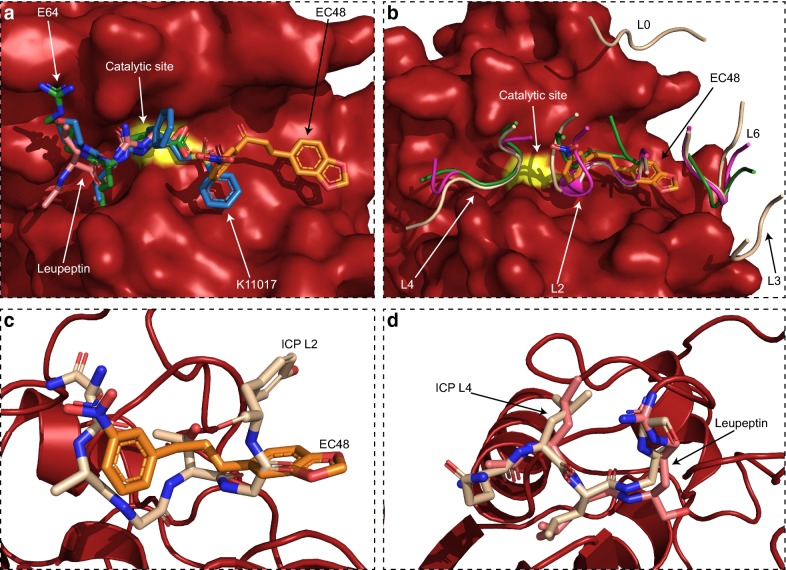
Fig. 3.
Comparison of EC48 binding to other FP2 inhibitors. a Superposition of small molecule inhibitors resolved in complex with Plasmodium parasite falcipain proteases on the FP2 structure of this study (surface representation, red; catalytic site C285 coloured yellow). Inhibitors are shown as sticks. EC48 (orange) from the present study; K11017 (blue) from PDB 3BWK; leupeptin (pink) from PDB 3BPM; E64 (green) from PDB 3BPF. b Superposition of protein-based inhibitors of falcipain (schematic representation) on the FP2 structure of this study (surface, as in panel a). Only the falcipain-interacting loops of these inhibitors are shown for clarity. Loops are numbered according to ICP nomenclature [27] and coloured light brown (ICP; PDB 3PNR), magenta (chagasin; PDB 2OUL) or green (cystatin; PDB 1YVB). EC48 (orange, sticks) from the present study is shown as reference. c Overlay of the EC48 (orange, sticks) binding position on FP2 with that of L2 from ICP (light brown, sticks). d Similar overlay of the leupeptin (pink) and ICP L4 (light brown) binding positions